A SEMINAR ON CONDUCTOMETRIC TITRATION PRESENTED BY DWIVEDI

A SEMINAR ON CONDUCTOMETRIC TITRATION PRESENTED BY- DWIVEDI ASHISH M. PHARM. 1 st SEM GUIDED BY-Mr. V. B. JADHAV 1

Contents: 1. Conductometry-: ØIntroduction ØOhm’s law. ØConductometric measurements. ØFactor affecting conductivity. ØApplication of conductometry. 2. Conductometric titration-: Ø Introduction. ØTypes of conductometric tiration. ØAdvantages of conductometric tiration. 3. Recent devlopement 4. References. 2

3

Total conductance of the solution is directly proportional to the sum of the n individual ion contributions. G = ci m, i 4

Ohm’s law 5

Conductivity measurements 1. Electrodes Two parallel platinized Pt. foil electrodes or Pt. black with electrodeposited a porous Pt. film which increases the surface area of the electrodes and further reduces faradaic polarization. 2. Primary standard solutions Primary standard KCl solution , at 25℃, 7. 419 g of KCl in 1000 g of solution has a specific conductivity of 0. 01286Ω-1/cm. 6
3. Conductivity Cell : Avoid the change of temperature during determination 4. Wheat stone bridge : 7

Factors affecting conductivity: v Size of ions v Temperature v Number of ions v Charge of ions ü Specific conductivity: -It is conductivity offered by a substance of 1 cm length and 1 sq. cm surface area. units are mhos/cm. ü Equivalent conductivity: -it is conductivity offered by a solution containing equivalent weight of solute in it. 8
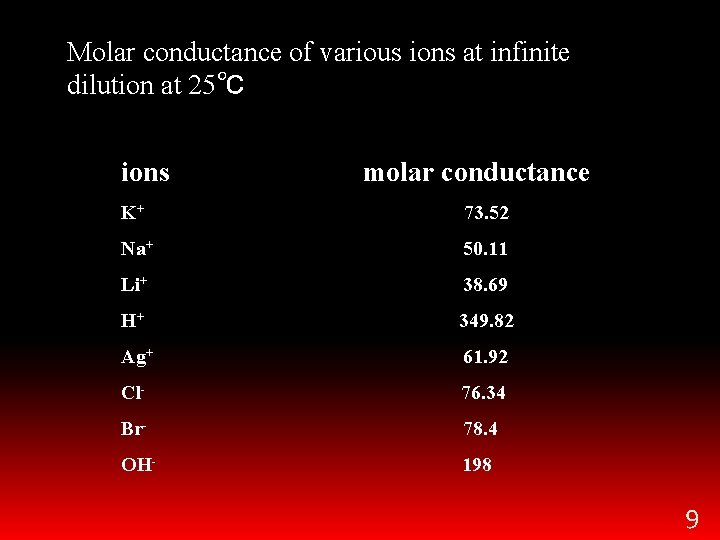
Molar conductance of various ions at infinite dilution at 25℃ ions molar conductance K+ 73. 52 Na+ 50. 11 Li+ 38. 69 H+ 349. 82 Ag+ 61. 92 Cl- 76. 34 Br- 78. 4 OH- 198 9

APPLICATIONS OF CONDUCTOMETRY It can be used for the determination of: Ø Solubility of sparingly soluble salts Ø Ionic product of water Ø Basicity of organic acids Ø Salinity of sea water (oceanographic work) Ø Chemical equilibrium in ionic reactions Ø Conductometric titration 10

CONDUCTOMETRIC TITRATIONS: Ø The determination of end point of a titration by means of conductivity measurements are known as conductometric titrations. 11

