A Primer on Anabolic Steroid Use in HIV

A Primer on Anabolic Steroid Use in HIV Infection Antonio E. Urbina, M. D. Medical Director of HIV/AIDS Education and Training St. Vincent Catholic Medical Center-Manhattan A Local Performance Site of the New York/New Jersey AETC

Anabolic Steroids Definitions n Commonly Used Agents n Indications/Diagnosis n Hypogonadism n HIV Wasting n Adverse Effects n Studies n Management n

Definitions Androgens: all male sex hormones, usually testosterone, but also testosterone derivatives n Androgenic: refers to masculinizing properties such as libido, aggression, acne, hair growth and loss n Anabolic: refers to assimilation of nitrogen into tissue (muscle growth) n Cannot completely separate one from the other n
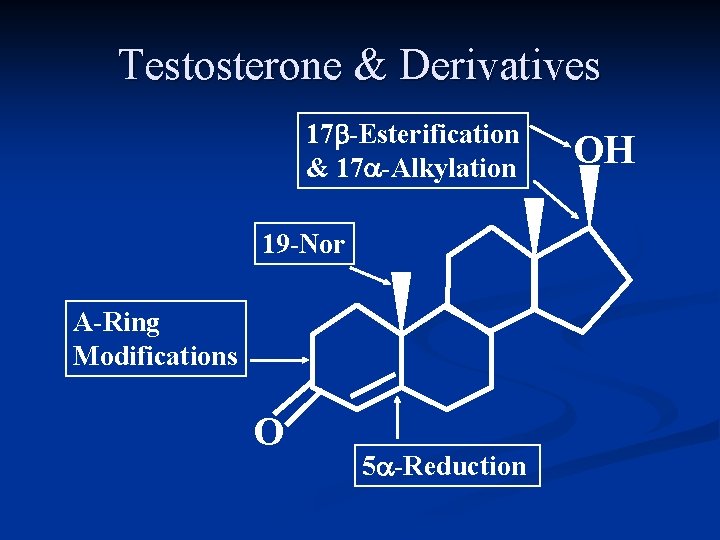
Testosterone & Derivatives 17 b-Esterification & 17 a-Alkylation 19 -Nor A-Ring Modifications O 5 a-Reduction OH

Target Organs and Physiological Effects of Testosterone and Metabolites n n n n CNS ( libido, well-being, aggression, spatial cognition) Hypothalamus/ Pituitary ( Gn. RH, LH, FSH; GH) Larynx (lowers voice) Breast (E 2 size) Liver ( SHBG, HDL) Kidney ( erythropoietin) Genitals ( development, spermatogenesis, erections) Prostate ( size, secretions) n n n Skin ( facial/ body hair, sebum production) Bone ( BMD) Muscle ( lean mass, strength) Adipose Tissue ( lipolysis, abdominal fat) Blood ( hematocrit) Immune system ( autoantibody production)

