A Randomized Prospective Intercontinental Evaluation of a Bioresorbable

A Randomized, Prospective, Intercontinental Evaluation of a Bioresorbable Polymer Sirolimus -eluting Stent: the CENTURY II Trial: an Update with 2 Years Outcomes Shigeru Saito, MD Department of Cardiology and Catheterization Laboratory, Shonan Kamakura General Hospital, Kamakura, Japan on behalf of CENTURY II investigators

Disclosure Statement of Financial Interest Within the past 12 months, I or my spouse/partner have had a financial interest/arrangement or affiliation with the organization(s) listed below. Affiliation/Financial Relationship • Grant/Research Support • Consulting Fees/Honoraria • Major Stock Shareholder/Equity • Royalty Income • Ownership/Founder • Intellectual Property Rights • Other Financial Benefit Company • Terumo Corporation • Non • Non

Background • CENTURY II is a large scale, prospective, multicentre, randomized (1: 1), single blind, controlled, non-inferiority trial with limited exclusion criteria; • The aims of this trial are: ü to establish safety and efficacy of a thin strut abluminally coated DES with bioresorbable polymer, Ultimaster, by comparing it with permanent polymer circumferentially coated DES, Xience, ü to make global clinical data compliant with regulatory requirements in Europe and Japan and address the gaps in timely access to new technology around the world • This is the first time report of two years data for total population enrolled from 58 sites from Europe, Japan and Korea.
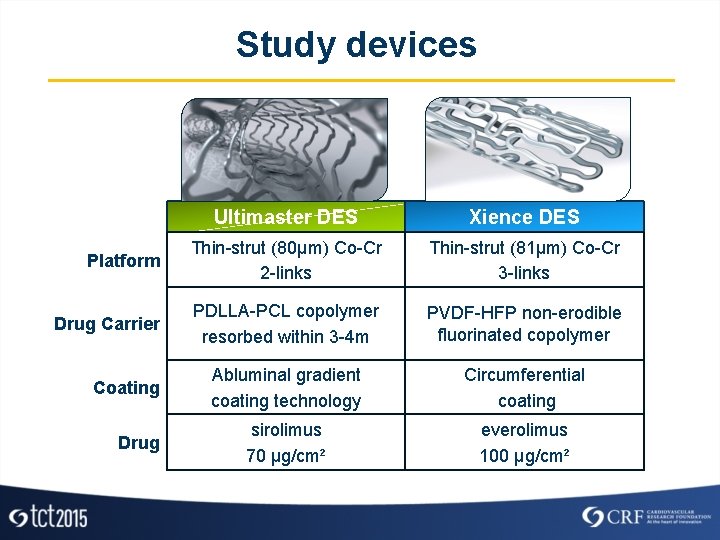
Study devices Ultimaster DES Xience DES Platform Thin-strut (80µm) Co-Cr 2 -links Thin-strut (81µm) Co-Cr 3 -links Drug Carrier PDLLA-PCL copolymer resorbed within 3 -4 m PVDF-HFP non-erodible fluorinated copolymer Abluminal gradient coating technology Circumferential coating sirolimus 70 µg/cm² everolimus 100 µg/cm² Coating Drug

Method Inclusion criteria Age ≥ 18 years Suitable for treatment with DES RVD matching stents 2. 5 -4. 0 mm Diameter stenosis >50% Eligible for DAPT Exclusion criteria-general* EF <25% Renal failure Cardiogenic shock Planned staged procedure Primary endpoint: freedom from TLF at 9 months, which is composite of cardiac death, TV-MI, and clinical indicated-TLR. * Additional exclusion criteria for Japan see reference Saito S. et al, Eur Heart J. 2014; 35, 2021 -31.
