A Review of Medtronic Core Valve TAVI Clinical








- Slides: 25

A Review of Medtronic Core. Valve TAVI Clinical Results: Focus on Learning Curve Issues and Outcomes Ganesh Manoharan MBBCh, MD, FRCP(I), FRCP(Edin) Consultant Cardiologist, Royal Victoria Hospital Co-Director, CACR, University of Ulster Hon. Senior Lecturer, Queens University Belfast, UK

Ganesh Manoharan, MBBCh, MD § Advisory Committee – Boston Scientific Corporation – Cordis Corporation § Consulting Fees – Medtronic, Inc. § Honoraria – St. Jude Medical, Inc.

Core. Valve Experience More than 12, 000 implants over 30 countries

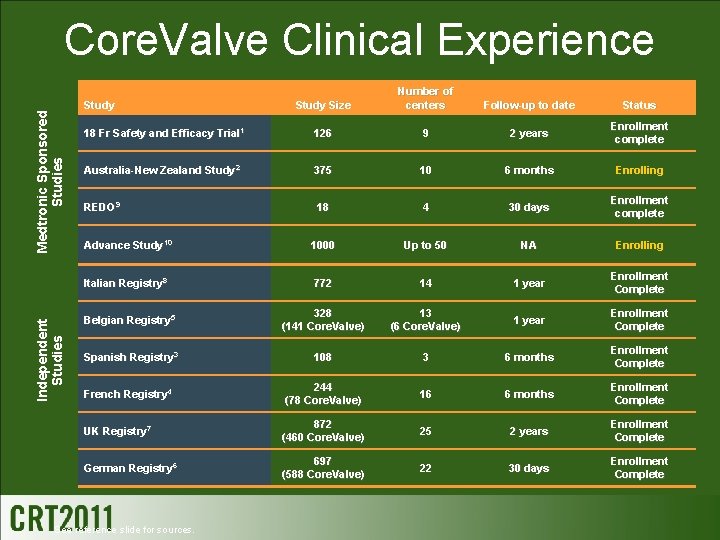
Independent Studies Medtronic Sponsored Studies Core. Valve Clinical Experience Study Size Number of centers Follow-up to date Status 18 Fr Safety and Efficacy Trial 1 126 9 2 years Enrollment complete Australia-New Zealand Study 2 375 10 6 months Enrolling REDO 9 18 4 30 days Enrollment complete Advance Study 10 1000 Up to 50 NA Enrolling Italian Registry 8 772 14 1 year Enrollment Complete Belgian Registry 5 328 (141 Core. Valve) 13 (6 Core. Valve) 1 year Enrollment Complete Spanish Registry 3 108 3 6 months Enrollment Complete French Registry 4 244 (78 Core. Valve) 16 6 months Enrollment Complete UK Registry 7 872 (460 Core. Valve) 25 2 years Enrollment Complete German Registry 6 697 (588 Core. Valve) 22 30 days Enrollment Complete Study See reference slide for sources.

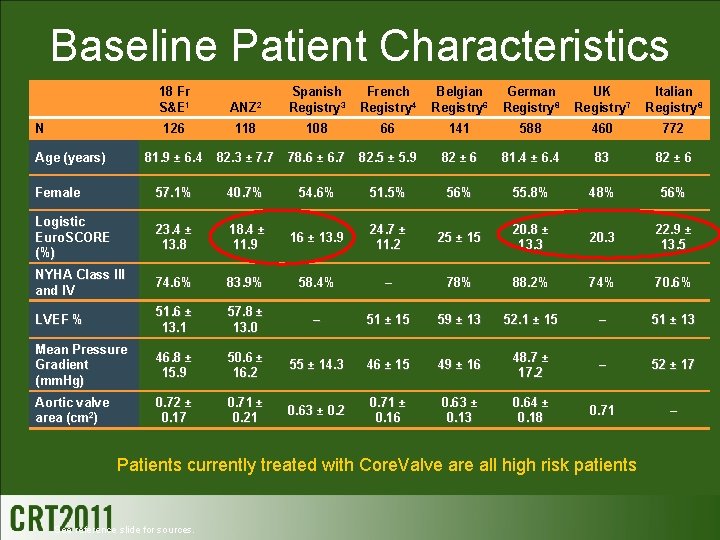
Baseline Patient Characteristics 18 Fr S&E 1 ANZ 2 Spanish Registry 3 French Registry 4 Belgian Registry 6 German Registry 8 UK Registry 7 Italian Registry 8 126 118 108 66 141 588 460 772 81. 9 ± 6. 4 82. 3 ± 7. 7 78. 6 ± 6. 7 82. 5 ± 5. 9 82 ± 6 81. 4 ± 6. 4 83 82 ± 6 Female 57. 1% 40. 7% 54. 6% 51. 5% 56% 55. 8% 48% 56% Logistic Euro. SCORE (%) 23. 4 ± 13. 8 18. 4 ± 11. 9 16 ± 13. 9 24. 7 ± 11. 2 25 ± 15 20. 8 ± 13. 3 20. 3 22. 9 ± 13. 5 NYHA Class III and IV 74. 6% 83. 9% 58. 4% – 78% 88. 2% 74% 70. 6% LVEF % 51. 6 ± 13. 1 57. 8 ± 13. 0 – 51 ± 15 59 ± 13 52. 1 ± 15 – 51 ± 13 Mean Pressure Gradient (mm. Hg) 46. 8 ± 15. 9 50. 6 ± 16. 2 55 ± 14. 3 46 ± 15 49 ± 16 48. 7 ± 17. 2 – 52 ± 17 Aortic valve area (cm 2) 0. 72 ± 0. 17 0. 71 ± 0. 21 0. 63 ± 0. 2 0. 71 ± 0. 16 0. 63 ± 0. 13 0. 64 ± 0. 18 0. 71 – N Age (years) Patients currently treated with Core. Valve are all high risk patients See reference slide for sources.

