A Multicenter Randomized Clinical Trial to evaluate the





















- Slides: 32

A Multicenter Randomized Clinical Trial to evaluate the Efficiency of small vessel coronary treatment using Paclitaxel drug-coated balloon vs. Zotarolimus-eluting stent: RAMSES trial Victor Alfonso Jimenez Diaz, MD, MPH Cardiology department, Hospital Alvaro Cunqueiro, University Hospital of Vigo, Spain On behalf of RAMSES study investigators

Victor A. Jimenez Diaz, MD, MPH I have no relevant financial relationships

BACKGROUND • Drug-coated balloons (DCB): value in different settings of CAD – Immediate and homogenous drug uptake – Avoidance of inflammatory reaction to stent struts or polymers – Respecting the normal vessel anatomy • Small coronary vessels: complex scenario – – • • Secondary branches / distal lesions Tortuosity No dedicated devices Prognostic / clinical benefit DES in small vessels are associated with higher rates of restenosis than in larger vessels Definition varies across studies: <3 mm / 2, 25 - 2, 75 mm / 2 - 2, 8 mm

BACKGROUND DCB in small vessels: Studies Late Lumen Loss at 6 month-FU BELLO trial 0. 08 ± 0. 38 mm PEPCAD I trial 0. 18 ± 0. 38 mm ü Vessel thrombosis: 1% (6 -9 months) VALENTINES II trial 0. 38 ± 0. 39 mm ü Bailout: 7. 5 to 26% DIOR Spanish reg. 0. 34 ± 0. 23 mm ü MACE: 6 -10% ü TLR: 3 -5% (6 -8 m) ü Late Loss: 0. 02 -0. 34 mm • Comparisons between studies are difficult and, as well, the applicability to standard practice o Different clinical populations ○ Different devices used o Different endpoints and timepoint of measurements ○ Non-homogeneous techniques • The less complexity, the greater the benefit • In vessels of <2. 25 mm we tend to underestimate their clinical relevance • Severe lesions in small vessels can be very symptomatic ! BELLO trial. Latib A, Colombo A. JACC 2012 PICOLETTO trial. Cortese B, et al. Heart 2010

METHODS RAMSES trial: Paclitaxel DCB vs. Zotarolimus DES in small coronary vessels • • Prospective, multicenter, randomized controlled trial Patients with de novo coronary lesions located in small vessels – Reference vessel diameter: 2. 00 to 2. 75 mm • Randomized to: IN. PACT Falcon-DCB – Paclitaxel-DCB (IN. PACT Falcon, Medtronic Inc); or – Zotarolimus-DES (Resolute Integrity, Medtronic Inc) • Primary endpoint: – Composite of major adverse cardiac events ([MACEs] - cardiac death, MI, CABG, and clinically-driven TVR or CD-TLR at 12 month follow-up Resolute Integrity-DES

METHODS RAMSES trial: Paclitaxel DCB vs. Zotarolimus DES in small coronary vessels • Investigator Initiated Study • Executive and Clinical Events Committee (5 members) • All patients/events monitorized by an independent CRO • 7 centers in Spain: – H. Álvaro Cunqueiro, Vigo: Andrés Íñiguez – H. Universitario Ciudad Real: Fernando Lozano – H. Universitario de Canarias: Francisco Bosa – H. Carlos Haya: José Urbano – H. Universitario León: Felipe Fernández – H. Virgen de la Victoria: José María Hernández – H. Virgen de las Nieves: Rafael Melgares IN. PACT Falcon-DCB Resolute Integrity-DES

METHODS RAMSES trial: Paclitaxel DCB vs. Zotarolimus DES in small coronary vessels • Inclusion criteria • Exclusion criteria • CAD with de novo lesions in native coronary • Left main disease, CTOs, Bifurcations arteries • Severely calcified lesions, ISR Stenosis severity of ≥ 70% by visual stimation or • Lesions in LIMA, RIMA or venous bypasess >50% stenosis by QCA • AMI in the 48 hr pre-PCI Coronary vessels diameter between 2, 00 and • Severe renal failure 2, 75 mm • Allergy or contraindications to ASA or P 2 Y 12 inhibitors • Allergy to contrast agents • Life expectancy <1 year • • • Lesion length of ≤ 25 mm