Androgenic vs Anabolic n Androgenic n n n Testosterone (IM) Androgel (transdermal) Androderm (transdermal) n Anabolic n n n Deca-Durabolin (IM) Oxandrin (oral) Anadrol (oral)
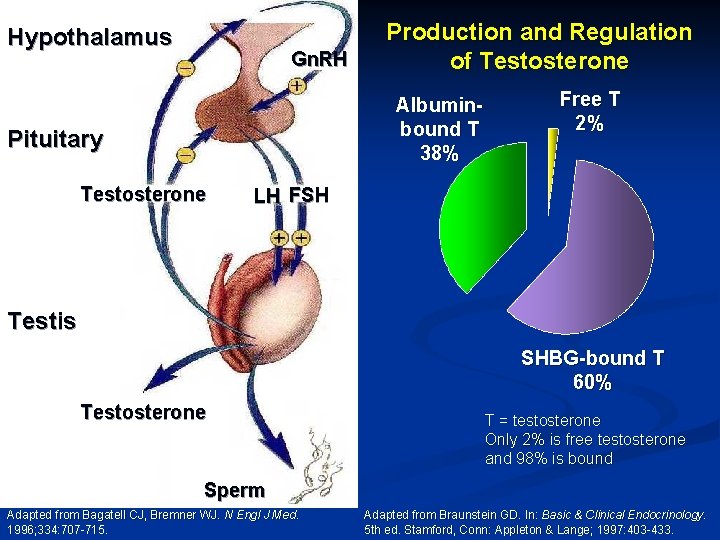
Hypothalamus Gn. RH Production and Regulation of Testosterone Albuminbound T 38% Pituitary Testosterone Free T 2% LH FSH Testis SHBG-bound T 60% Testosterone T = testosterone Only 2% is free testosterone and 98% is bound Sperm Adapted from Bagatell CJ, Bremner WJ. N Engl J Med. 1996; 334: 707 -715. Adapted from Braunstein GD. In: Basic & Clinical Endocrinology. 5 th ed. Stamford, Conn: Appleton & Lange; 1997: 403 -433.

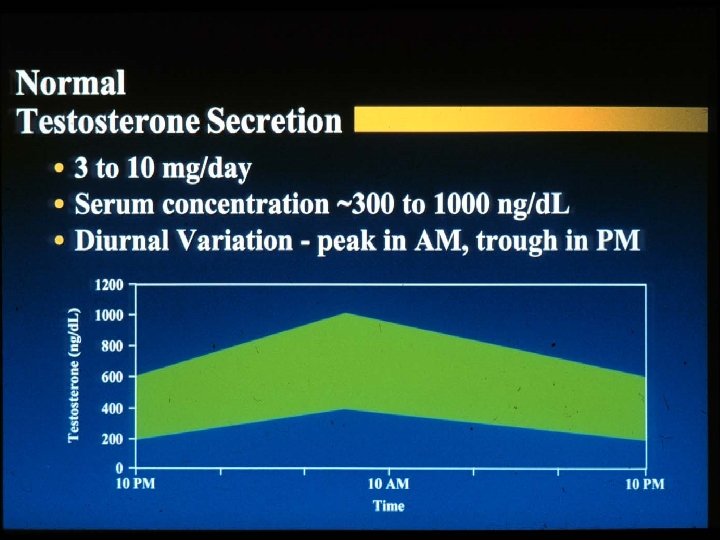
Laboratory Diagnosis and Workup of Primary vs. Secondary Hypogonadism n Hypogonadism in adult male - presence of signs or symptoms of hypogonadism with confirmation by laboratory testing n Laboratory Testing: AM total testosterone x 2 n n Normally diurnal rhythm with highest levels in AM Free testosterone (2%) - (sometimes even if total normal) Bioavailable testosterone - free (2%) plus loosely bound to albumin (38%) - (total 40%) n 60% tightly bound to SHBG

Diagnosis and Workup of Primary vs. Secondary Hypogonadism (Cont. ) n LH and FSH - (if low T is established or as initial workup); Repeat with 2 samples taken 20 -30 min. apart and pooled n FSH and LH secreted in short pulses n Prolactin ; Estradiol (if gynecomastia or testicular or adrenal tumor suspected) n Definitive diagnosis of T deficiency on the basis of laboratory tests for the aging male has not been established n n n <200 ng/d. L clearcut total T may not be an accurate measurement if there is increased or decreased SHBG deficiency considered at 200 -350 ng/d. L (depending on assay) or if the T or bioavailable T (or free T) is in the lower range of normal

Diagnosis and Workup of Primary vs. Secondary Hypogonadism (Cont. ) n n If studies indicate clear primary hypogonadism n Low T with reciprocal elevated FSH and LH n Then pituitary workup not indicated If studies indicate secondary hypogonadism or combined: n Low T with low FSL/LH or n Low T with normal or high-normal FSH/LH - not appropriately elevated Then MRI of pituitary indicated n MRI of pituitary always indicated if elevated prolactin n Other pituitary testing may be necessary Stimulation tests generally of limited clinical value to distinguish 1º from 2º or pituitary from hypothalamic defect AACE Guidelines, Endocrine Practice: 8, 439, 2002