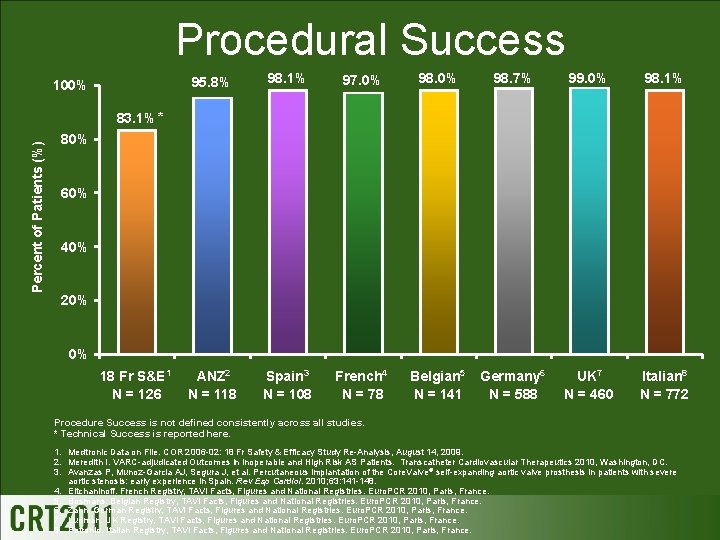
Procedural Success 100% 95. 8% 98. 1% 97. 0% 98. 7% 99. 0% 98. 1% ANZ 2 N = 118 Spain 3 N = 108 French 4 N = 78 Belgian 5 N = 141 Germany 6 N = 588 UK 7 N = 460 Italian 8 N = 772 Percent of Patients (%) 83. 1% * 80% 60% 40% 20% 0% 18 Fr S&E 1 N = 126 Procedure Success is not defined consistently across all studies. * Technical Success is reported here. 1. Medtronic Data on File. COR 2006 -02: 18 Fr Safety & Efficacy Study Re-Analysis, August 14, 2009. 2. Meredith I. VARC-adjudicated Outcomes in Inoperable and High Risk AS Patients. Transcatheter Cardiovascular Therapeutics 2010, Washington, DC. 3. Avanzas P, Munoz-Garcia AJ, Segura J, et al. Percutaneous implantation of the Core. Valve® self-expanding aortic valve prosthesis in patients with severe aortic stenosis: early experience in Spain. Rev Esp Cardiol. 2010; 63: 141 -148. 4. Eltchaninoff. French Registry, TAVI Facts, Figures and National Registries. Euro. PCR 2010, Paris, France. 5. Bosmans. Belgian Registry, TAVI Facts, Figures and National Registries. Euro. PCR 2010, Paris, France. 6. Zahn. German Registry, TAVI Facts, Figures and National Registries. Euro. PCR 2010, Paris, France. 7. Ludman. UK Registry, TAVI Facts, Figures and National Registries. Euro. PCR 2010, Paris, France. 8. Petronio. Italian Registry, TAVI Facts, Figures and National Registries. Euro. PCR 2010, Paris, France.

Has experience reduced complication rates?

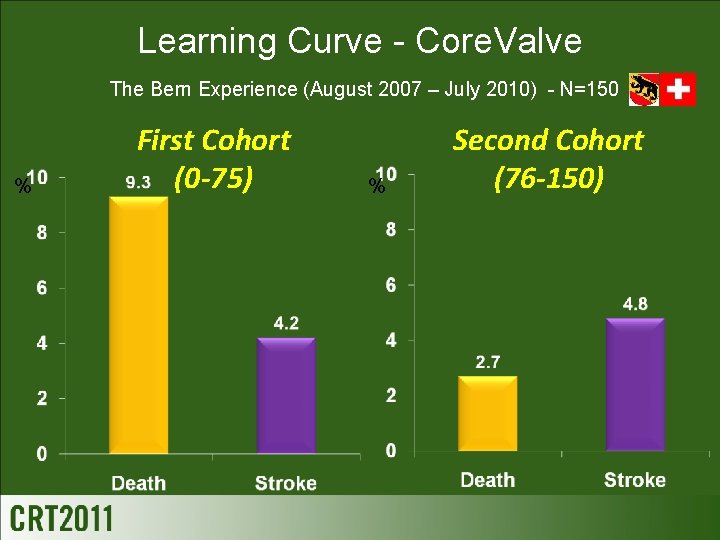
Learning Curve - Core. Valve The Bern Experience (August 2007 – July 2010) - N=150 % First Cohort (0 -75) % Second Cohort (76 -150)

