A Prospective Observational Pilot Study Evaluating the Impact
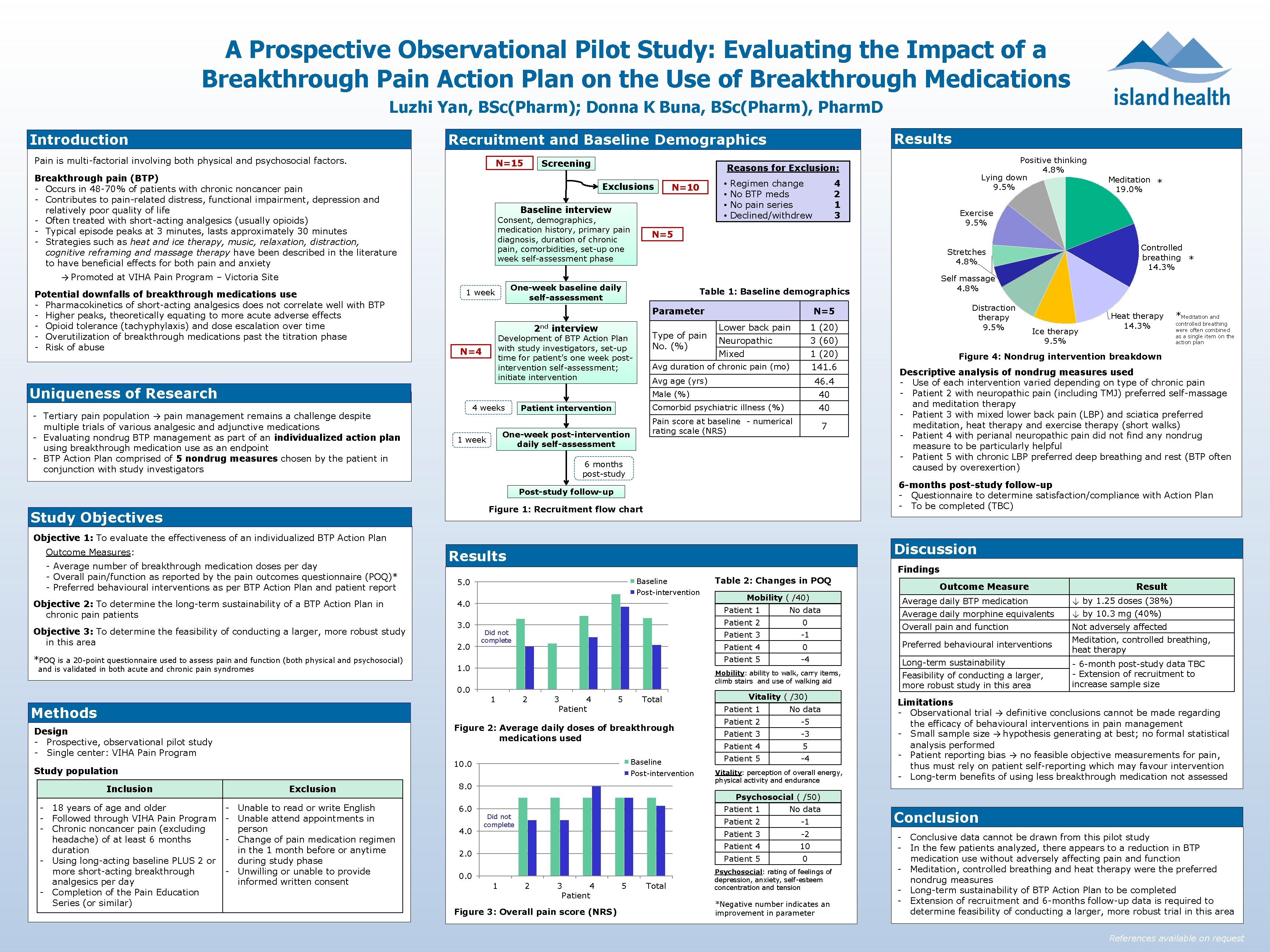
A Prospective Observational Pilot Study: Evaluating the Impact of a Breakthrough Pain Action Plan on the Use of Breakthrough Medications Luzhi Yan, BSc(Pharm); Donna K Buna, BSc(Pharm), Pharm. D Introduction Results Recruitment and Baseline Demographics Pain is multi-factorial involving both physical and psychosocial factors. N=15 Breakthrough pain (BTP) - Occurs in 48 -70% of patients with chronic noncancer pain - Contributes to pain-related distress, functional impairment, depression and relatively poor quality of life - Often treated with short-acting analgesics (usually opioids) - Typical episode peaks at 3 minutes, lasts approximately 30 minutes - Strategies such as heat and ice therapy, music, relaxation, distraction, cognitive reframing and massage therapy have been described in the literature to have beneficial effects for both pain and anxiety Screening Positive thinking 4. 8% Lying down 9. 5% Reasons for Exclusion: Exclusions N=10 Baseline interview Consent, demographics, medication history, primary pain diagnosis, duration of chronic pain, comorbidities, set-up one week self-assessment phase • • Regimen change No BTP meds No pain series Declined/withdrew 4 2 1 3 Exercise 9. 5% N=5 One-week baseline daily self-assessment 1 week interview Type of pain No. (%) Development of BTP Action Plan with study investigators, set-up time for patient’s one week postintervention self-assessment; initiate intervention N=4 Lower back pain Neuropathic Mixed 1 (20) 3 (60) 1 (20) 141. 6 46. 4 40 40 Avg age (yrs) Male (%) - Tertiary pain population → pain management remains a challenge despite multiple trials of various analgesic and adjunctive medications - Evaluating nondrug BTP management as part of an individualized action plan using breakthrough medication use as an endpoint - BTP Action Plan comprised of 5 nondrug measures chosen by the patient in conjunction with study investigators Comorbid psychiatric illness (%) Patient intervention Pain score at baseline - numerical rating scale (NRS) One-week post-intervention daily self-assessment 1 week 7 6 months post-study Figure 1: Recruitment flow chart Objective 1: To evaluate the effectiveness of an individualized BTP Action Plan Outcome Measures: - Average number of breakthrough medication doses per day - Overall pain/function as reported by the pain outcomes questionnaire (POQ)* - Preferred behavioural interventions as per BTP Action Plan and patient report Objective 2: To determine the long-term sustainability of a BTP Action Plan in chronic pain patients Objective 3: To determine the feasibility of conducting a larger, more robust study in this area *POQ is a 20 -point questionnaire used to assess pain and function (both physical and psychosocial) and is validated in both acute and chronic pain syndromes Baseline Post-intervention 4. 0 Did not complete 1. 0 0. 0 2 Methods 3 4 Patient 5 Total Figure 2: Average daily doses of breakthrough medications used Design - Prospective, observational pilot study - Single center: VIHA Pain Program Baseline 10. 0 Study population Post-intervention Inclusion Exclusion 8. 0 - 18 years of age and older - Followed through VIHA Pain Program - Chronic noncancer pain (excluding headache) of at least 6 months duration - Using long-acting baseline PLUS 2 or more short-acting breakthrough analgesics per day - Completion of the Pain Education Series (or similar) - Unable to read or write English - Unable attend appointments in person - Change of pain medication regimen in the 1 month before or anytime during study phase - Unwilling or unable to provide informed written consent 6. 