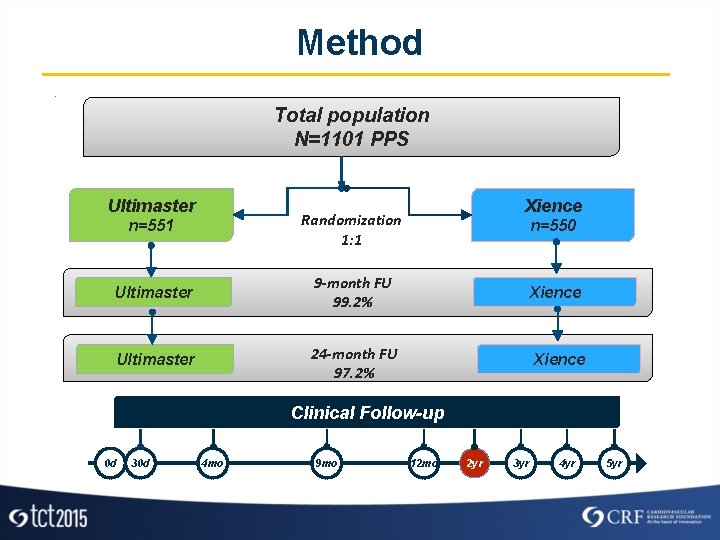
Method Total population N=1101 PPS Ultimaster Xience n=551 Randomization 1: 1 Ultimaster 9 -month FU 99. 2% Xience Ultimaster 24 -month FU 97. 2% Xience n=550 Clinical Follow-up 0 d 0 d 30 d 4 mo 9 mo 12 mo 2 yr 3 yr 4 yr 5 yr
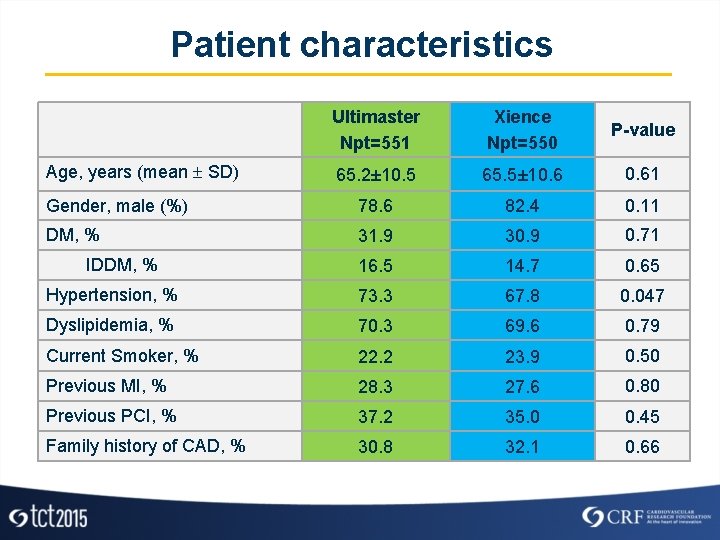
Patient characteristics Ultimaster Npt=551 Xience Npt=550 P-value 65. 2± 10. 5 65. 5± 10. 61 Gender, male (%) 78. 6 82. 4 0. 11 DM, % 31. 9 30. 9 0. 71 16. 5 14. 7 0. 65 Hypertension, % 73. 3 67. 8 0. 047 Dyslipidemia, % 70. 3 69. 6 0. 79 Current Smoker, % 22. 2 23. 9 0. 50 Previous MI, % 28. 3 27. 6 0. 80 Previous PCI, % 37. 2 35. 0 0. 45 Family history of CAD, % 30. 8 32. 1 0. 66 Age, years (mean SD) IDDM, %
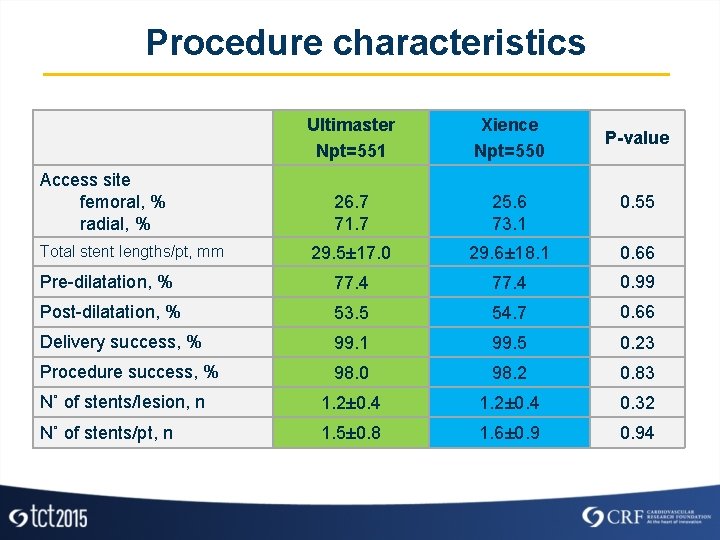
Procedure characteristics Ultimaster Npt=551 Xience Npt=550 26. 7 71. 7 25. 6 73. 1 0. 55 29. 5± 17. 0 29. 6± 18. 1 0. 66 Pre-dilatation, % 77. 4 0. 99 Post-dilatation, % 53. 5 54. 7 0. 66 Delivery success, % 99. 1 99. 5 0. 23 Procedure success, % 98. 0 98. 2 0. 83 N˚ of stents/lesion, n 1. 2± 0. 4 0. 32 N˚ of stents/pt, n 1. 5± 0. 8 1. 6± 0. 94 Access site femoral, % radial, % Total stent lengths/pt, mm P-value