Medications (common) contribute to hypogonadism n n n n Glucocoticoids - testicular and pituitary/hypothalamic ketoconazole - inhibitor of gonadal and adrenal steroidogenesis spironolactone - aldosterone antagonist; and blocks androgen at receptor, inhibits androgen biosynthesis, interferes with binding T to SHBG cimetidine - weak antiandrogen finasteride (propecia) - inhibitor of type. II 5 alpha reductase, antiandrogen flutamide and other antiandrogens megastrol acatate (megace) - decreased androgen production androgen mediated action

Testosterone Deficiency with Aging n Decline in Testosterone with age n n n Decrease in testosterone production Decrease in testosterone clearance Increase in SHBG n n n may be due to higher serum estradiol levels from increased adipose tissue Therefore, bioavailable T decreases more than total T Circadian rhythm (higher T values in AM) lost with aging Tenover, L. J. End. Metab. Clinics NA: 27, 969, 1998

Prevalence and Diagnosis of Hypogonadism In HIV n Approximately 30% of HIV+ men and 50% of men with AIDS are hypogonadal n Correlated with stage of disease, lymphocyte depletion, weight loss, reduced muscle mass, and decreased functional status n Free testosterone is the preferred measurement n Sex hormone binding globulin (SHBG) increases in men with HIV-infection Dobs AS. Baillière’s Clin Endocrinol Metab. 1998; 12: 379 -390. Grinspoon S, et al. J Clin Endocrinol Metab. 2000; 85: 60 -65. Wiley S, et al. AIDS. 2003; 17(2): 183 -8. Habasque C, et al. Mol Hum Reprod 2002 8(5): 419 -25.

Effects of Testosterone in Hypogonadal Men With AIDS Wasting Study design n 6 -month, randomized, placebo-controlled trial n 51 men with hypogonadism and AIDS wasting n Randomly assigned to receive testosterone enanthate 300 mg or placebo IM every 3 weeks Grinspoon S, et al. Ann Intern Med. 1998; 129: 18 -26.
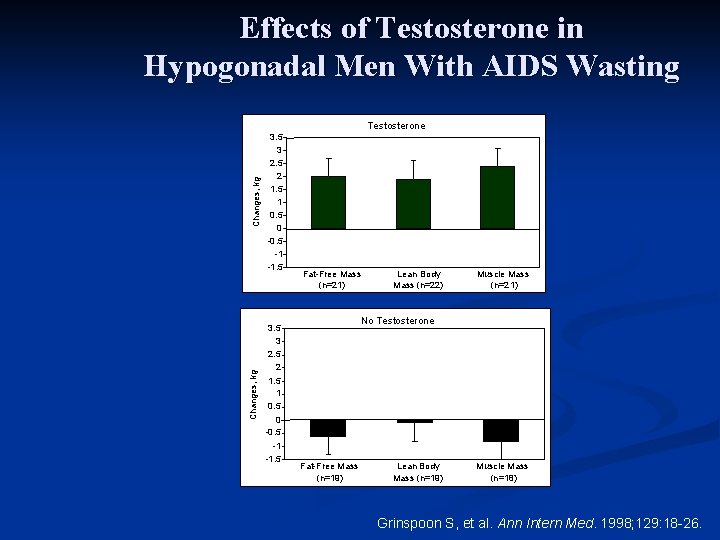
Effects of Testosterone in Hypogonadal Men With AIDS Wasting Changes, kg Testosterone 3. 5 3 2. 5 2 1. 5 1 0. 5 0 -0. 5 -1 -1. 5 Fat-Free Mass (n=21) Lean Body Mass (n=22) Muscle Mass (n=21) No Testosterone 3. 5 3 2. 5 2 1. 5 1 0. 5 0 -0. 5 -1 -1. 5 Fat-Free Mass (n=19) Lean Body Mass (n=19) Muscle Mass (n=18) Grinspoon S, et al. Ann Intern Med. 1998; 129: 18 -26.