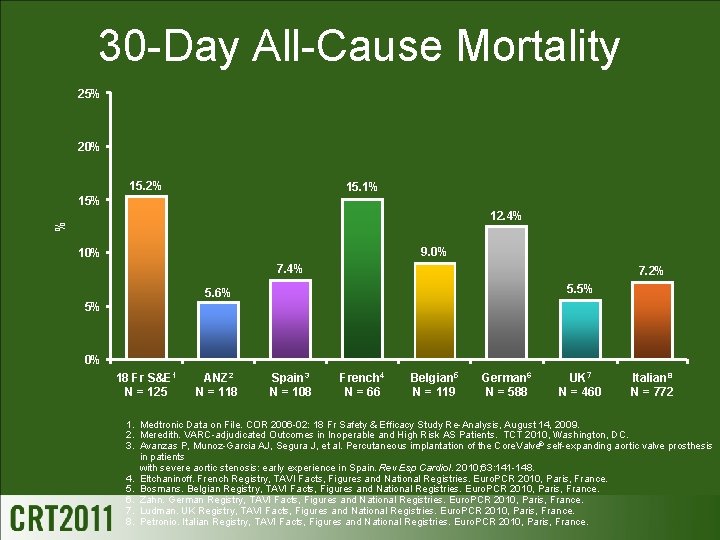
30 -Day All-Cause Mortality 25% 20% 15. 2% 15. 1% 15% % 12. 4% 9. 0% 10% 7. 4% 7. 2% 5. 5% 5. 6% 5% 0% 18 Fr S&E 1 N = 125 ANZ 2 N = 118 Spain 3 N = 108 French 4 N = 66 Belgian 5 N = 119 German 6 N = 588 UK 7 N = 460 Italian 8 N = 772 1. Medtronic Data on File. COR 2006 -02: 18 Fr Safety & Efficacy Study Re-Analysis, August 14, 2009. 2. Meredith. VARC-adjudicated Outcomes in Inoperable and High Risk AS Patients. TCT 2010, Washington, DC. 3. Avanzas P, Munoz-Garcia AJ, Segura J, et al. Percutaneous implantation of the Core. Valve® self-expanding aortic valve prosthesis in patients with severe aortic stenosis: early experience in Spain. Rev Esp Cardiol. 2010; 63: 141 -148. 4. Eltchaninoff. French Registry, TAVI Facts, Figures and National Registries. Euro. PCR 2010, Paris, France. 5. Bosmans. Belgian Registry, TAVI Facts, Figures and National Registries. Euro. PCR 2010, Paris, France. 6. Zahn. German Registry, TAVI Facts, Figures and National Registries. Euro. PCR 2010, Paris, France. 7. Ludman. UK Registry, TAVI Facts, Figures and National Registries. Euro. PCR 2010, Paris, France. 8. Petronio. Italian Registry, TAVI Facts, Figures and National Registries. Euro. PCR 2010, Paris, France.

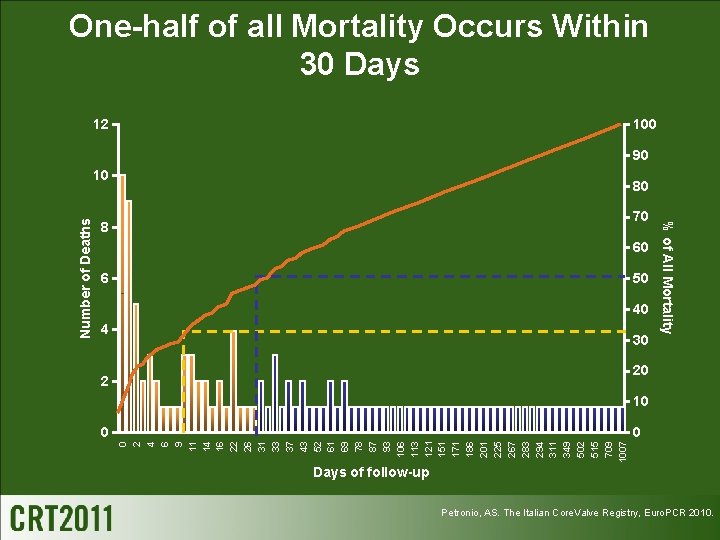
One-half of all Mortality Occurs Within 30 Days 12 100 90 80 70 8 60 6 50 40 4 30 % of All Mortality 20 2 10 0 2 4 6 9 11 14 16 22 26 31 33 37 43 52 61 69 78 87 93 106 113 121 151 171 186 201 225 267 283 294 311 349 502 515 709 1007 Number of Deaths 10 Days of follow-up Petronio, AS. The Italian Core. Valve Registry, Euro. PCR 2010.

