A Phase 3 Doubleblind Placebocontrolled Study of the

A Phase 3, Double-blind, Placebocontrolled Study of the Effectiveness and Safety of Single Oral Doses of Secnidazole 2 g for the Treatment of Women with Bacterial Vaginosis JR Schwebke 1; FG Morgan 2; W Koltun 3; P Nyirjesy 4 University of Alabama at Birmingham School of Medicine, Birmingham, AL 1; Tidewater Physicians for Women, Virginia Beach, VA 2; Medical Center for Clinical Research, San Diego, CA 3; Drexel University College of Medicine, Philadelphia, PA 4
Background • SYM-1219 is an oral granule formulation of secnidazole, a 5 -nitroimidazole Secnidazole – Under development in the US as a single-dose treatment for bacterial vaginosis (BV) • Secnidazole has a longer halflife (~17 h) than metronidazole (~8 h) Pentikis HS et al. Adv Ther. 2017; 33: 2229 -2241. Metronidazole 2

Study Design • 189 women were randomized 2: 1 at 21 US sites to a single oral dose of secnidazole 2 g (SYM-1219 granules; N=125) or placebo (N=64) Study Inclusion Criteria • Adult females or postmenarchal adolescent girls ≥ 12 years of age • Clinical diagnosis of BV based on 4 Amsel criteria for BV (discharge; p. H ≥ 4. 7; clue cells ≥ 20%; positive KOH whiff test) • Nugent scores ≥ 4 Study Exclusion Criteria • Patients who were: • Pregnant or lactating • Menstruating or menopausal • Suspected clinically of having candidiasis, Chlamydia trachomatis, Trichomonas vaginalis, Neisseria gonorrhoeae, or herpes simplex infection KOH=potassium hydroxide. Data on file. Symbiomix Therapeutics, LLC. 3

Study Outcomes Primary Endpoint Proportion of clinical outcome responders (CORs): • Normal discharge, negative KOH whiff test, and clue cells <20% at TOC/EOS visit (study days 21– 30) An alternate definition of responder defined as: Secondary Endpoints • Normal discharge after treatment or abnormal discharge that is inconsistent with BV; negative KOH whiff test; clue cells <20% assessed at the interim visit (study days 7– 14) and TOC/EOS (study days 21– 30) Safety Evaluations • Rates of adverse events (AEs), serious AEs, vital signs, physical examination findings, and laboratory test results • Efficacy was evaluated at 7– 14 days (interim visit) and 21– 30 days (TOC/EOS visit) posttreatment • The efficacy analyses were performed on the m. ITT* population, defined as all randomized patients who met all study selection criteria • Secondary analyses were performed on the m. ITT population who received study drug, met the entry criteria, had an interim visit between days 7 and 14 and a TOC/EOS visit between days 21 and 30 EOS=end of study visit; m. ITT=modified intent to treat; TOC=test of cure visit. *All statistical comparisons used a stratified Cochran-Mantel-Haenszel test performed at a 0. 05 level of significance (2 -sided). Data on file. Symbiomix Therapeutics, LLC. 4