IM Testosterone Therapy and Resistance Exercise in Hypogonadal HIV+ Men Study design n A 16 -week, placebo-controlled, double-blind, randomized trial 61 HIV+ men, aged 18 to 50 years old Randomized to 1 of 4 groups n Placebo, no exercise (n=14) n Testosterone enanthate 100 mg/wk, no exercise (n=17) n Placebo and exercise (n=15) n Testosterone and exercise (n=15) Bhasin S, et al. JAMA. 2000; 283: 763 -770.

IM Testosterone Therapy and Resistance Exercise in Hypogonadal HIV+ Men Study results n n n weight in testosterone alone or exercise alone maximum voluntary muscle strength in all 4 treatment groups Greater in thigh muscle volume in T alone or PRE alone lean body mass with testosterone or T + PRE hemoglobin in testosterone recipients Bhasin S, et al. JAMA. 2000; 283: 763 -770.

IM Testosterone and/or Exercise in Eugonadal Men With AIDS Wasting Study design n 12 -week randomized, controlled trial n 54 eugonadal men with AIDS wasting n Randomized to testosterone enanthate 200 mg/wk or placebo and progressive resistance training (3 x/wk) or no exercise Grinspoon S, et al. Ann Intern Med. 2000; 133: 348 -355.
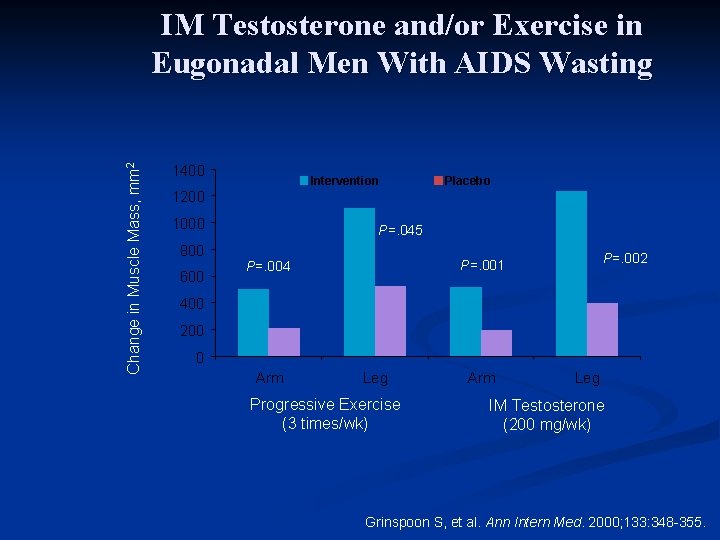
Change in Muscle Mass, mm 2 IM Testosterone and/or Exercise in Eugonadal Men With AIDS Wasting 1400 Intervention Placebo 1200 1000 800 600 P=. 045 P=. 002 P=. 001 P=. 004 400 200 0 Arm Leg Progressive Exercise (3 times/wk) Arm Leg IM Testosterone (200 mg/wk) Grinspoon S, et al. Ann Intern Med. 2000; 133: 348 -355.

Background n n Despite HAART, HIV-wasting is still very common, affecting up to 30% of patients in the US and Europe (Wanke et al. 2000, Balslef et al. 1997) Death due to wasting in patients with AIDS is related to the magnitude of tissue depletion, independent of the underlying cause (Kotler DP et al. Am J Clin Nutr. 1989)

AIDS-Wasting Syndrome (AWS) n 10% involuntary weight loss in last 12 months n 7. 5% involuntary weight loss in last 6 months n 5% loss of BCM in last 6 months n Men: BCM <35% B. W. and BMI <27 kg/m 2 Women: BCM <23% B. W. and BMI <27 kg/m 2 Polsky, Kotler and Steinhart.