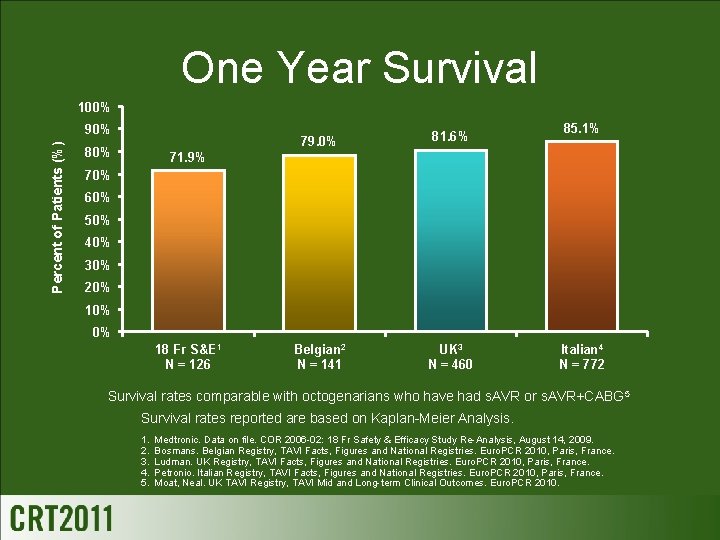
One Year Survival 100% Percent of Patients (%) 90% 80% 79. 0% 81. 6% Belgian 2 N = 141 UK 3 N = 460 85. 1% 71. 9% 70% 60% 50% 40% 30% 20% 10% 0% 18 Fr S&E 1 N = 126 Italian 4 N = 772 Survival rates comparable with octogenarians who have had s. AVR or s. AVR+CABG 5 Survival rates reported are based on Kaplan-Meier Analysis. 1. 2. 3. 4. 5. Medtronic. Data on file. COR 2006 -02: 18 Fr Safety & Efficacy Study Re-Analysis, August 14, 2009. Bosmans. Belgian Registry, TAVI Facts, Figures and National Registries. Euro. PCR 2010, Paris, France. Ludman. UK Registry, TAVI Facts, Figures and National Registries. Euro. PCR 2010, Paris, France. Petronio. Italian Registry, TAVI Facts, Figures and National Registries. Euro. PCR 2010, Paris, France. Moat, Neal. UK TAVI Registry, TAVI Mid and Long-term Clinical Outcomes. Euro. PCR 2010.

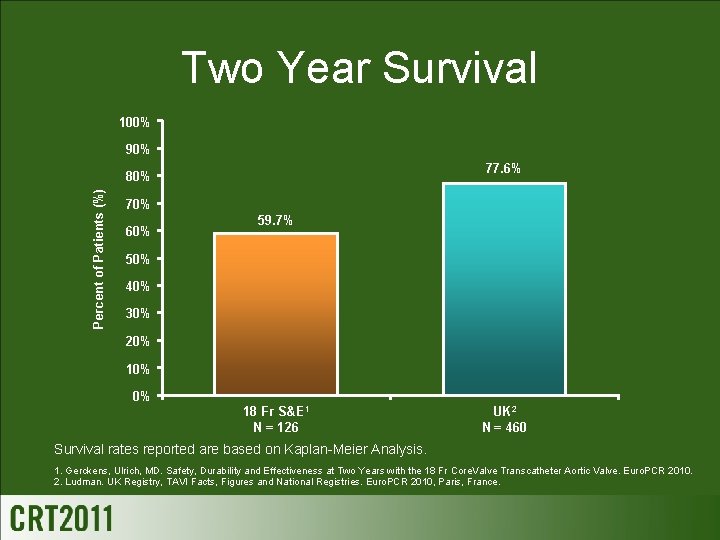
Two Year Survival 100% 90% 77. 6% Percent of Patients (%) 80% 70% 60% 59. 7% 50% 40% 30% 20% 10% 0% 18 Fr S&E 1 N = 126 UK 2 N = 460 Survival rates reported are based on Kaplan-Meier Analysis. 1. Gerckens, Ulrich, MD. Safety, Durability and Effectiveness at Two Years with the 18 Fr Core. Valve Transcatheter Aortic Valve. Euro. PCR 2010. 2. Ludman. UK Registry, TAVI Facts, Figures and National Registries. Euro. PCR 2010, Paris, France.