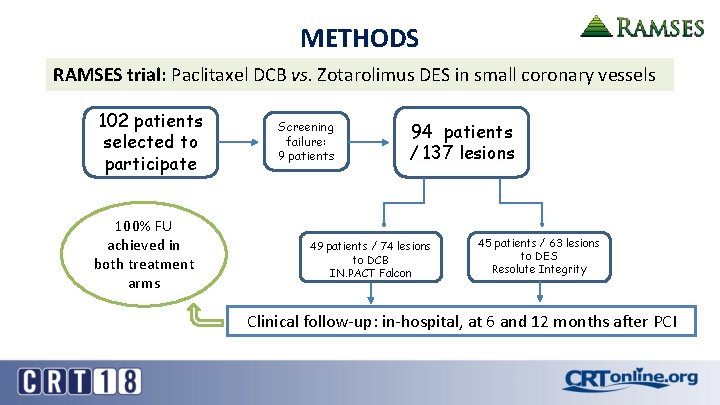
METHODS RAMSES trial: Paclitaxel DCB vs. Zotarolimus DES in small coronary vessels 102 patients selected to participate 100% FU achieved in both treatment arms Screening failure: 9 patients 94 patients / 137 lesions 49 patients / 74 lesions to DCB IN. PACT Falcon 45 patients / 63 lesions to DES Resolute Integrity Clinical follow-up: in-hospital, at 6 and 12 months after PCI

METHODS RAMSES trial: Paclitaxel DCB vs. Zotarolimus DES in small coronary vessels Clinically and anatomically elegible patient Predilation with a balloon diameter 0, 25 mm less than the RVD and less than total lesion length No vessel occlusion or ≥ type C dissection Randomization DCB: balloon diameter 0. 8 -1. 0 to the RVD, and at least 5 mm length longer than lesion length, with inflation duration for 60 seg, increasing atmosferes progresively until reach nominal balloon size DES: stent diameter 1: 1 to RVD, and at least 5 mm length longer than lesion length

METHODS RAMSES trial: Paclitaxel DCB vs. Zotarolimus DES in small coronary vessels In-PACT Falcon-DCB Zotarolimus-DES N: 49 pts /74 lesions N: 45 pts /63 lesions Angiographic success: residual stenosis <30% in the treated segment and abscense of ≥ type C dissection No angiographic success: BMS implantation In-hospital clinical follow-up 6 -month and 1 -year clinical follow-up

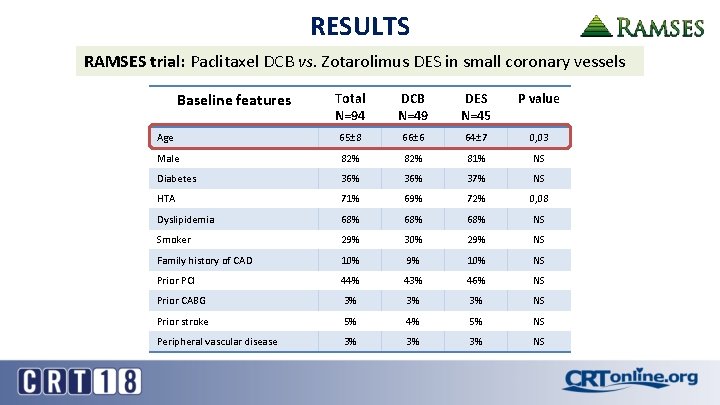
RESULTS RAMSES trial: Paclitaxel DCB vs. Zotarolimus DES in small coronary vessels Baseline features Total N=94 DCB N=49 DES N=45 P value Age 65± 8 66± 6 64± 7 0, 03 Male 82% 81% NS Diabetes 36% 37% NS HTA 71% 69% 72% 0, 08 Dyslipidemia 68% 68% NS Smoker 29% 30% 29% NS Family history of CAD 10% 9% 10% NS Prior PCI 44% 43% 46% NS Prior CABG 3% 3% 3% NS Prior stroke 5% 4% 5% NS Peripheral vascular disease 3% 3% 3% NS

