A Randomized Trial of a Bioabsorbable PolymerBased Metallic

A Randomized Trial of a Bioabsorbable Polymer-Based Metallic DES vs. a BMS with Short DAPT in Patients with Coronary Artery Disease Older than 75 Years. The SENIOR Trial O. Varenne, S. Cook, G. Sideris, S. Kedev, T. Cuisset, D. Carrié, T. Hovasse, P. Garot, R. El Mahmoud, C. Spaulding, G. Helft, J. Diaz Fernandez, S. Brugaletta, E. Pinar Bermudez, J. Mauri Ferre, P. Commeau, E. Teiger, K. Bogearts, M. Sabate, M-C. Morice and P. Sinnaeve, for the SENIOR investigators.

Disclosure Statement of Financial Interest Within the past 12 months, I or my partner have had a financial interest with the organizations listed below Financial Relationship Company • Lectures Fees • • Boston Scientific Abbott Vascular Astra Zeneca Servier

SENIOR Study Hypothesis To demonstrate that a bioabsorbable polymer DES & short DAPT in elderly patients is associated with: - a lower rate of MACCE at 1 year vs. BMS (efficacy) - a similar risk of bleeding at 1 year vs. BMS (safety) - a similar risk of stent thrombosis at 1 year vs. BMS (safety) www. Clinical. Trial. gov: NCT 02099617
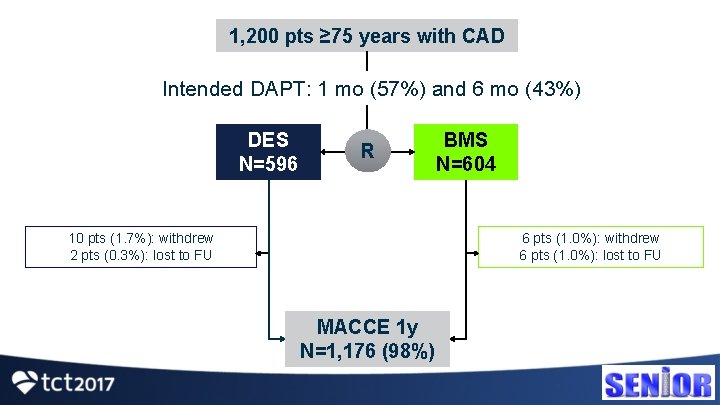
1, 200 pts ≥ 75 years with CAD Intended DAPT: 1 mo (57%) and 6 mo (43%) DES N=596 R 10 pts (1. 7%): withdrew 2 pts (0. 3%): lost to FU BMS N=604 6 pts (1. 0%): withdrew 6 pts (1. 0%): lost to FU MACCE 1 y N=1, 176 (98%)