Patient Disposition 189 Women Enrolled (Safety Population) • STIs at baseline (n=13) • Nugent score ≥ 4 (n=12) • Other (n=1*) 164 Women Modified Intent-to-Treat (m. ITT) Population Secnidazole 2 g (N=107) Placebo (N=57) STIs=sexually transmitted infections. *Patients can have multiple reasons for exclusion. Data on file. Symbiomix Therapeutics, LLC. 5
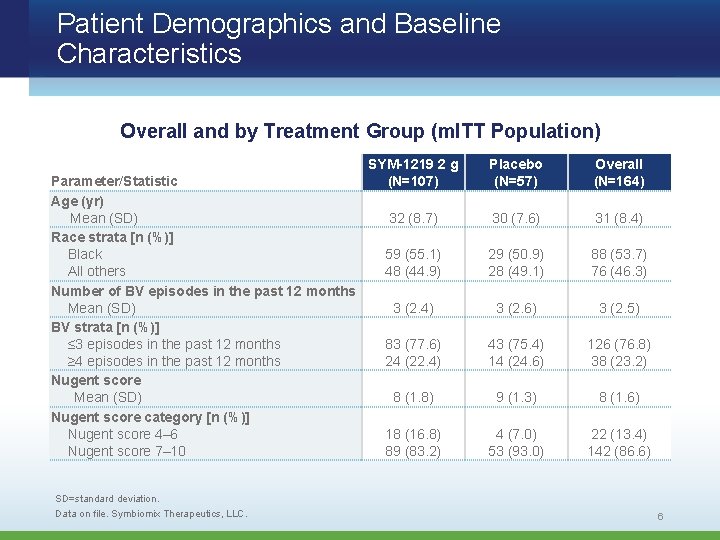
Patient Demographics and Baseline Characteristics Overall and by Treatment Group (m. ITT Population) Parameter/Statistic Age (yr) Mean (SD) Race strata [n (%)] Black All others Number of BV episodes in the past 12 months Mean (SD) BV strata [n (%)] ≤ 3 episodes in the past 12 months ≥ 4 episodes in the past 12 months Nugent score Mean (SD) Nugent score category [n (%)] Nugent score 4– 6 Nugent score 7– 10 SD=standard deviation. Data on file. Symbiomix Therapeutics, LLC. SYM-1219 2 g (N=107) Placebo (N=57) Overall (N=164) 32 (8. 7) 59 (55. 1) 48 (44. 9) 3 (2. 4) 83 (77. 6) 24 (22. 4) 30 (7. 6) 29 (50. 9) 28 (49. 1) 3 (2. 6) 43 (75. 4) 14 (24. 6) 31 (8. 4) 88 (53. 7) 76 (46. 3) 3 (2. 5) 126 (76. 8) 38 (23. 2) 8 (1. 8) 18 (16. 8) 89 (83. 2) 9 (1. 3) 4 (7. 0) 53 (93. 0) 8 (1. 6) 22 (13. 4) 142 (86. 6) 6