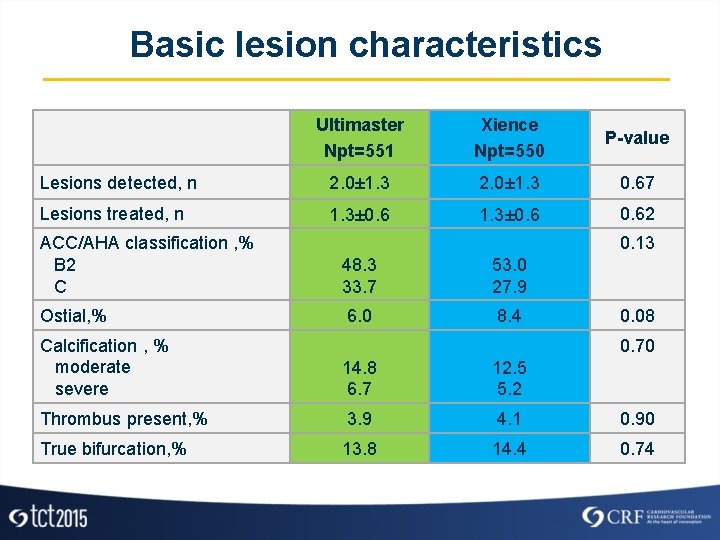
Basic lesion characteristics Ultimaster Npt=551 Xience Npt=550 P-value Lesions detected, n 2. 0± 1. 3 0. 67 Lesions treated, n 1. 3± 0. 62 0. 13 ACC/AHA classification , % B 2 C 48. 3 33. 7 53. 0 27. 9 Ostial, % 6. 0 8. 4 0. 08 0. 70 Calcification , % moderate severe 14. 8 6. 7 12. 5 5. 2 Thrombus present, % 3. 9 4. 1 0. 90 True bifurcation, % 13. 8 14. 4 0. 74
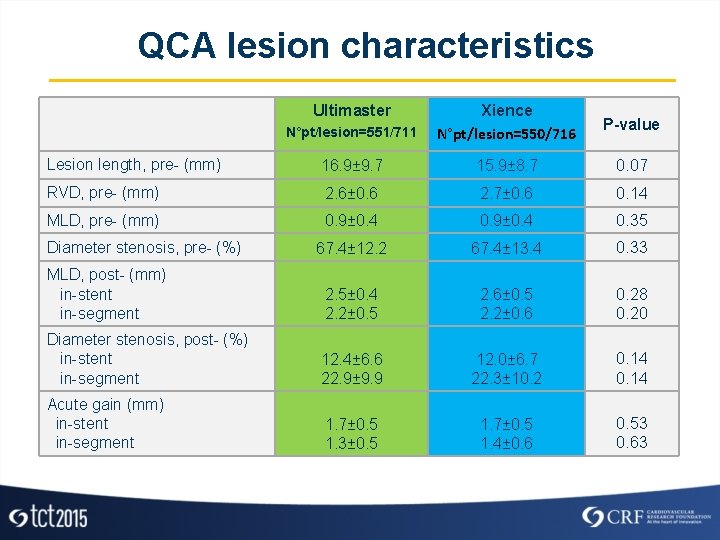
QCA lesion characteristics Ultimaster N°pt/lesion=551/711 Xience N°pt/lesion=550/716 P-value Lesion length, pre- (mm) 16. 9± 9. 7 15. 9± 8. 7 0. 07 RVD, pre- (mm) 2. 6± 0. 6 2. 7± 0. 6 0. 14 MLD, pre- (mm) 0. 9± 0. 4 0. 35 67. 4± 12. 2 67. 4± 13. 4 0. 33 MLD, post- (mm) in-stent in-segment 2. 5± 0. 4 2. 2± 0. 5 2. 6± 0. 5 2. 2± 0. 6 0. 28 0. 20 Diameter stenosis, post- (%) in-stent in-segment 12. 4± 6. 6 22. 9± 9. 9 12. 0± 6. 7 22. 3± 10. 2 0. 14 Acute gain (mm) in-stent in-segment 1. 7± 0. 5 1. 3± 0. 5 1. 7± 0. 5 1. 4± 0. 6 0. 53 0. 63 Diameter stenosis, pre- (%)