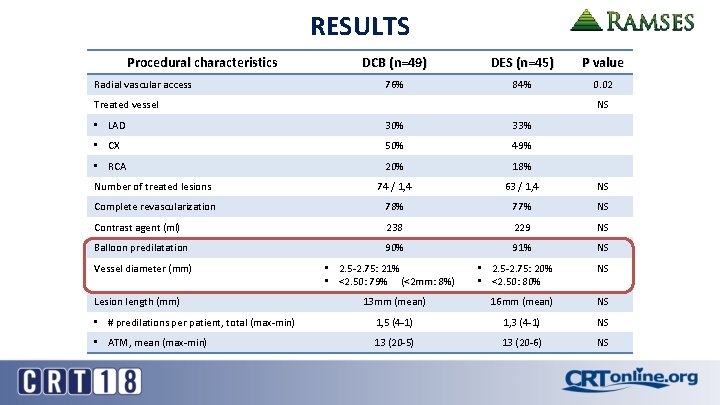
RESULTS Procedural characteristics Radial vascular access DCB (n=49) DES (n=45) P value 76% 84% 0. 02 Treated vessel NS • LAD 30% 33% • CX 50% 49% • RCA 20% 18% Number of treated lesions 74 / 1, 4 63 / 1, 4 NS Complete revascularization 78% 77% NS Contrast agent (ml) 238 229 NS Balloon predilatation 90% 91% NS Vessel diameter (mm) • 2. 5 -2. 75: 20% • <2. 50: 80% NS 13 mm (mean) 16 mm (mean) NS • # predilations per patient, total (max-min) 1, 5 (4 -1) 1, 3 (4 -1) NS • ATM, mean (max-min) 13 (20 -5) 13 (20 -6) NS Lesion length (mm) • 2. 5 -2. 75: 21% • <2. 50: 79% (<2 mm: 8%)

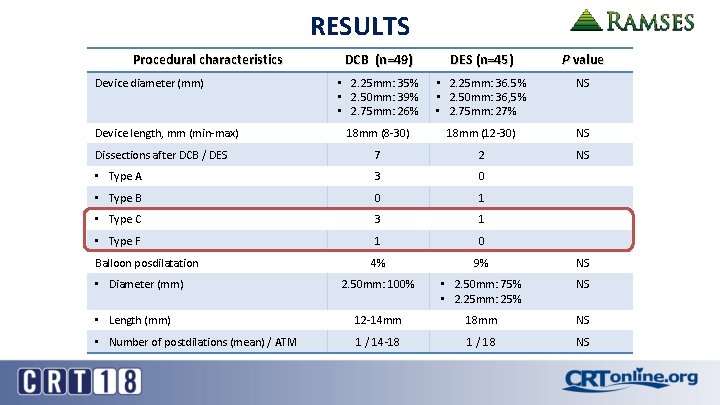
RESULTS Procedural characteristics DCB (n=49) DES (n=45) P value • 2. 25 mm: 35% • 2. 50 mm: 39% • 2. 75 mm: 26% • 2. 25 mm: 36. 5% • 2. 50 mm: 36, 5% • 2. 75 mm: 27% NS 18 mm (8 -30) 18 mm (12 -30) NS Dissections after DCB / DES 7 2 NS • Type A 3 0 • Type B 0 1 • Type C 3 1 • Type F 1 0 4% 9% NS 2. 50 mm: 100% • 2. 50 mm: 75% • 2. 25 mm: 25% NS • Length (mm) 12 -14 mm 18 mm NS • Number of postdilations (mean) / ATM 1 / 14 -18 1 / 18 NS Device diameter (mm) Device length, mm (min-max) Balloon posdilatation • Diameter (mm)

