A Randomized Phase II Trial of Salvage Radiotherapy








- Slides: 10

A Randomized Phase II Trial of Salvage Radiotherapy with Standard vs Enhanced Androgen Deprivation Therapy (with Enzalutamide) in Patients with Post-Prostatectomy PSA Recurrences with Aggressive Disease Features RTOG 3506 Edwin Posadas, Hiram Gay, Ying Xiao, Todd Morgan, James Yu, Stephanie Pugh An RTOG Foundation collaboration with Pfizer/Astellas

Study Team Medical Oncology Edwin Posadas, MD Radiation Oncology Hiram Gay, MD Radiation Oncology Felix Feng, MD Medical Physics Ying Xiao, Ph. D, FAAPM Urology Todd Morgan, MD Quality of Life James B. Yu, MD Senior Statistician Stephanie Pugh, Ph. D 2

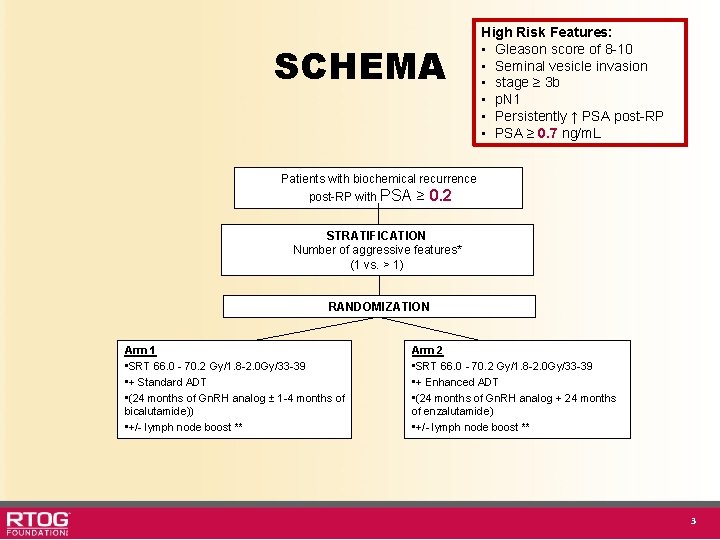
SCHEMA High Risk Features: • Gleason score of 8 -10 • Seminal vesicle invasion • stage ≥ 3 b • p. N 1 • Persistently ↑ PSA post-RP • PSA ≥ 0. 7 ng/m. L Patients with biochemical recurrence post-RP with PSA ≥ 0. 2 STRATIFICATION Number of aggressive features* (1 vs. > 1) RANDOMIZATION Arm 1 • SRT 66. 0 - 70. 2 Gy/1. 8 -2. 0 Gy/33 -39 • + Standard ADT • (24 months of Gn. RH analog ± 1 -4 months of bicalutamide)) • +/- lymph node boost ** Arm 2 • SRT 66. 0 - 70. 2 Gy/1. 8 -2. 0 Gy/33 -39 • + Enhanced ADT • (24 months of Gn. RH analog + 24 months of enzalutamide) • +/- lymph node boost ** 3

Study Endpoints Primary Objective: progression free survival • first occurrence of biochemical failure, clinical failure, or initiation of new unplanned anticancer treatment. • Biochemical failure: – Phoenix definition (PSA ≥ nadir+2 ng/m. L) – initiation of salvage hormones in the absence of BF Clinical failure: local, regional, or distant failure. time from randomization to the date of progression, death from any cause, or last known follow-up time • • Hypothesis • HR 0. 65 (enhanced/standard ADT) – Alpha error 0. 10 – Power 80% • 5 -year PFS (enhanced ADT) 56. 6% [hazard rate of 0. 114] • Assumptions: 5 year PFS(standard ADT) 41. 7% [ hazard rate of 0. 175] 4

Enrollment Updates • Currently we have 17 network sites activated and a total of 57 sites that are able to accrue. • • UT Southwestern Washington University Arizona Center for Cancer Care Nancy N & JC Lewis Cancer & Research Pavilion Decatur Memorial Hospital Well. Span Health Arizona Center for Cancer Care SUNY Upstate Medical University • William Beaumont • CIUSS de I’Estrie – Centre Hospitalier • Wayne State Universitaire de Sherbrooke- Fleurimont University/Karmanos Cancer • CHU de Quebec- L’Hotel-Dieu de Institute Quebec • Maine Medical Center • Sutter Medical Center • Valley View • Marin Cancer Care, Inc. • Dartmouth-Hitchcock Medical Center 5

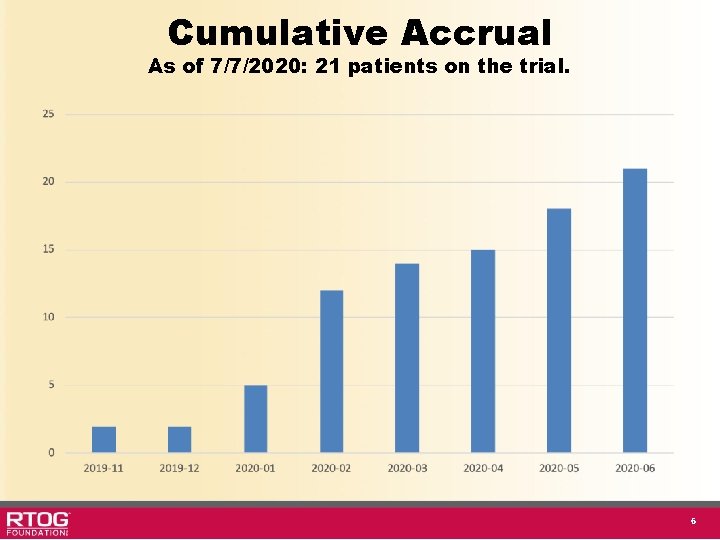
Cumulative Accrual As of 7/7/2020: 21 patients on the trial. 6

Enrollment Updates The RTOG Foundation Study Team and Study Chairs would like to congratulate and thank all the Investigators and the research staff at the following institutions for leading the charge and enrolling patients to this study! 7

Enrollment Updates • Top U. S. Site (9 patients enrolled). Brian C. Baumann, MD 8

Enrollment Updates • Top Canadian Site (5 patients enrolled). Myriam Bouchard, MD 9

Additional Points • RTOG Foundation study requirements: • Site feasibility questionnaire • Executed Master Purchased Service Agreement (MPSA) and Statement of Work (SOW) with RTOG Foundation must be in place • Site must meet RT credentialing requirements (IMRT/3 DCRT) • Complied with all regulatory requirements (Please see Study Specific Study Guide) • Reimbursements: RTOGF rates (Not NRG) • $7, 700/patient according to protocol milestones. • Startup costs: up to $3000 • IRB costs: up to $2000 • Maintenance costs: up to $1500 • Contacts: • RTOG-Membership@acr. org for MPSA/SOW/CDA • Erin Moye, Project Manager (RTOG 3506@acr. org) 10