Major Causes of AWS n n n Reduced food intake Malabsorption/diarrhea Infections HIV-enteropathy Altered metabolism Medications

Treatment Strategies of AWS n Appetite stimulants (megestrol acetate, dronabinol) n Nutritional supplements (beta-hydroxy-beta-methylbutyrate, glutamine, arginine, vitamins, micronutrients, protein) n Cytokine inhibitors (thalidomide, pentoxifyllin) n Anabolic proteins (human growth hormone, Insulinlike growth factor) n Anabolic steroids n Physical exercise

Oxymetholone as Therapy to Maintain Body Composition in HIV-Positive Subjects (Urbina, A. 2003) n n n Open label, single center, Phase III study involving pts who have received at least 4 months of prior anabolic (nandrolone or oxandrolone) for a past or current dx of wasting Pts were then switched to oxymetholone 50 mg QD and followed for 6 months Efficacy and safety evaluations performed at 4 week interval from baseline through week 12, then q 6 weeks until week 24

Oxymetholone as Therapy to Maintain (Urbina, A 2003) n Study Objectives Maintenance (no change) or improvement (increase) in BCM as measured by BIA n Evaluate the effects on HIV replication as measured by change in CD 4 and viral load from baseline n Evaluate clinical laboratory (hematology, lipids, LFTs, testosterone, PSA) and vital sign measurements n

Oxymetholone as Therapy to Maintain (Urbina, A 2003) 16 HIV+ men were successfully switched to oxymetholone n BCM was maintained over the 24 week period with a mean increase of 2. 2 lbs (p=. 091) n Increase in FFM for all weeks with significant increase at 24 weeks (3. 1 lbs, p=0. 027) n