Types of conductometric titrations: Ø Ø Ø Acid-base titration Precipitation titration Replacement titration Redox (oxidation-reduction) titration Complexometric titration 12
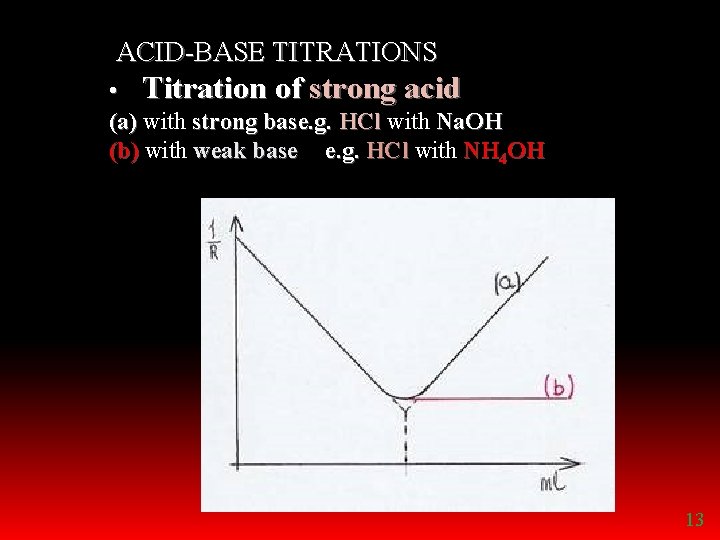
ACID-BASE TITRATIONS • Titration of strong acid (a) with strong base. g. HCl with Na. OH (b) with weak base e. g. HCl with NH 4 OH 13
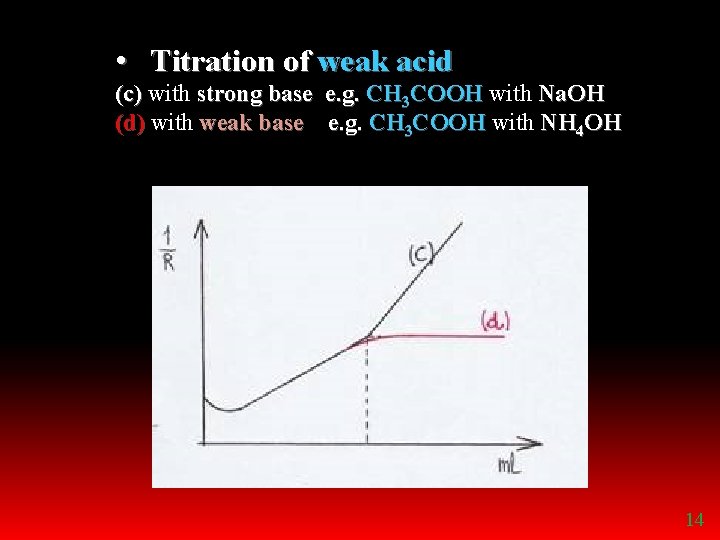
• Titration of weak acid (c) with strong base e. g. CH 3 COOH with Na. OH (d) with weak base e. g. CH 3 COOH with NH 4 OH 14
![PRECIPITATION TITRATIONS: [K++Cl-]+[Ag++No 3_] 15 PRECIPITATION TITRATIONS: [K++Cl-]+[Ag++No 3_] 15](http://slidetodoc.com/presentation_image_h/bbacdec21a600cab245f17906128761a/image-15.jpg)
PRECIPITATION TITRATIONS: [K++Cl-]+[Ag++No 3_] 15