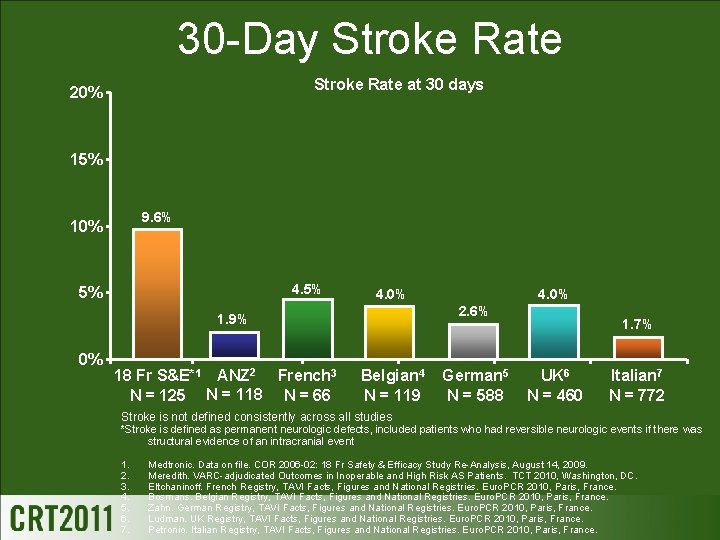
30 -Day Stroke Rate at 30 days 20% 15% 9. 6% 10% 4. 5% 5% 4. 0% 2. 6% 1. 9% 0% 18 Fr S&E*1 ANZ 2 French 3 N = 125 N = 118 N = 66 4. 0% Belgian 4 N = 119 German 5 N = 588 1. 7% UK 6 N = 460 Italian 7 N = 772 Stroke is not defined consistently across all studies *Stroke is defined as permanent neurologic defects, included patients who had reversible neurologic events if there was structural evidence of an intracranial event 1. 2. 3. 4. 5. 6. 7. Medtronic. Data on file. COR 2006 -02: 18 Fr Safety & Efficacy Study Re-Analysis, August 14, 2009. Meredith. VARC-adjudicated Outcomes in Inoperable and High Risk AS Patients. TCT 2010, Washington, DC. Eltchaninoff. French Registry, TAVI Facts, Figures and National Registries. Euro. PCR 2010, Paris, France. Bosmans. Belgian Registry, TAVI Facts, Figures and National Registries. Euro. PCR 2010, Paris, France. Zahn. German Registry, TAVI Facts, Figures and National Registries. Euro. PCR 2010, Paris, France. Ludman. UK Registry, TAVI Facts, Figures and National Registries. Euro. PCR 2010, Paris, France. Petronio. Italian Registry, TAVI Facts, Figures and National Registries. Euro. PCR 2010, Paris, France.

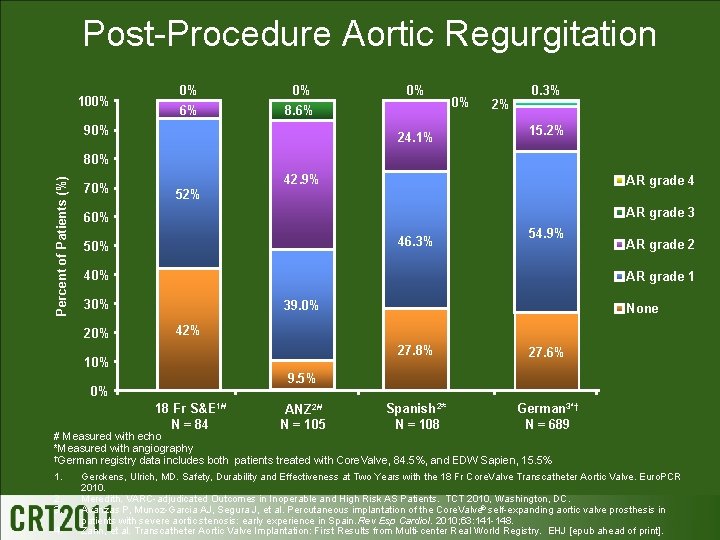
Post-Procedure Aortic Regurgitation 100% 0% 0% 6% 8. 6% 90% 0% 24. 1% 0% 2% 0. 3% 15. 2% Percent of Patients (%) 80% 70% 42. 9% AR grade 4 52% AR grade 3 60% 46. 3% 50% 54. 9% 40% AR grade 1 30% 20% AR grade 2 39. 0% None 42% 10% 27. 8% 27. 6% Spanish 2* N = 108 German 3*† N = 689 9. 5% 0% 18 Fr S&E 1# N = 84 ANZ 2# N = 105 # Measured with echo *Measured with angiography †German registry data includes both patients treated with Core. Valve, 84. 5%, and EDW Sapien, 15. 5% 1. Gerckens, Ulrich, MD. Safety, Durability and Effectiveness at Two Years with the 18 Fr Core. Valve Transcatheter Aortic Valve. Euro. PCR 2. 3. 4. 2010. Meredith. VARC-adjudicated Outcomes in Inoperable and High Risk AS Patients. TCT 2010, Washington, DC. Avanzas P, Munoz-Garcia AJ, Segura J, et al. Percutaneous implantation of the Core. Valve® self-expanding aortic valve prosthesis in patients with severe aortic stenosis: early experience in Spain. Rev Esp Cardiol. 2010; 63: 141 -148. Zahn, et al. Transcatheter Aortic Valve Implantation: First Results from Multi-center Real World Registry. EHJ [epub ahead of print].

