A stable carbon atom has the following parts

A stable carbon atom has the following parts: – 6 protons – 6 neutrons – 6 electrons Describe the location of each of these parts in a carbon atom. Seconds Left: 140 120 130 30 40 50 60 70 10 2 3 4 5 6 7 8 9 0 80 180 20 160 90 150 170 100 110 1

Protons and neutrons are found in the nucleus of the atom, at the very center. Electrons are found in an electron cloud that surrounds the nucleus.

Describe three kinds of particles found in atoms. Where are they located & what are their charges? This screen will disappear in 3 minutes. 140 120 130 30 40 50 60 70 10 320 4 5 6 7 8 9 180 20 160 90 80 170 150 100 110 1 Seconds Remaining.

• Protons – in the nucleus, positive charge • Neutrons – in the nucleus, no charge • Electrons – outside the nucleus, negative charge

An electrically neutral atom undergoes a change to become negatively charged. The atom's atomic mass decreases. A student explains that the atom most likely lost a neutron. – Describe one strength and one weakness of the student's explanation. Seconds left: 140 120 130 30 40 50 60 70 10 080 2 3 4 5 6 7 8 9 180 20 160 90

One strength of the explanation is that an atom's mass consists mostly of the mass of its protons and neutrons. Electrons have very little mass. So, the atom must have lost either a proton or neutron. One weakness, however, is that neutrons have no charge. So, removing the neutron should not affect the atom's charge. It is more likely that a proton was removed.
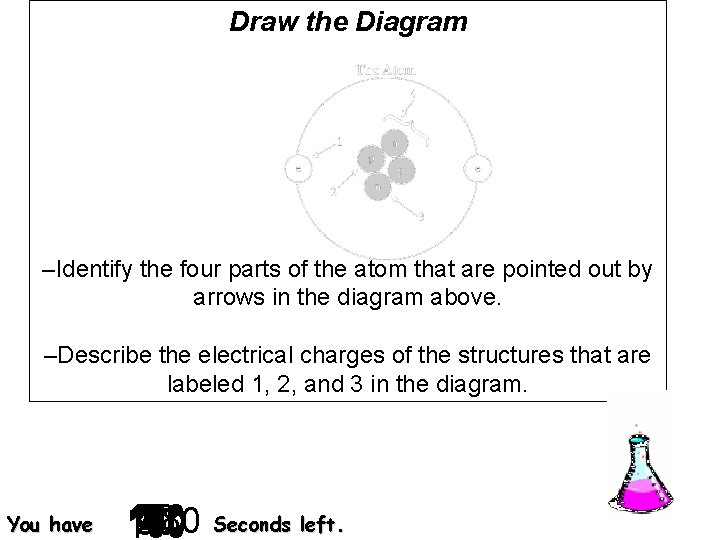
Draw the Diagram –Identify the four parts of the atom that are pointed out by arrows in the diagram above. –Describe the electrical charges of the structures that are labeled 1, 2, and 3 in the diagram. You have 140 120 130 30 40 50 60 70 10 2 3 4 5 6 7 8 9 0 180 20 160 90 150 170 100 110 180 Seconds left.

Number 1 is the electron, 2 is the proton, 3 is the neutron, and 4 is the nucleus. The electron is negative. The proton is positive. The neutron is neutral.
Define these terms: Atom Isotope Ion Time left: 140 120 130 30 40 50 60 70 10 2 3 4 5 6 7 8 9 0 180 20 160 90 170 150 100 110 180 Seconds

ATOM – the smallest part of matter that has the properties of the matter ISOTOPE – different forms of the same atom; they have the same numbers of protons, but different numbers of neutrons ION – an atom that has lost or gained electrons

An atom consists of 7 neutrons, 7 electrons, and a number of protons. The atom has a charge of -1. What is its mass number? Seconds Left: 140 120 130 30 40 50 60 70 10 2 3 4 5 6 7 8 9 0 180 20 160 90 150 170 100 110 180

The atom has a mass number of 13 • To find the mass number we have to know the # of protons & the # of neutrons. – We know there are 7 neutrons; ? ? ? protons • An atom with a charge of -1 has gained an electron (added a – charge) • Atoms begin w/ = numbers of protons & electrons, so the atom began with 6 electrons & 6 protons. • 6 protons + 7 neutrons = • a mass # of 13

What are the biggest parts of an atom? Seconds Remaining: 120 140 130 70 60 50 40 30 10 9 8 7 6 5 4 3 2 0 180 20 160 90 80 170 150 110 100 180

Protons & Neutrons

A stable helium atom contains 2 protons, 2 neutrons, and 2 electrons. – Draw and label a diagram of a stable helium atom. Be sure to include the protons, neutrons, electrons, and nucleus of the atom in your diagram. Seconds Left: 140 120 130 30 40 50 60 70 10 2 3 4 5 6 7 8 9 0 180 80 20 160 90 150 170 100 110 1
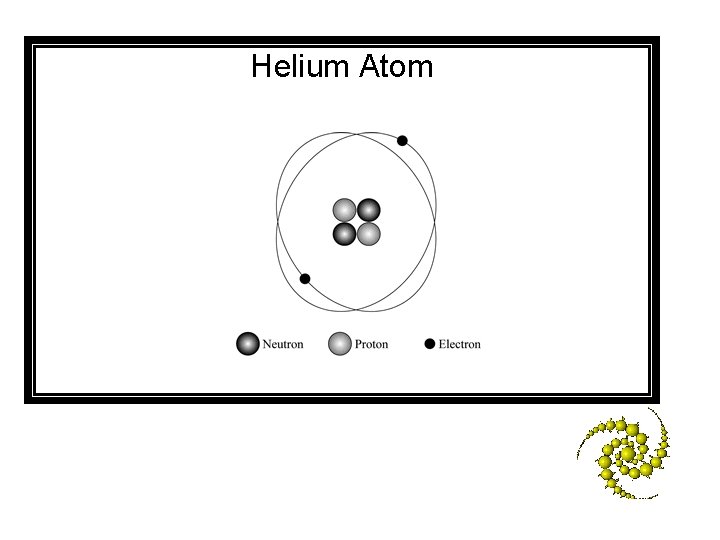
Helium Atom
- Slides: 16