RESULTS RAMSES trial: Paclitaxel DCB vs. Zotarolimus DES in small coronary vessels v All 4 crossovers were from the DCB arm and the strategy was BMS implantation DCB N: 49 DES N: 45 ü There were 2 dissections in the DES arm (1 type B and Crossover 1 type C) sealed with assigned DES ü There were 7 dissections in the DEB arm: 3 type C and 1 type F (bailout) sealed with BMS (2 of 2. 25 and 2 of 2. 5 mm), and 3 type A dissections under conservative treatment 0 pts DCB N: 45 4 pts (8, 1%) DES N: 49

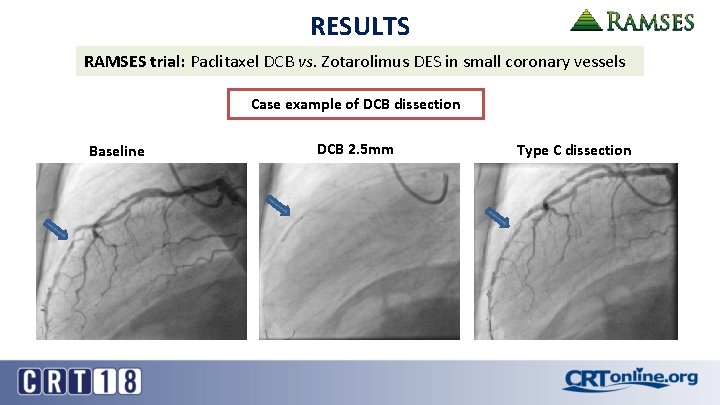
RESULTS RAMSES trial: Paclitaxel DCB vs. Zotarolimus DES in small coronary vessels Case example of DCB dissection Baseline DCB 2. 5 mm Type C dissection

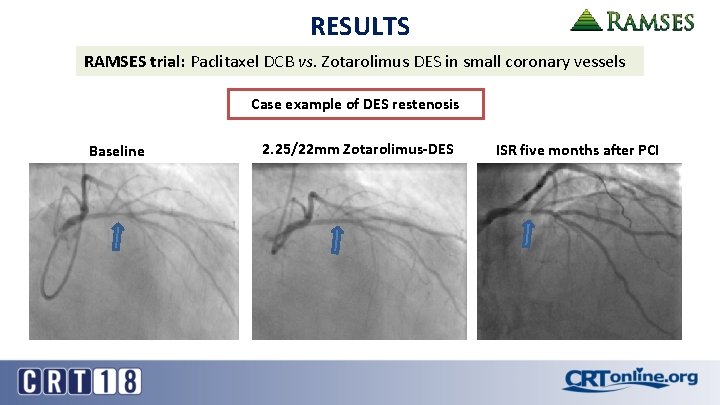
RESULTS RAMSES trial: Paclitaxel DCB vs. Zotarolimus DES in small coronary vessels Case example of DES restenosis Baseline 2. 25/22 mm Zotarolimus-DES ISR five months after PCI

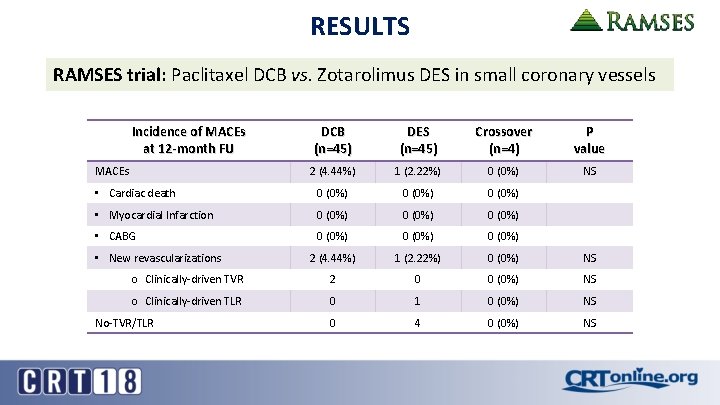
RESULTS RAMSES trial: Paclitaxel DCB vs. Zotarolimus DES in small coronary vessels Incidence of MACEs at 12 -month FU DCB (n=45) DES (n=45) Crossover (n=4) P value 2 (4. 44%) 1 (2. 22%) 0 (0%) NS • Cardiac death 0 (0%) • Myocardial Infarction 0 (0%) • CABG 0 (0%) 2 (4. 44%) 1 (2. 22%) 0 (0%) NS o Clinically-driven TVR 2 0 0 (0%) NS o Clinically-driven TLR 0 1 0 (0%) NS 0 4 0 (0%) NS MACEs • New revascularizations No-TVR/TLR