Oxymetholone to Maintain (Urbina, A 2003) Lipids decreased over time (especially HDL and LDL) n Overall, no clinically significant effect on LFTs n CD 4 values increased over time (mean of 21 cell increase) n Testosterone levels increased by week 18 and 24 n
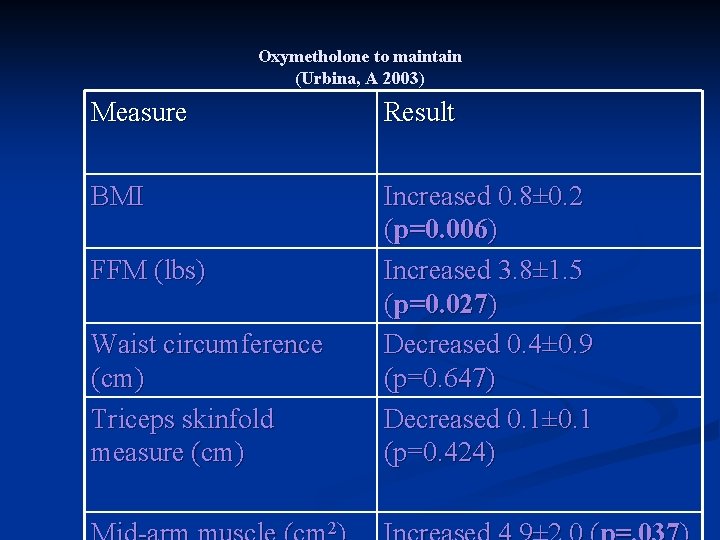
Oxymetholone to maintain (Urbina, A 2003) Measure Result BMI Increased 0. 8± 0. 2 (p=0. 006) Increased 3. 8± 1. 5 (p=0. 027) Decreased 0. 4± 0. 9 (p=0. 647) Decreased 0. 1± 0. 1 (p=0. 424) FFM (lbs) Waist circumference (cm) Triceps skinfold measure (cm) 2
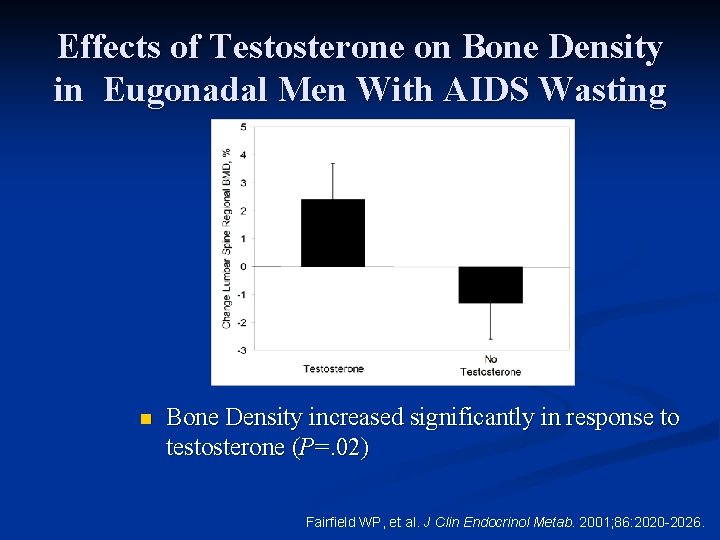
Effects of Testosterone on Bone Density in Eugonadal Men With AIDS Wasting n Bone Density increased significantly in response to testosterone (P=. 02) Fairfield WP, et al. J Clin Endocrinol Metab. 2001; 86: 2020 -2026.
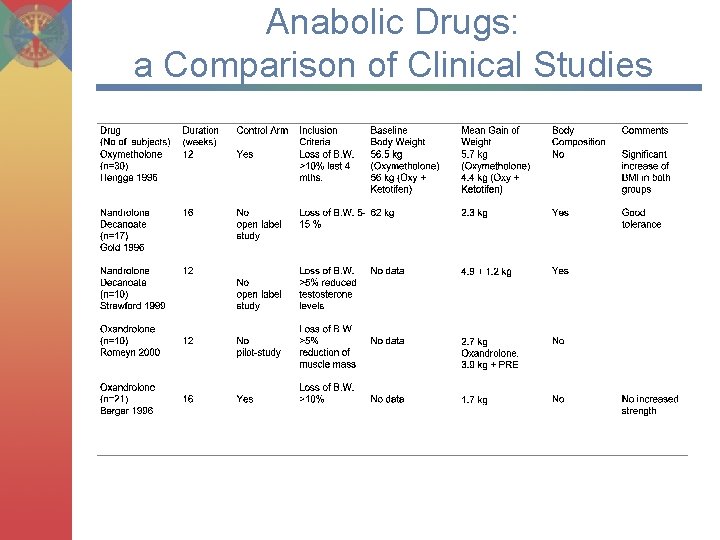
Anabolic Drugs: a Comparison of Clinical Studies

Depression Indices in Hypogonadal HIV-Infected Men Study design n 6 -month, randomized, placebo-controlled trial n 51 men with hypogonadism and AIDS wasting n Randomly assigned to receive testosterone enanthate 300 mg or placebo IM every 3 weeks n 10 age and weight matched men with AIDS wasting who were not hypogonadal were recruited as a control group for baseline comparison only and did not receive testosterone Grinspoon S. et al. J Clin Endocrinol Metab. 2000; 85: 60 -65.

