A question about hepatic encephalopathy and SAEs in

A question about hepatic encephalopathy and SAEs in the PROSPECT Pilot Trial Dr. Erick Duan Presented at the CCCTG Halifax, NS, June 2016

Case • • • Patient enrolled in PROSPECT, with history of end stage liver disease (listed for tx), Recent variceal bleed + now worsening hepatic encephalopathy Is study product contributing to hepatic encephalopathy? Should they still be receiving study product? Hold? Discontinue? Is this a reportable serious adverse event (SAE)?
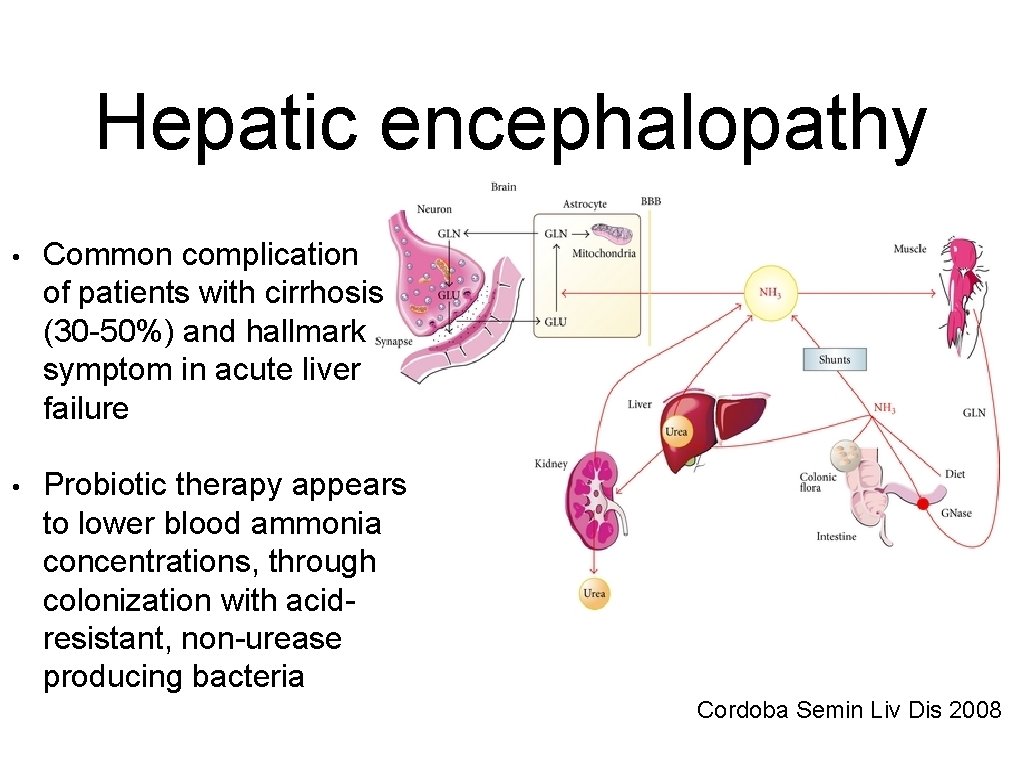
Hepatic encephalopathy • Common complication of patients with cirrhosis (30 -50%) and hallmark symptom in acute liver failure • Probiotic therapy appears to lower blood ammonia concentrations, through colonization with acidresistant, non-urease producing bacteria Cordoba Semin Liv Dis 2008