CONCLUSIONS 1) The use of Paclitaxel-DCB for the treatment of de novo lesions in small coronary vessels seems to be as safe and effective as 2 nd gen. Zotarolimus-DES o Similar procedural success rates o No significant differences in periprocedural complications and MACEs at 12 -month FU o Low crossover rate in the DCB group 2) Incidence of MACEs is very low at 12 -months FU, in both groups 3) Larger population of patients and longer FU period is desirable

PERSONAL TAKE-HOME MESSAGES • Reassuring and encouraging data • With the use of DCB in de novo lesions, predictable results are possible: – Adequate patient / lesion selection – Optimal PCI technique – Careful balloon predilation looking for residual stenosis <30% • In light of the disappointing performance of current BRS in small vessels and similar results with current DES generation, DCB may play an important role in this setting • The aim of “PCI without leaving any metal behind“ seems to be feasible and safe with DCB

THANK YOU FOR YOUR ATTENTION Hospital Alvaro Cunqueiro University Hospital of Vigo, Spain

BACKUP SLIDES

Methods COST ESTIMATION (included): Ø Direct hospitalization costs Ø Costs of procedures Ø Follow-up costs / patient Ø Costs of complications Ref. DOG num. 96, miércoles 21 de mayo, 2014

Definitions Average C/E: Cost per patient with initial procedure success, absence of cross-over and without MACE during follow-up.

Definitions Effectiveness: An initial successful revascularization procedure without need of cross-over and absence of cardiac death, myocardial infarction, or need for new revascularization on the treated lesion (TLF) at 1 -year follow-up

Definitions Incremental C/E: Extra-cost of one of each strategy in comparison to the alternative per each extra-unit of benefit achieved. Cost Treatment A - Cost Treatment B C/E i = ---------------------------Effectiveness Treat. A – Effectiveness Treat. B

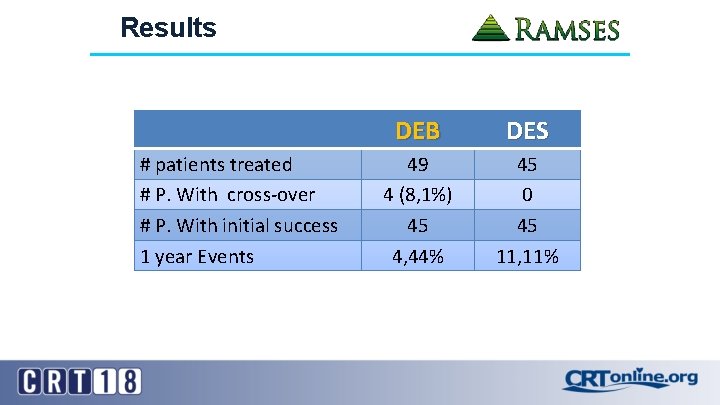
Results # patients treated # P. With cross-over # P. With initial success 1 year Events DEB DES 49 4 (8, 1%) 45 4, 44% 45 0 45 11, 11%