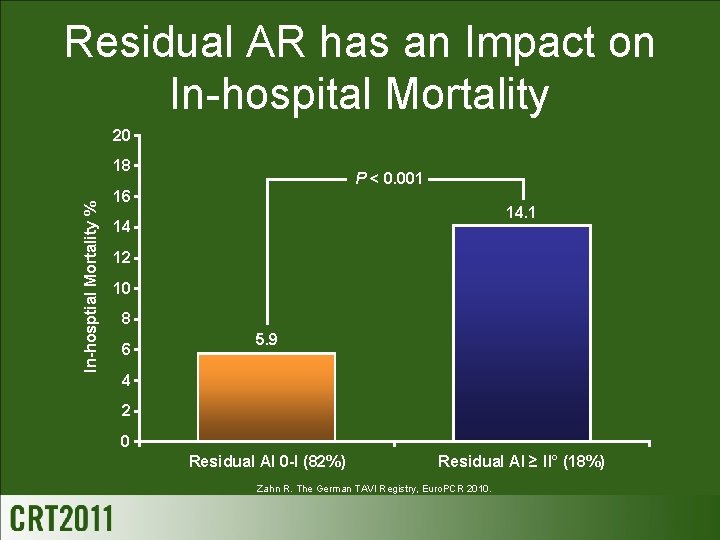
Residual AR has an Impact on In-hospital Mortality 20 In-hosptial Mortality % 18 P < 0. 001 16 14. 1 14 12 10 8 6 5. 9 4 2 0 Residual AI 0 -I (82%) Residual AI ≥ II° (18%) Zahn R. The German TAVI Registry, Euro. PCR 2010.

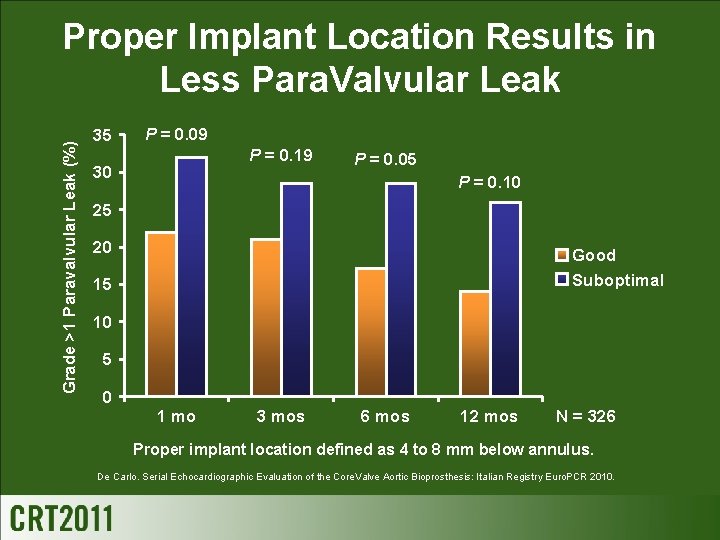
Grade >1 Paravalvular Leak (%) Proper Implant Location Results in Less Para. Valvular Leak 35 P = 0. 09 P = 0. 19 30 P = 0. 05 P = 0. 10 25 20 Good Suboptimal 15 10 5 0 1 mo 3 mos 6 mos 12 mos N = 326 Proper implant location defined as 4 to 8 mm below annulus. De Carlo. Serial Echocardiographic Evaluation of the Core. Valve Aortic Bioprosthesis: Italian Registry Euro. PCR 2010.

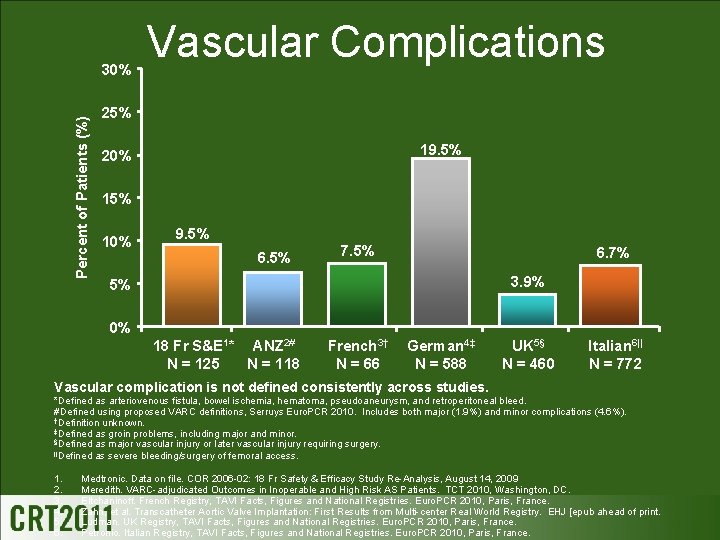
Percent of Patients (%) 30% Vascular Complications 25% 19. 5% 20% 15% 10% 9. 5% 6. 5% 7. 5% 6. 7% 3. 9% 5% 0% 18 Fr S&E 1* ANZ 2# N = 125 N = 118 French 3† N = 66 German 4‡ N = 588 UK 5§ N = 460 Italian 6|| N = 772 Vascular complication is not defined consistently across studies. *Defined as arteriovenous fistula, bowel ischemia, hematoma, pseudoaneurysm, and retroperitoneal bleed. #Defined using proposed VARC definitions, Serruys Euro. PCR 2010. Includes both major (1. 9%) and minor complications (4. 6%). †Definition unknown. ‡Defined as groin problems, including major and minor. §Defined as major vascular injury or later vascular injury requiring surgery. ||Defined as severe bleeding/surgery of femoral access. 1. 2. 3. 4. 5. 6. Medtronic. Data on file. COR 2006 -02: 18 Fr Safety & Efficacy Study Re-Analysis, August 14, 2009 Meredith. VARC-adjudicated Outcomes in Inoperable and High Risk AS Patients. TCT 2010, Washington, DC. Eltchaninoff. French Registry, TAVI Facts, Figures and National Registries. Euro. PCR 2010, Paris, France. Zahn, et al. Transcatheter Aortic Valve Implantation: First Results from Multi-center Real World Registry. EHJ [epub ahead of print. Ludman. UK Registry, TAVI Facts, Figures and National Registries. Euro. PCR 2010, Paris, France. Petronio. Italian Registry, TAVI Facts, Figures and National Registries. Euro. PCR 2010, Paris, France.

