A Presentation By Ms Priya Vishwas waghambare MSc



A Presentation By Ms. Priya Vishwas waghambare. MSc. B. Ed. (Chemistry)

Metals and Nonmetals Laboratory preparation of Carbon dioxide

Aim- To prepare and study the properties of carbon dioxide. Apparatus- Flat bottom flask, two holed rubber cork , thistle funnel, gas delivery tube, gas jar with lid , stand candles, match box, test tubes, deflagrating spoon. Chemicals- Pieces of marble / shahabad stone , dil. Hydrochloric acid, blue and red litmus paper, freshly prepared lime water, magnesium ribbon.
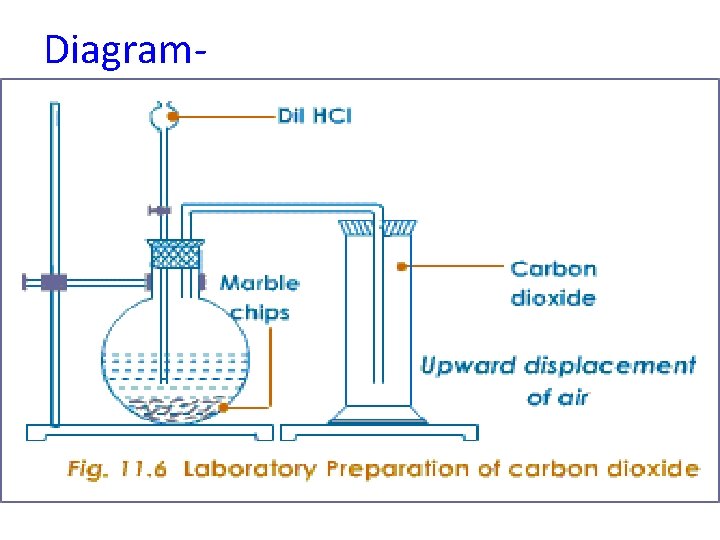
Diagram-
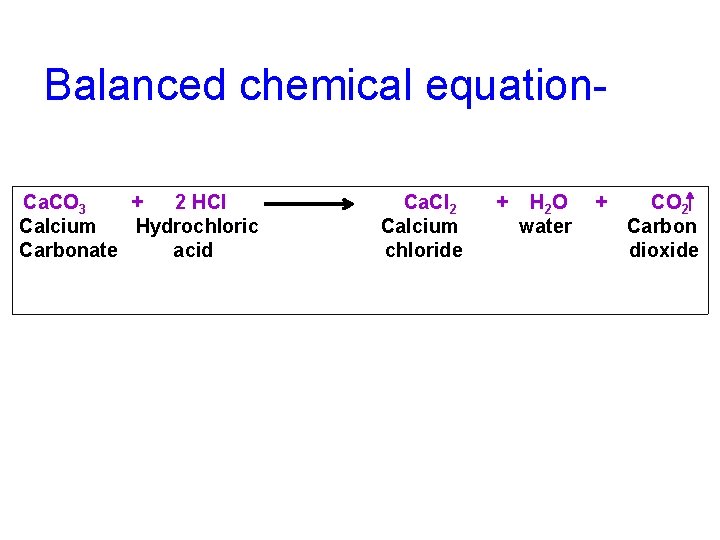
Balanced chemical equation. Ca. CO 3 + 2 HCl Calcium Hydrochloric Carbonate acid Ca. Cl 2 Calcium chloride + H 2 O water + CO 2 Carbon dioxide

Observation and Inference – Sr. Test No. 1 Colour of the gas 2 3 Effect of gas on moist blue & red litmus paper Insert the lightened candle in gas jar with the help of deflagrating spoon Observation Colourless Inference Co 2 is colourless Moist blue litmus paper CO 2 is acidic turns red Candle extinguished and CO 2 neither gas doesn’t burn supports combustion not is combustible
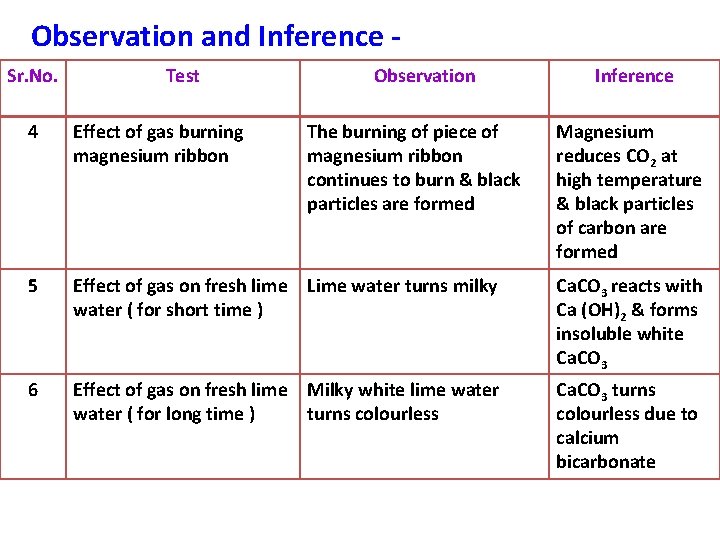
Observation and Inference Sr. No. Test Observation The burning of piece of magnesium ribbon continues to burn & black particles are formed Inference 4 Effect of gas burning magnesium ribbon Magnesium reduces CO 2 at high temperature & black particles of carbon are formed 5 Effect of gas on fresh lime Lime water turns milky water ( for short time ) Ca. CO 3 reacts with Ca (OH)2 & forms insoluble white Ca. CO 3 6 Effect of gas on fresh lime Milky white lime water ( for long time ) turns colourless Ca. CO 3 turns colourless due to calcium bicarbonate
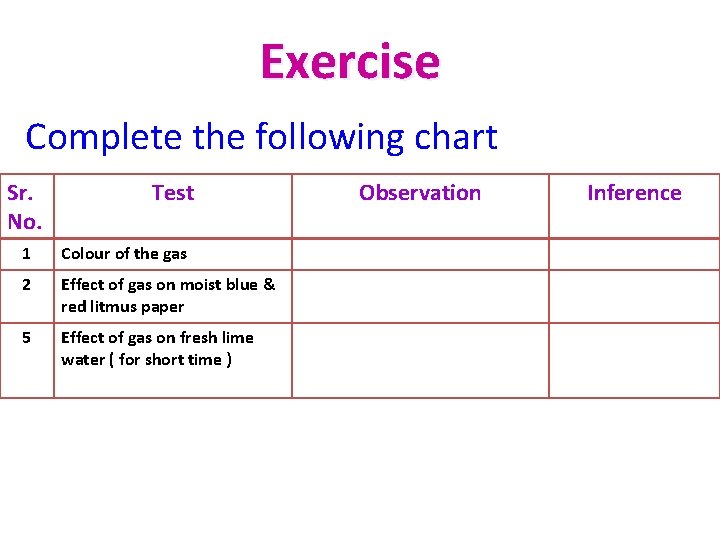
Exercise Complete the following chart Sr. No. Test 1 Colour of the gas 2 Effect of gas on moist blue & red litmus paper 5 Effect of gas on fresh lime water ( for short time ) Observation Inference

Thank You
- Slides: 11