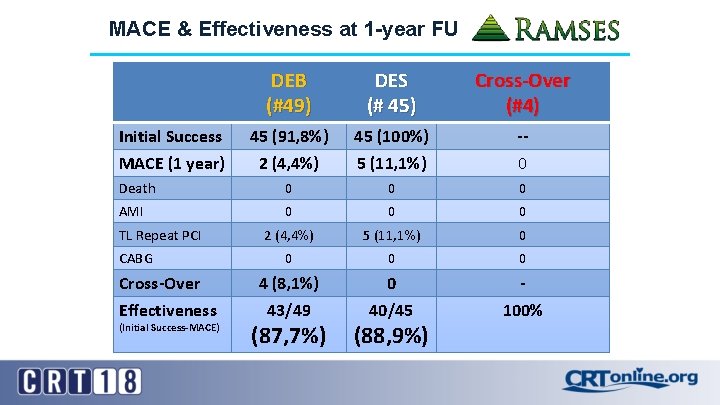
MACE & Effectiveness at 1 -year FU DEB (#49) DES (# 45) Cross-Over (#4) 45 (91, 8%) 2 (4, 4%) 45 (100%) 5 (11, 1%) -0 Death 0 0 0 AMI 0 0 0 2 (4, 4%) 5 (11, 1%) 0 0 4 (8, 1%) 43/49 0 40/45 100% Initial Success MACE (1 year) TL Repeat PCI CABG Cross-Over Effectiveness (Initial Success-MACE) (87, 7%) (88, 9%)

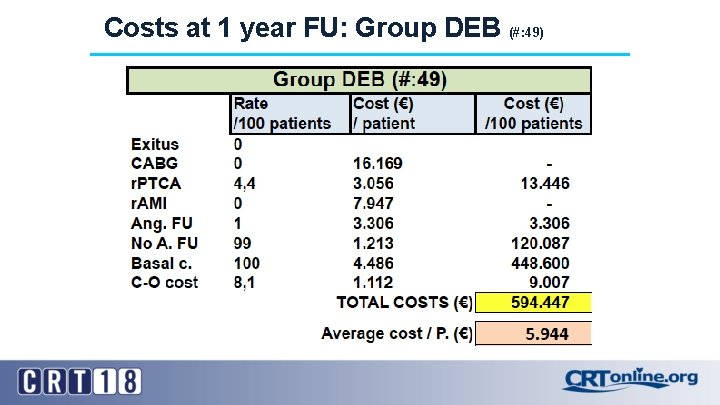
Costs at 1 year FU: Group DEB (#: 49)

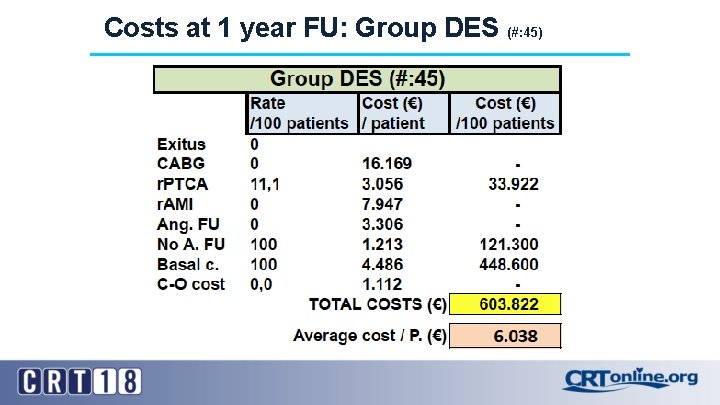
Costs at 1 year FU: Group DES (#: 45)

Results: C/E at 1 year FU (€) (%) (€)

Methods Direct Cost *: ¨ Basal Costs: 4. 486, 00 € ¨ Coronary angiogram 721, 45 € ¨ PTCA 3. 088, 54 € ¨ CABG 15. 330, 97 € ¨ AMI 7. 947, 19 € ¨ Follow-up of Patient without angina 1. 213, 10 € ¨ Hospital admission of patient with angina 3. 306, 67 € *: excluded salaries.

Methods Basal Cost / unit : ¨ Hospitalization ¨ EKG ¨ RX ¨ CK ¨ Blood counter ¨ Biochemistry ¨ Echocardiogram ¨ Treadmil test (CCU: 358, 92 €, Gen. : 147, 41 €), 24, 89 € 37, 33 € 58, 33 € 14, 93 € 49, 78 € 84, 63 € 126, 25 €