Study Population (m. ITT) • Stratified by race (black vs all other) – 54% black • Stratified by recurrent BV sufferers vs infrequent sufferers (defined as ≥ 4 episodes of BV in past 12 months vs ≤ 3, including current episode) – 23% recurrent BV sufferers • 87% had Nugent score ≥ 7 at baseline Data on file. Symbiomix Therapeutics, LLC. 7
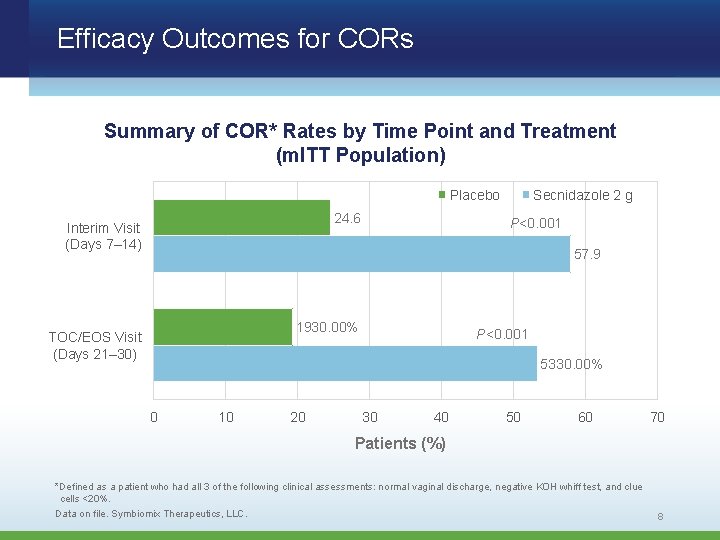
Efficacy Outcomes for CORs Summary of COR* Rates by Time Point and Treatment (m. ITT Population) Placebo 24. 6 Interim Visit (Days 7– 14) Secnidazole 2 g P<0. 001 57. 9 1930. 00% TOC/EOS Visit (Days 21– 30) P<0. 001 5330. 00% 0 10 20 30 40 50 60 70 Patients (%) *Defined as a patient who had all 3 of the following clinical assessments: normal vaginal discharge, negative KOH whiff test, and clue cells <20%. Data on file. Symbiomix Therapeutics, LLC. 8
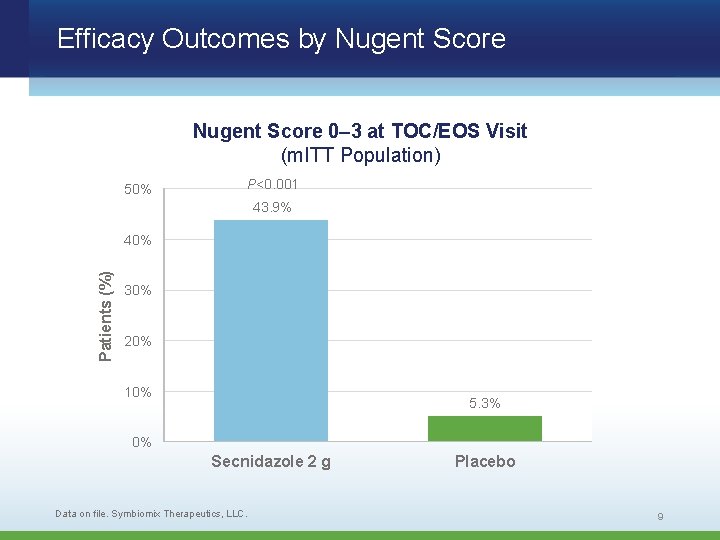
Efficacy Outcomes by Nugent Score 0– 3 at TOC/EOS Visit (m. ITT Population) 50% P<0. 001 43. 9% Patients (%) 40% 30% 20% 10% 5. 3% 0% Secnidazole 2 g Data on file. Symbiomix Therapeutics, LLC. Placebo 9
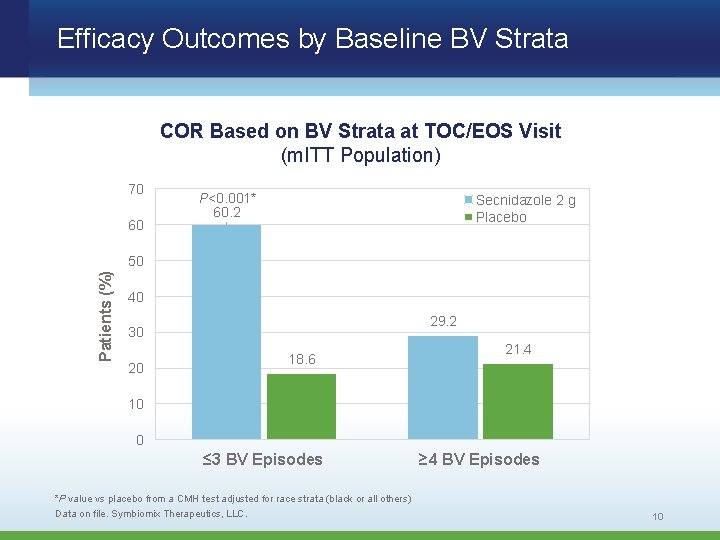
Efficacy Outcomes by Baseline BV Strata COR Based on BV Strata at TOC/EOS Visit (m. ITT Population) 70 60 P<0. 001* 60. 2 Secnidazole 2 g Placebo Patients (%) 50 40 29. 2 30 20 18. 6 21. 4 10 0 ≤ 3 BV Episodes *P value vs placebo from a CMH test adjusted for race strata (black or all others) Data on file. Symbiomix Therapeutics, LLC. ≥ 4 BV Episodes 10