Probiotics in hepatic encephalopathy: the evidence • • • 14 RCTs (8/14 in India) Mostly outpatients Used JDAD for ROB assessment
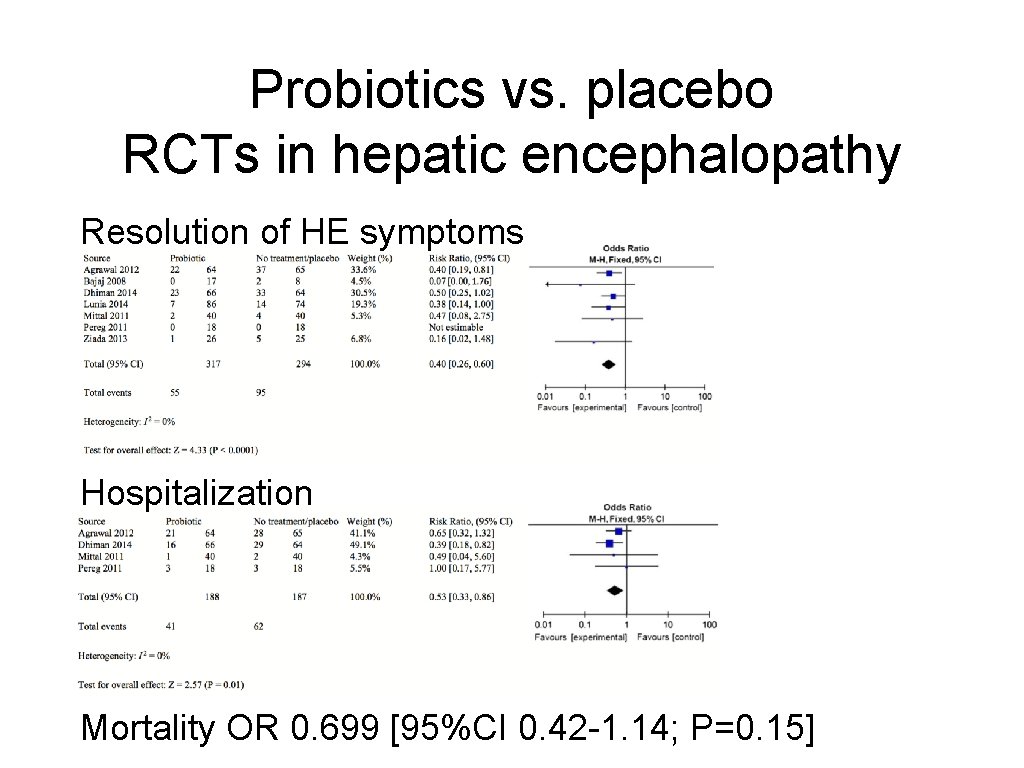
Probiotics vs. placebo RCTs in hepatic encephalopathy Resolution of HE symptoms Hospitalization Mortality OR 0. 699 [95%CI 0. 42 -1. 14; P=0. 15]
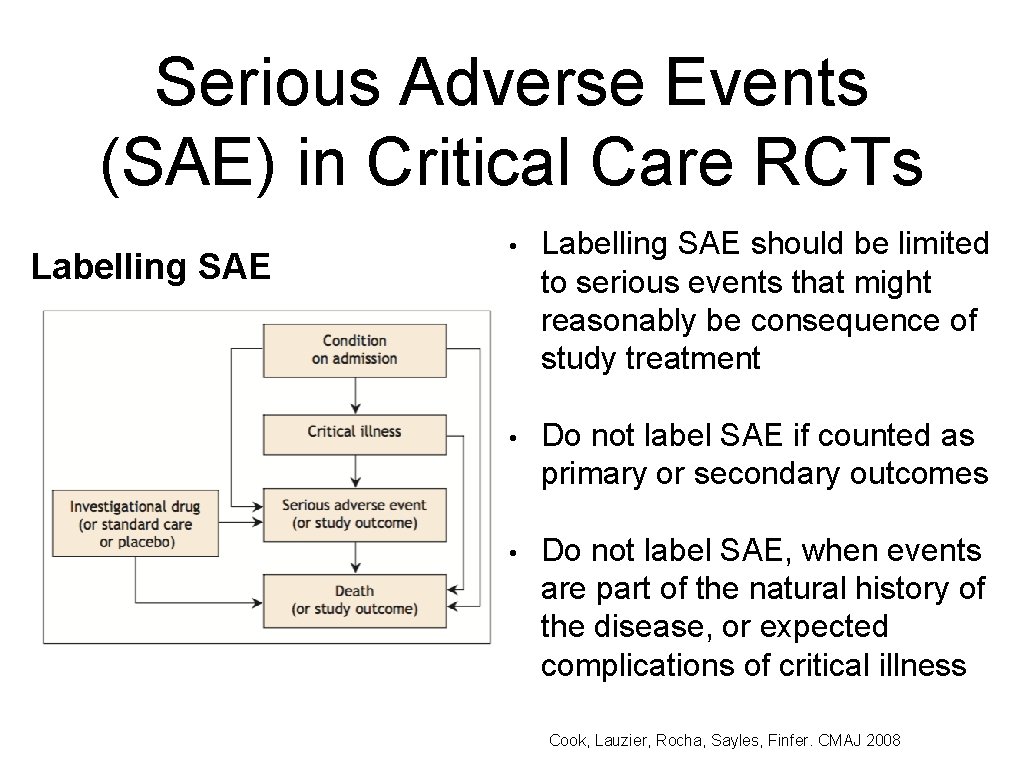
Serious Adverse Events (SAE) in Critical Care RCTs Labelling SAE • Labelling SAE should be limited to serious events that might reasonably be consequence of study treatment • Do not label SAE if counted as primary or secondary outcomes • Do not label SAE, when events are part of the natural history of the disease, or expected complications of critical illness Cook, Lauzier, Rocha, Sayles, Finfer. CMAJ 2008