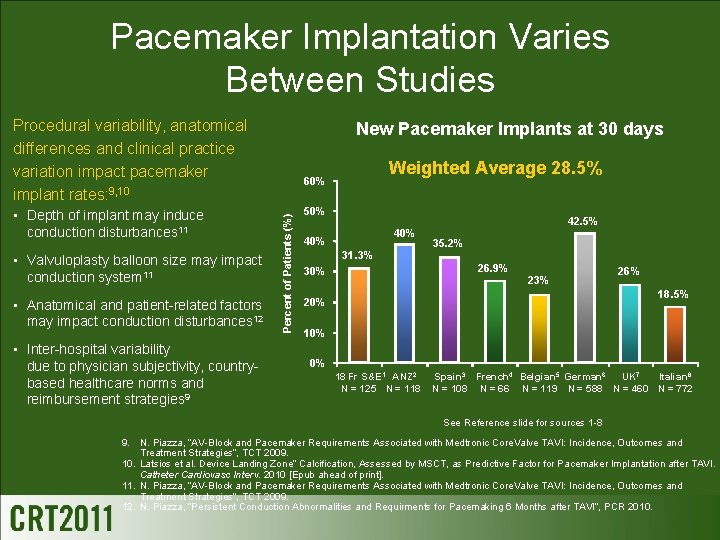
Pacemaker Implantation Varies Between Studies Procedural variability, anatomical differences and clinical practice variation impact pacemaker implant rates: 9, 10 • Valvuloplasty balloon size may impact conduction system 11 • Anatomical and patient-related factors may impact conduction disturbances 12 • Inter-hospital variability due to physician subjectivity, countrybased healthcare norms and reimbursement strategies 9 Weighted Average 28. 5% 60% Percent of Patients (%) • Depth of implant may induce conduction disturbances 11 New Pacemaker Implants at 30 days 50% 42. 5% 40% 31. 3% 35. 2% 26. 9% 30% 23% 26% 18. 5% 20% 10% 0% 18 Fr S&E 1 ANZ 2 N = 125 N = 118 Spain 3 French 4 Belgian 5 German 6 UK 7 Italian 8 N = 108 N = 66 N = 119 N = 588 N = 460 N = 772 See Reference slide for sources 1 -8 9. N. Piazza, “AV-Block and Pacemaker Requirements Associated with Medtronic Core. Valve TAVI: Incidence, Outcomes and Treatment Strategies”, TCT 2009. 10. Latsios et al. Device Landing Zone” Calcification, Assessed by MSCT, as Predictive Factor for Pacemaker Implantation after TAVI. Catheter Cardiovasc Interv. 2010 [Epub ahead of print]. 11. N. Piazza, “AV-Block and Pacemaker Requirements Associated with Medtronic Core. Valve TAVI: Incidence, Outcomes and Treatment Strategies”, TCT 2009. 12. N. Piazza, “Persistent Conduction Abnormalities and Requirments for Pacemaking 6 Months after TAVI”, PCR 2010.