REPLACEMENT TITRATIONS Ø Salt of strong acid and weak base vs. strong base Ex: ammonium chloride vs. sodium hydroxide Ø Salt of strong base and weak acid vs. strong acid Eg: sodium acetate vs. hydrochloric acid 16
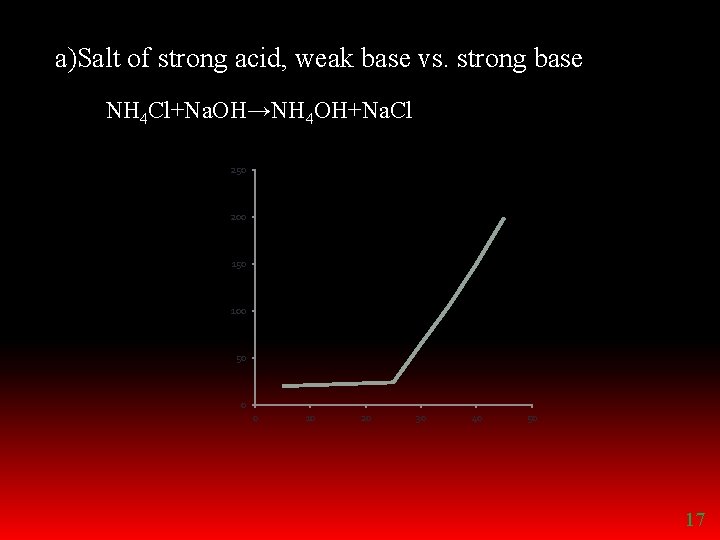
a)Salt of strong acid, weak base vs. strong base NH 4 Cl+Na. OH→NH 4 OH+Na. Cl 250 200 150 100 50 0 0 10 20 30 40 50 17
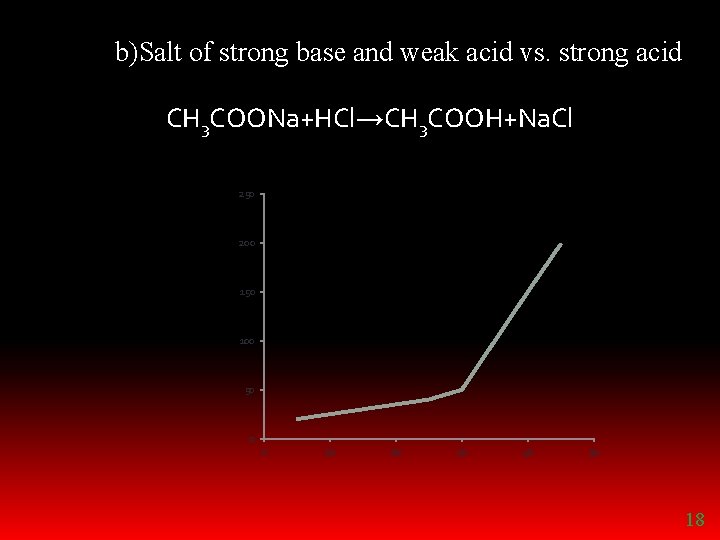
b)Salt of strong base and weak acid vs. strong acid CH 3 COONa+HCl→CH 3 COOH+Na. Cl 250 200 150 100 50 0 0 10 20 30 40 50 18
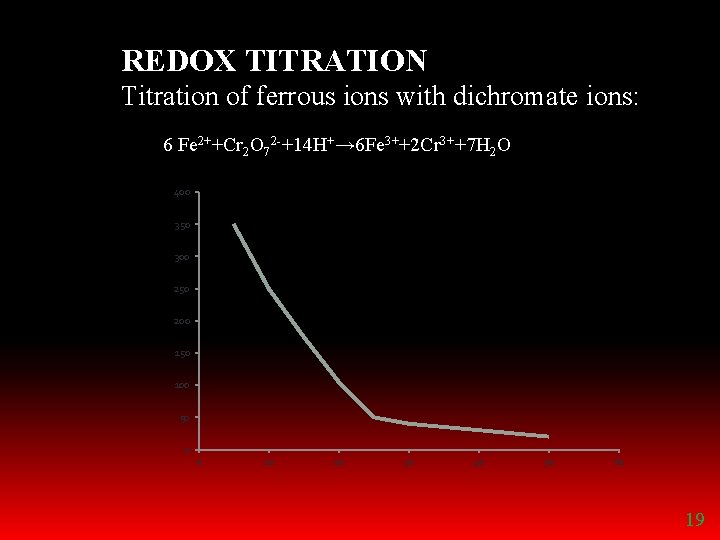
REDOX TITRATION Titration of ferrous ions with dichromate ions: 6 Fe 2++Cr 2 O 72 -+14 H+→ 6 Fe 3++2 Cr 3++7 H 2 O 400 350 300 250 200 150 100 50 0 0 10 20 30 40 50 60 19

COMPLEXOMETRIC TITRATION Ex. : -KCl vs. Hg(Cl. O 4)2 Ø Non-aqueous titrations can also be measured using conductometry. Ex: a)titration of weak bases vs. perchloric acid in dioxan-formic acid. b)Titration of weak organic acids in methanol vs. tetra methyl ammonium hydroxide in methanolbenzene. 20

ADVANTAGES OF CONDUCTOMETRIC TITRATIONS Ø No need of indicator Ø Colored or dilute solutions or turbid suspensions can be used for titrations. Ø Temperature is maintained constant throughout the titration. Ø End point can be determined accurately and errors are minimized as the end point is being determined graphically. 21

RECENT DEVLOPEMNTS ØIn refinary industries. ØEstimation of polyelectrolytic solution. ØBiotechnology. ØMicrobiosensors for enviromental monitoring. 22

References Ø Gurdeep. R. chatwal, sham k. anand, instrumental method of chemical analysis, himalaya publishing house, 2008, p. no. 2. 482 -2. 497. Ø Hovert H. willard, lynne L. merritt, john A. dean, frank A. settle, jr. , instrumental method of analysis CBS publishers 1986, p. no. 732 -750. Ø Kenneth A. connors, e textbook of pharmaceutical analysis, third edition, wiley india, p. no. 334. Ø Danniel christein, analytical chemistry, 2 nd edition, wiley india, p. no. 274. Ø www. pharmapaedia. com 23

Continued…. Ø www. authorstream. com Ø Kissinger, P. T. , AND W. W. Heineman, eds. , Laboratory Techniques in Electroanalytical Chemistry, Dekker, New York, 1984. Ø A. H. beckett , J. B. stenlake, practical pharmaceutical chemistry, fourth edition , part –two, p. no-91. Ø Lingane, J. J. , Electroanalytical Chemistry, 2 nd ed. , Wiley- Interscience, New York, 1958 24

THANK YOU 25
- Slides: 25