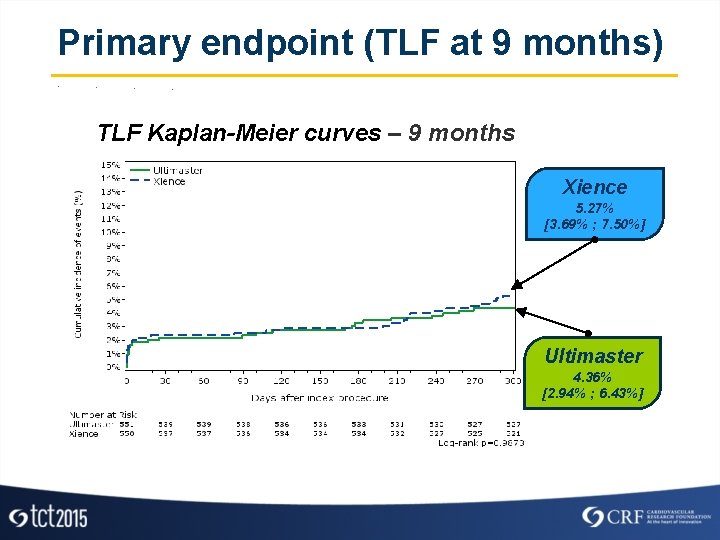
Primary endpoint (TLF at 9 months) TLF Kaplan-Meier curves – 9 months Xience 5. 27% [3. 69% ; 7. 50%] Ultimaster 4. 36% [2. 94% ; 6. 43%]
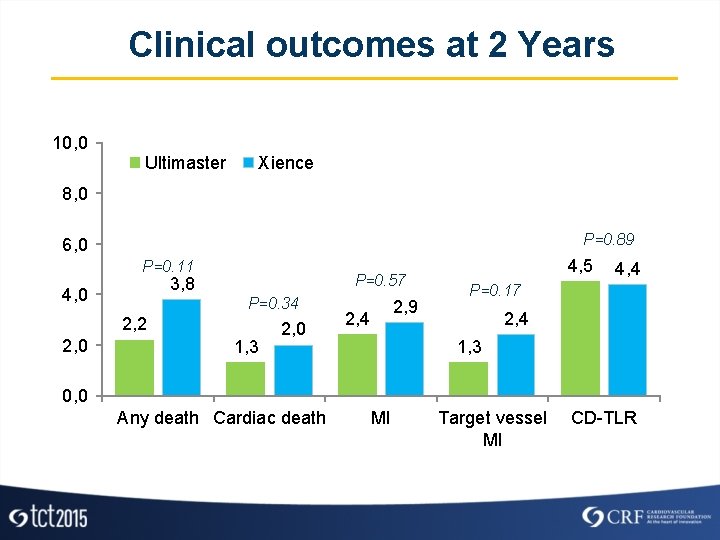
Clinical outcomes at 2 Years 10, 0 Ultimaster Xience 8, 0 P=0. 89 6, 0 P=0. 11 P=0. 57 3, 8 4, 0 P=0. 34 2, 2 2, 0 1, 3 2, 0 2, 9 2, 4 4, 5 4, 4 P=0. 17 2, 4 1, 3 0, 0 Any death Cardiac death MI Target vessel MI CD-TLR
![TLF at 24 months Ultimaster: 6. 5% [4. 8% ; 8. 9%] Xience: 6. TLF at 24 months Ultimaster: 6. 5% [4. 8% ; 8. 9%] Xience: 6.](http://slidetodoc.com/presentation_image_h2/aa44b77c568478b6d89cb0f477b7379b/image-13.jpg)
TLF at 24 months Ultimaster: 6. 5% [4. 8% ; 8. 9%] Xience: 6. 6% [4. 8% ; 9. 0%]
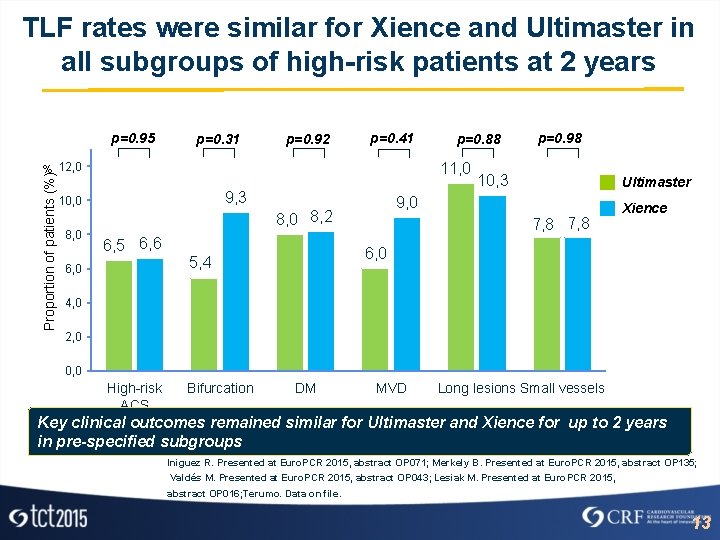