Synergy™ DES used in SENIOR 74μm Everolimus PLGA Polymer SEM of coating (x 5000) Meredith I. et al. Euro. Interv 2017 (in Press) doi: 10. 4244/EU-D-17 -00529 BMS
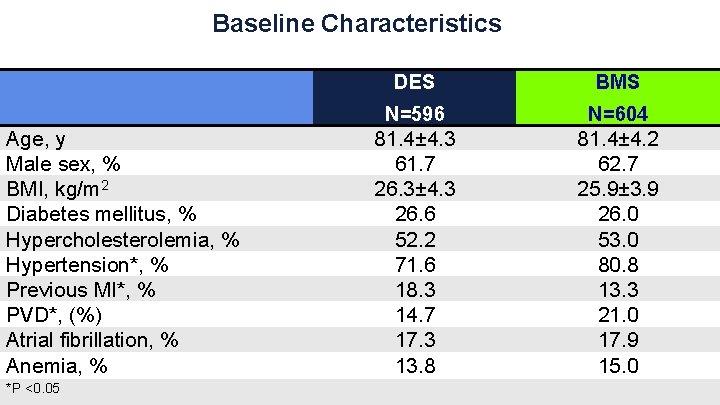
Baseline Characteristics Age, y Male sex, % BMI, kg/m 2 Diabetes mellitus, % Hypercholesterolemia, % Hypertension*, % Previous MI*, % PVD*, (%) Atrial fibrillation, % Anemia, % *P <0. 05 DES BMS N=596 N=604 81. 4± 4. 3 61. 7 26. 3± 4. 3 26. 6 52. 2 71. 6 18. 3 14. 7 17. 3 13. 8 81. 4± 4. 2 62. 7 25. 9± 3. 9 26. 0 53. 0 80. 8 13. 3 21. 0 17. 9 15. 0
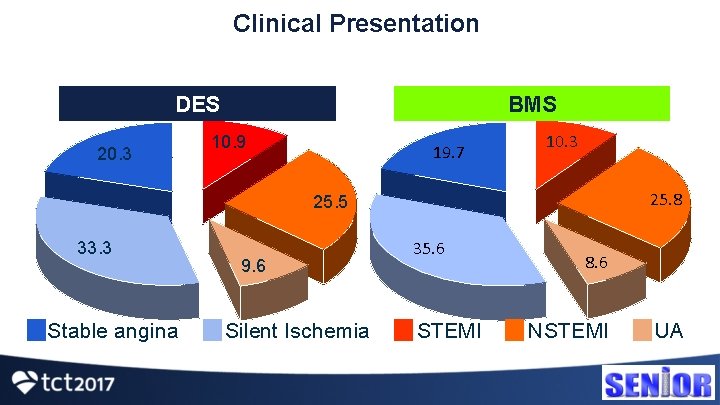
Clinical Presentation DES 20. 3 BMS 10. 9 19. 7 10. 3 25. 8 25. 5 33. 3 Stable angina 9. 6 Silent Ischemia 35. 6 STEMI 8. 6 NSTEMI UA
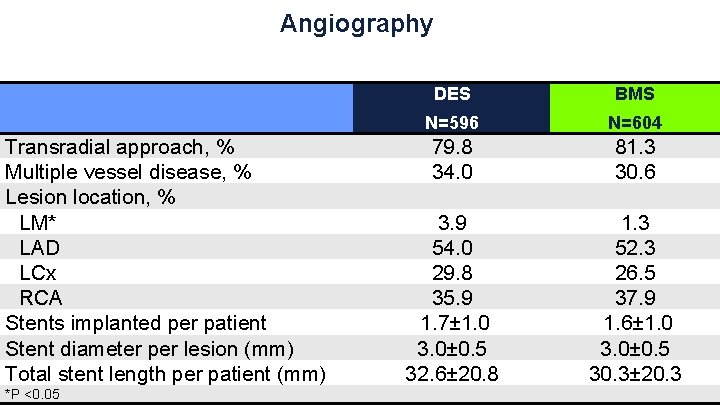
Angiography Transradial approach, % Multiple vessel disease, % Lesion location, % LM* LAD LCx RCA Stents implanted per patient Stent diameter per lesion (mm) Total stent length per patient (mm) *P <0. 05 DES BMS N=596 N=604 79. 8 34. 0 3. 9 54. 0 29. 8 35. 9 1. 7± 1. 0 3. 0± 0. 5 32. 6± 20. 8 81. 3 30. 6 1. 3 52. 3 26. 5 37. 9 1. 6± 1. 0 3. 0± 0. 5 30. 3± 20. 3
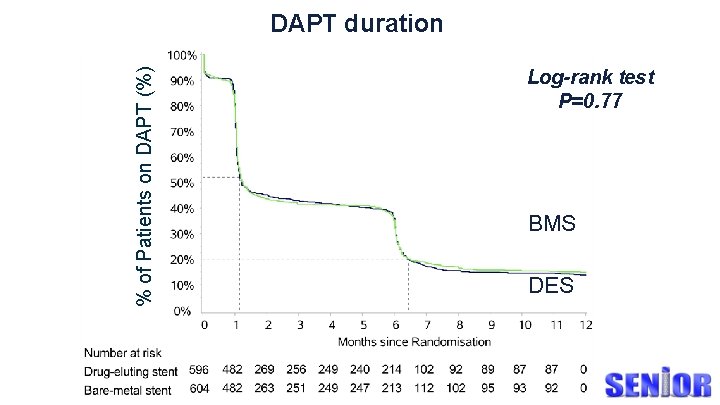
DAPT duration % of Patients on DAPT (%) Log-rank test P=0. 77 BMS DES
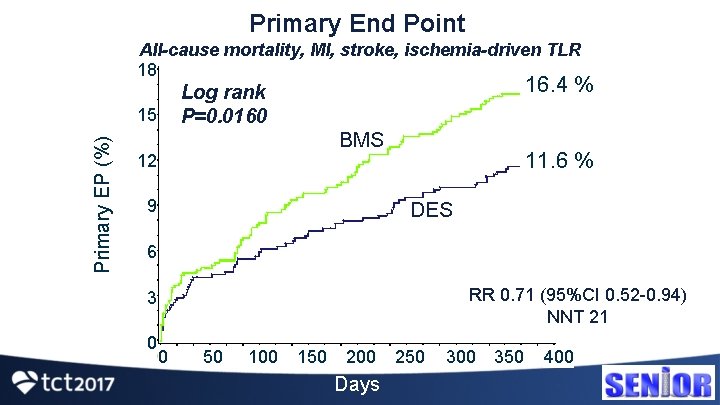
Primary End Point All-cause mortality, MI, stroke, ischemia-driven TLR 18 15 Primary EP (%) 16. 4 % Log rank P=0. 0160 BMS 12 9 11. 6 % DES 6 RR 0. 71 (95%CI 0. 52 -0. 94) NNT 21 3 0 0 50 100 150 200 Days 250 300 350 400