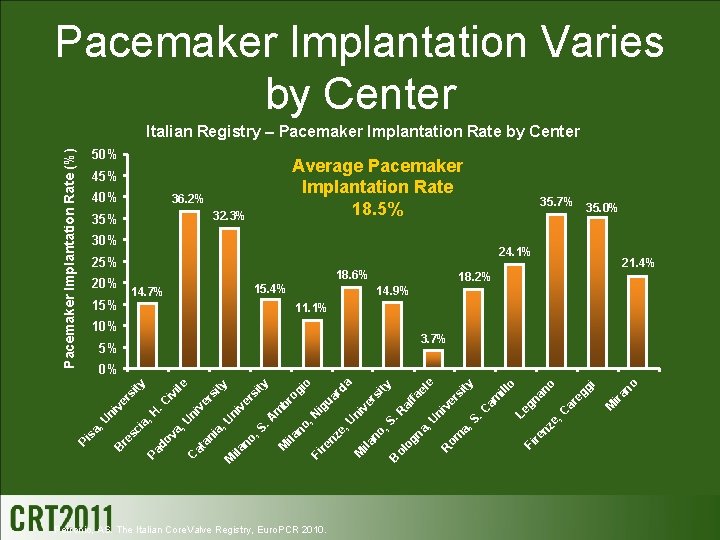
Pacemaker Implantation Varies by Center 50% Average Pacemaker Implantation Rate 18. 5% 40% 36. 2% 32. 3% 35. 7% 30% 20% 15% 35. 0% 24. 1% 25% 18. 6% 21. 4% 18. 2% 15. 4% 14. 7% 14. 9% 11. 1% 10% 3. 7% 5% Petronio, AS. The Italian Core. Valve Registry, Euro. PCR 2010. re nz no ira M e, C ar e gg i o an gn Le S. C am ill o Fi R om a, , U ni ve rs ity el e af fa na og ol B M ila no , S . R ni ve r si ty a ua rd nz re Fi M ila no e, U , N ig br m . A , S no ila M og ity ve rs ia , U ni at an C ni ve r si ty iv ile , U va Pa do B re sc i a, H . C ve rs ni , U Pi sa io 0% ity Pacemaker Implantation Rate (%) Italian Registry – Pacemaker Implantation Rate by Center

Is TAVI outcome related to Euroscore?

Core. Valve Australia-New Zealand Study Baseline Characteristics Characteristic Age (years) Logistic Euro. SCORE (%) Mean ± SD, or % 82. 3 ± 7. 7 18. 4 ± 11. 9 STS (%) (Mortality) 5. 6 ± 2. 7 Female 40. 7% Class I: 0. 0% Class II: 16. 1% Class III: 62. 5% Class IV: 21. 4% 0. 71 ± 0. 21 81. 5 ± 23. 6 50. 6 ± 16. 2 57. 8 ± 13. 0 NYHA Aortic valve area (cm 2) Peak gradient (mm. Hg) Mean gradient (mm. Hg) LVEF (%)

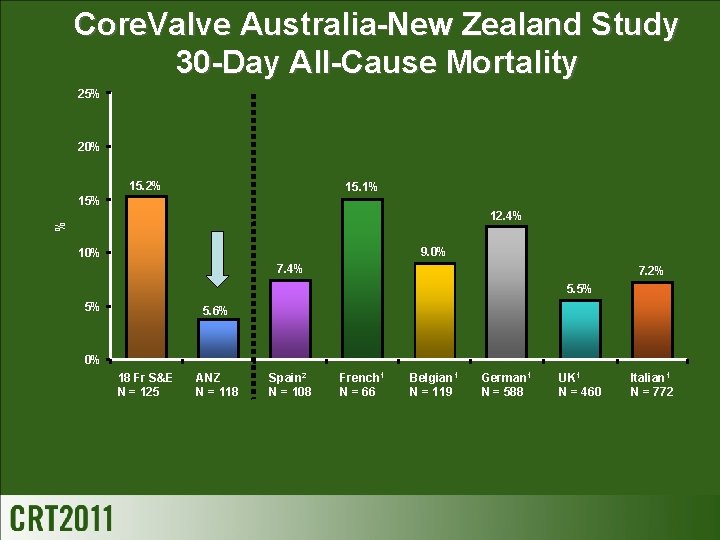
Core. Valve Australia-New Zealand Study 30 -Day All-Cause Mortality 25% 20% 15. 2% 15. 1% 15% % 12. 4% 9. 0% 10% 7. 4% 7. 2% 5. 5% 5% 5. 6% 0% 18 Fr S&E N = 125 ANZ N = 118 Spain 2 N = 108 French 1 N = 66 Belgian 1 N = 119 German 1 N = 588 UK 1 N = 460 Italian 1 N = 772

Core. Valve Clinical Portfolio Evaluate Long-term Performance & QOL Evaluate Procedural Best Practices Evaluate Performance in Expanded Patient Populations 18 Fr Safety & Efficacy ADVANCE II Study SURTAVI ANZ Study ADVANCE Study US Pivotal Extreme Risk (Inoperable) Cohort US Pivotal High Risk Surgical Cohort REDO

Conclusions (1) • TAVI with the Medtronic Core. Valve is safe and effective • Continued improvements in technique, understanding patient selection (clinical and technical suitability) and post procedural management impacts on outcome • Continued emphasis on teaching, training and dissemination of information will impact on outcome

Conclusions (2) • Standardisation of event reporting (VARC) will allow more transparent comparisons to be made • Technological iterations will further enhance the safety and efficacy of this technique • Long term durability and outcome awaited