TLF rates were similar for Xience and Ultimaster in all subgroups of high-risk patients at 2 years p=0. 95 p=0. 31 p=0. 92 p=0. 41 11, 0 Proportion of patients (%) % 12, 0 9, 3 10, 0 8, 0 p=0. 88 6, 0 10, 3 Ultimaster 9, 0 8, 2 6, 5 6, 6 p=0. 98 7, 8 Xience 6, 0 5, 4 4, 0 2, 0 0, 0 High-risk ACS Bifurcation DM MVD Long lesions Small vessels Key clinical outcomes remained similar for Ultimaster and Xience for up to 2 years in pre-specified subgroups Iniguez R. Presented at Euro. PCR 2015, abstract OP 071; Merkely B. Presented at Euro. PCR 2015, abstract OP 135; Valdés M. Presented at Euro. PCR 2015, abstract OP 043; Lesiak M. Presented at Euro. PCR 2015, abstract OP 016; Terumo. Data on file. 13
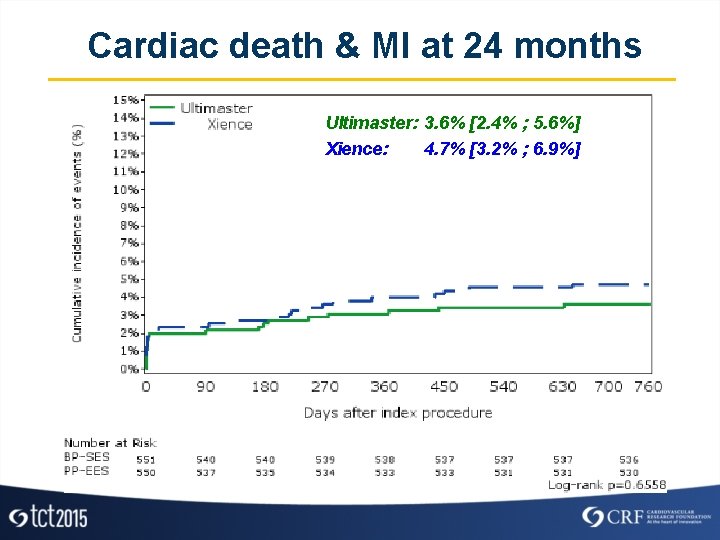
Cardiac death & MI at 24 months Ultimaster: 3. 6% [2. 4% ; 5. 6%] Xience: 4. 7% [3. 2% ; 6. 9%]
![POCE* at 24 months Ultimaster: 13. 3% [10. 7%; 16. 4%] Xience: 16. 9% POCE* at 24 months Ultimaster: 13. 3% [10. 7%; 16. 4%] Xience: 16. 9%](http://slidetodoc.com/presentation_image_h2/aa44b77c568478b6d89cb0f477b7379b/image-16.jpg)
POCE* at 24 months Ultimaster: 13. 3% [10. 7%; 16. 4%] Xience: 16. 9% [14. 0% ; 20. 3%] *POCE(Patient-Oriented Composite Endpoints) : all death, MI and revascularization
