A PBMC Control PBMC Ptptide Tumor Volume mm
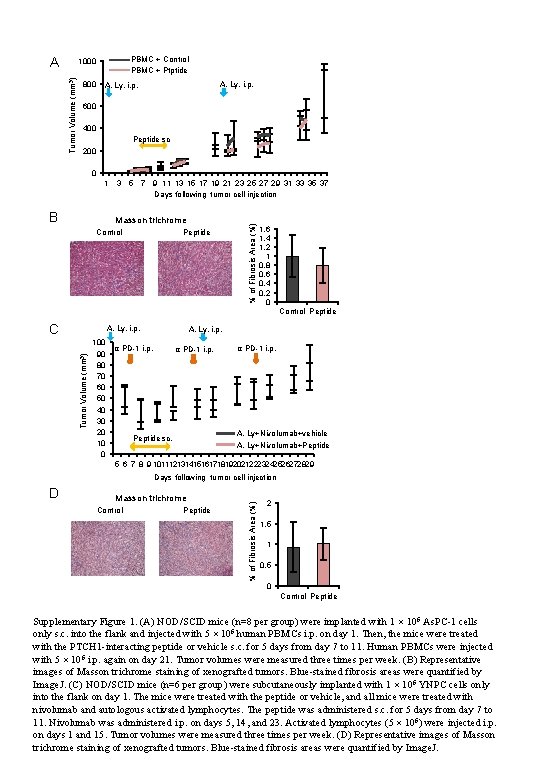
A PBMC + Control PBMC + Ptptide Tumor Volume (mm 3) 1000 800 A. Ly. i. p. 600 400 Peptide sc 200 0 B Masson trichrome Control Peptide % of Fibrosis Area (%) 1 3 5 7 9 11 13 15 17 19 21 23 25 27 29 31 33 35 37 Days following tumor cell injection 1, 6 1, 4 1, 2 1 0, 8 0, 6 0, 4 0, 2 0 Control Peptide C Tumor Volume (mm 3) A. Ly. i. p. 100 90 80 70 60 50 40 30 20 10 0 A. Ly. i. p. α PD-1 i. p. A. Ly+Nivolumab+vehicle A. Ly+Nivolumab+Peptide sc. 5 6 7 8 9 1011121314151617181920212223242526272829 D Masson trichrome Control Peptide % of Fibrosis Area (%) Days following tumor cell injection 2 1, 5 1 0, 5 0 Control Peptide Supplementary Figure 1. (A) NOD/SCID mice (n=8 per group) were implanted with 1 × 106 As. PC-1 cells only s. c. into the flank and injected with 5 × 106 human PBMCs i. p. on day 1. Then, the mice were treated with the PTCH 1 -interacting peptide or vehicle s. c. for 5 days from day 7 to 11. Human PBMCs were injected with 5 × 106 i. p. again on day 21. Tumor volumes were measured three times per week. (B) Representative images of Masson trichrome staining of xenografted tumors. Blue-stained fibrosis areas were quantified by Image. J. (C) NOD/SCID mice (n=6 per group) were subcutaneously implanted with 1 × 106 YNPC cells only into the flank on day 1. The mice were treated with the peptide or vehicle, and all mice were treated with nivolumab and autologous activated lymphocytes. The peptide was administered s. c. for 5 days from day 7 to 11. Nivolumab was administered i. p. on days 5, 14, and 23. Activated lymphocytes (5 × 106) were injected i. p. on days 1 and 15. Tumor volumes were measured three times per week. (D) Representative images of Masson trichrome staining of xenografted tumors. Blue-stained fibrosis areas were quantified by Image. J.
- Slides: 1