0 Table 2: Changes in POQ Mobility ( /40) Figure 4: Nondrug intervention breakdown Descriptive analysis of nondrug measures used - Use of each intervention varied depending on type of chronic pain - Patient 2 with neuropathic pain (including TMJ) preferred self-massage and meditation therapy - Patient 3 with mixed lower back pain (LBP) and sciatica preferred meditation, heat therapy and exercise therapy (short walks) - Patient 4 with perianal neuropathic pain did not find any nondrug measure to be particularly helpful - Patient 5 with chronic LBP preferred deep breathing and rest (BTP often caused by overexertion) Outcome Measure ↓ by 1. 25 doses (38%) Average daily morphine equivalents ↓ by 10. 3 mg (40%) Overall pain and function Not adversely affected Meditation, controlled breathing, heat therapy No data Patient 2 0 Patient 3 -1 Patient 4 0 Preferred behavioural interventions Patient 5 -4 Long-term sustainability Vitality ( /30) Patient 1 No data Patient 2 -5 Patient 3 -3 Patient 4 5 Patient 5 -4 Vitality: perception of overall energy, physical activity and endurance Result Average daily BTP medication Patient 1 Mobility: ability to walk, carry items, climb stairs and use of walking aid 1 controlled breathing were often combined as a single item on the action plan Findings 5. 0 2. 0 *Meditation and Discussion Results 3. 0 Ice therapy 9. 5% Heat therapy 14. 3% 6 -months post-study follow-up - Questionnaire to determine satisfaction/compliance with Action Plan - To be completed (TBC) Post-study follow-up Study Objectives Distraction therapy 9. 5% N=5 Avg duration of chronic pain (mo) Uniqueness of Research 4 weeks Self massage 4. 8% Table 1: Baseline demographics Parameter 2 nd Controlled breathing * 14. 3% Stretches 4. 8% → Promoted at VIHA Pain Program – Victoria Site Potential downfalls of breakthrough medications use - Pharmacokinetics of short-acting analgesics does not correlate well with BTP - Higher peaks, theoretically equating to more acute adverse effects - Opioid tolerance (tachyphylaxis) and dose escalation over time - Overutilization of breakthrough medications past the titration phase - Risk of abuse Meditation * 19. 0% Feasibility of conducting a larger, more robust study in this area - 6 -month post-study data TBC - Extension of recruitment to increase sample size Limitations - Observational trial → definitive conclusions cannot be made regarding the efficacy of behavioural interventions in pain management - Small sample size → hypothesis generating at best; no formal statistical analysis performed - Patient reporting bias → no feasible objective measurements for pain, thus must rely on patient self-reporting which may favour intervention - Long-term benefits of using less breakthrough medication not assessed Psychosocial ( /50) 4. 0 Did not complete 2. 0 0. 0 1 2 3 4 Patient Figure 3: Overall pain score (NRS) 5 Total Patient 1 No data Patient 2 -1 Patient 3 -2 Patient 4 10 Patient 5 0 Psychosocial: rating of feelings of depression, anxiety, self-esteem concentration and tension *Negative number indicates an improvement in parameter Conclusion - Conclusive data cannot be drawn from this pilot study - In the few patients analyzed, there appears to a reduction in BTP medication use without adversely affecting pain and function - Meditation, controlled breathing and heat therapy were the preferred nondrug measures - Long-term sustainability of BTP Action Plan to be completed - Extension of recruitment and 6 -months follow-up data is required to determine feasibility of conducting a larger, more robust trial in this area References available on request
- Slides: 1