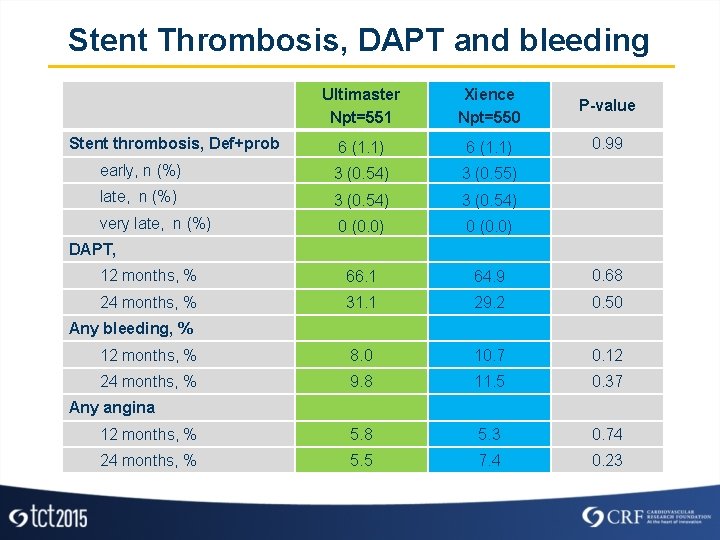
Stent Thrombosis, DAPT and bleeding Ultimaster Npt=551 Xience Npt=550 P-value 6 (1. 1) 0. 99 early, n (%) 3 (0. 54) 3 (0. 55) late, n (%) 3 (0. 54) very late, n (%) 0 (0. 0) 12 months, % 66. 1 64. 9 0. 68 24 months, % 31. 1 29. 2 0. 50 12 months, % 8. 0 10. 7 0. 12 24 months, % 9. 8 11. 5 0. 37 12 months, % 5. 8 5. 3 0. 74 24 months, % 5. 5 7. 4 0. 23 Stent thrombosis, Def+prob DAPT, Any bleeding, % Any angina

Conclusions • The long term follow up to 2 years of this large-scale global trial enrolling a patient population representing daily clinical practice, demonstrated good safety and efficacy profile of the thin strut abluminally coated bioresorbable polymer DES, Ultimaster; • The results were similar in both studied stents • There are no very late stent thrombosis reported between 12 and 24 months for Ultimaster, which further demonstrated good safety profile for this stent.

Back up slides

- Slides: 20