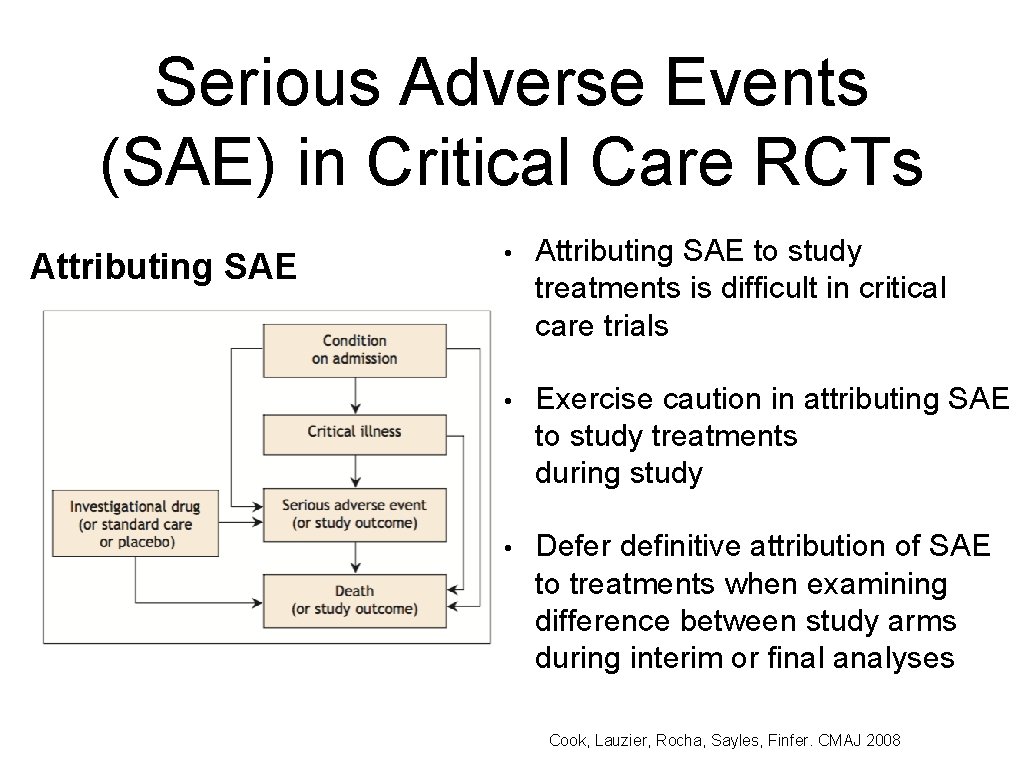
Serious Adverse Events (SAE) in Critical Care RCTs Attributing SAE • Attributing SAE to study treatments is difficult in critical care trials • Exercise caution in attributing SAE to study treatments during study • Defer definitive attribution of SAE to treatments when examining difference between study arms during interim or final analyses Cook, Lauzier, Rocha, Sayles, Finfer. CMAJ 2008

Case resolution • Study product unlikely to be contributing to hepatic encephalopathy • Option for the clinical team to hold study product if continued clinical concern • Do not label as SAE as hepatic encephalopathy is part of the natural history of end stage liver disease and not plausibly consequence of the study product
- Slides: 8