Efficacy Outcomes for Secondary Analyses The 2016 FDA-defined alternate clinical cure rates utilized data from patients that had a baseline Nugent score of 7– 10 • Single-dose secnidazole 2 g (64. 0% [n=57]) was superior to placebo (26. 4% [n=14]) Investigator’s clinical assessment at the TOC/EOS visit demonstrated • Significantly more patients in the secnidazole 2 g group (68. 0% [68/107]) compared with placebo (29. 6% [16/57]) required no additional BV treatment (P<0. 001) • Clinical cure=normal discharge after treatment or abnormal discharge that is inconsistent with BV, a normal KOH whiff test, and clue cells <20% by day 7– 14 visit FDA=US Food and Drug Administration. Data on file. Symbiomix Therapeutics, LLC. 11
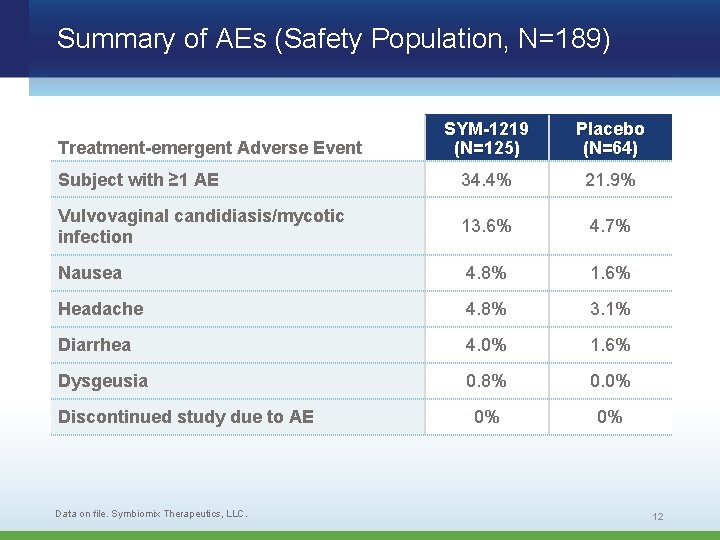
Summary of AEs (Safety Population, N=189) SYM-1219 (N=125) Placebo (N=64) Subject with ≥ 1 AE 34. 4% 21. 9% Vulvovaginal candidiasis/mycotic infection 13. 6% 4. 7% Nausea 4. 8% 1. 6% Headache 4. 8% 3. 1% Diarrhea 4. 0% 1. 6% Dysgeusia 0. 8% 0. 0% 0% 0% Treatment-emergent Adverse Event Discontinued study due to AE Data on file. Symbiomix Therapeutics, LLC. 12

Conclusion The 2 g dose of SYM-1219 granules was statistically superior to placebo on primary and secondary endpoints Statistical superiority was demonstrated for the single-dose secnidazole 2 g group in both race strata for all efficacy outcomes Secnidazole 2 g was well tolerated Data on file. Symbiomix Therapeutics, LLC. 13
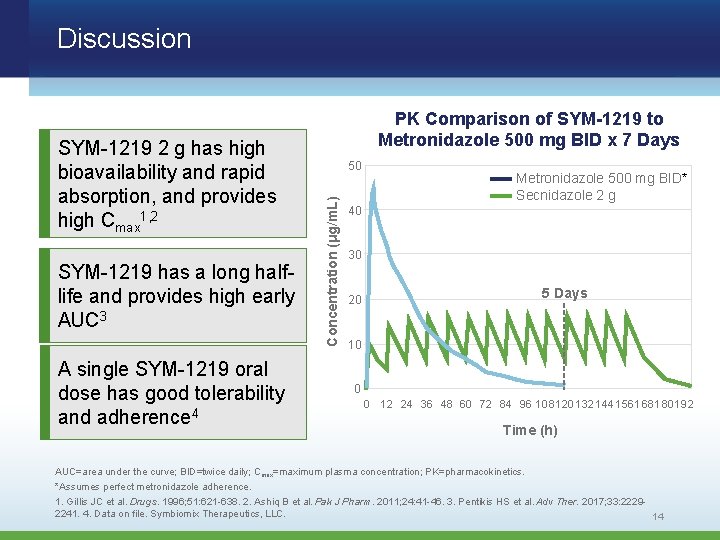
Discussion SYM-1219 has a long halflife and provides high early AUC 3 A single SYM-1219 oral dose has good tolerability and adherence 4 50 Concentration (μg/m. L) SYM-1219 2 g has high bioavailability and rapid absorption, and provides high Cmax 1, 2 PK Comparison of SYM-1219 to Metronidazole 500 mg BID x 7 Days Metronidazole 500 mg BID* Secnidazole 2 g 40 30 5 Days 20 10 0 0 12 24 36 48 60 72 84 96 108 120 132 144 156 168 180 192 Time (h) AUC=area under the curve; BID=twice daily; Cmax=maximum plasma concentration; PK=pharmacokinetics. *Assumes perfect metronidazole adherence. 1. Gillis JC et al. Drugs. 1996; 51: 621 -638. 2. Ashiq B et al. Pak J Pharm. 2011; 24: 41 -46. 3. Pentikis HS et al. Adv Ther. 2017; 33: 22292241. 4. Data on file. Symbiomix Therapeutics, LLC. 14

Funding and Conflict of Interest Funding for this study was provided to the University of Alabama (Schwebke), Tidewater Physicians for Women (Morgan), Medical Center for Clinical Research (Koltun), and Drexel University School of Medicine (Nyirjesy) by Symbiomix Therapeutics, LLC, Baltimore, MD 15
- Slides: 15