Depression Indices in Hypogonadal HIV-Infected Men n Beck Depression Inventory n Administered to all patients (hypogondal and eugonadal) at baseline and again after 6 months to the hypogonadal patients in the randomized study n Normal range <10 Grinspoon S. et al. J Clin Endocrinol Metab. 2000; 85: 60 -65.
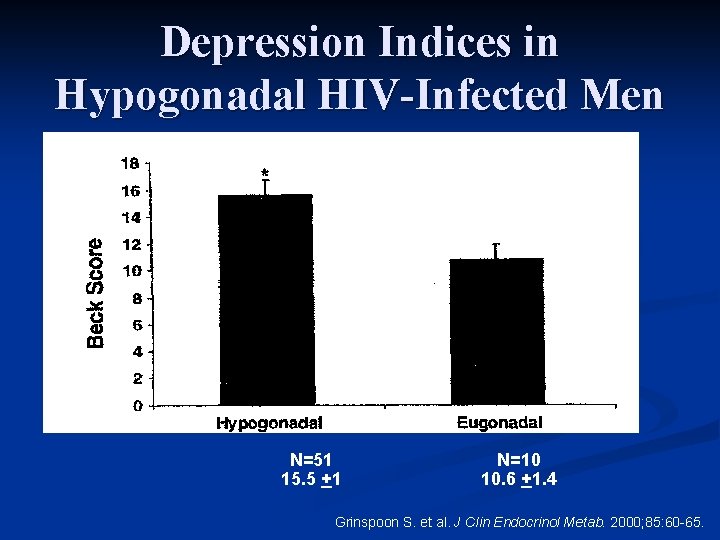
Depression Indices in Hypogonadal HIV-Infected Men *P=. 02 N=51 15. 5 +1 N=10 10. 6 +1. 4 Grinspoon S. et al. J Clin Endocrinol Metab. 2000; 85: 60 -65.
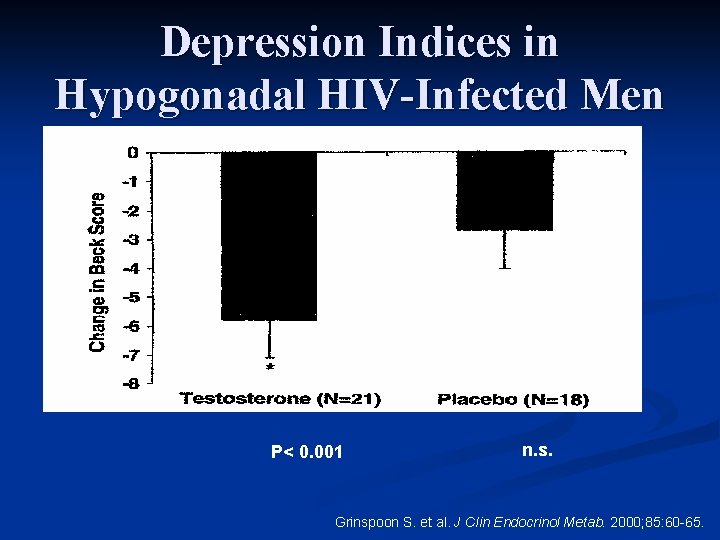
Depression Indices in Hypogonadal HIV-Infected Men P< 0. 001 n. s. Grinspoon S. et al. J Clin Endocrinol Metab. 2000; 85: 60 -65.

ADVERSE EFFECTS Acne n Hair loss n Increased libido (supraphysiologic) n Insomnia n Testicular atrophy n Agressiveness (supraphysiologic) n Hypertension n

ADVERSE EFFECTS Gynecomastia n Virilization n Polycythemia n Increase in transaminases n Hepatis peliosis n Inceased risk with co-infected n Hyperlipidemia (↓HDL) n Prostatic enlargement n

Algorithim for Use of Anabolics n Select appropriate patient Wasting, post-inpatient, after tx of OI n Hypogonadol vs eugonadol n n Free or bioavilable n Prior to initiation n Check LFTs, CBC, PSA and DRE

Algorithim for Use of Anabolic Steroids n Treatment for short duration n n 3 -6 months Monitoring of lab values Testosterone n LFT’s n CBC n Lipid panel n PSA n

Monitoring PSA during Androgen Therapy n Elevated serum PSA levels before or during therapy must be investigated. n Measure PSA at baseline, 6 months, then annually n Interval increase of PSA of > 0. 75 ng/ml (even if still in “normal” range) requires investigation
- Slides: 42