MACCE Components 20 Patients (%) P=0. 0172 16. 4 15 10 11. 6 P=0. 20 P=0. 0002 8 5 P=0. 92 6. 1 P=0. 08 2 0 Mortality 5. 9 3. 6 3. 7 1. 7 0. 8 Stroke MI ID-TLR MACCE DES BMS
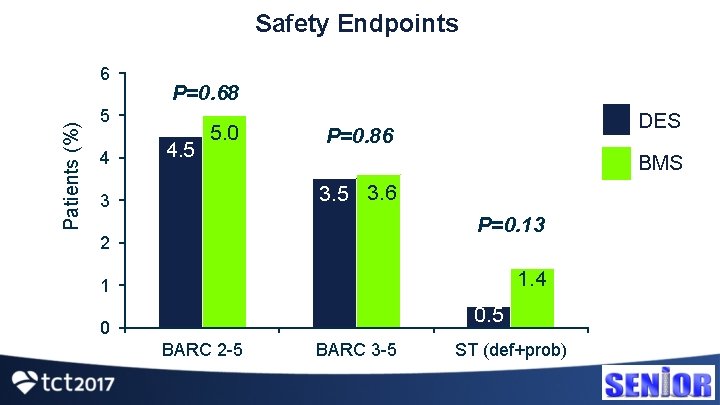
Safety Endpoints Patients (%) 6 P=0. 68 5 4 4. 5 5. 0 DES P=0. 86 BMS 3. 5 3. 6 3 P=0. 13 2 1. 4 1 0. 5 0 BARC 2 -5 BARC 3 -5 ST (def+prob)

Conclusion • PCI with a contemporary bioabsorbable polymer DES is more effective, and as safe as BMS in elderly patients with CAD, on a short DAPT tailored to their clinical presentation BMS should no longer be used as a strategy to reduce DAPT duration in elderly patients All TVT 2016 faculty disclosures are listed online and on the app.
- Slides: 13