A Matter Flowchart MATTER no Can it be
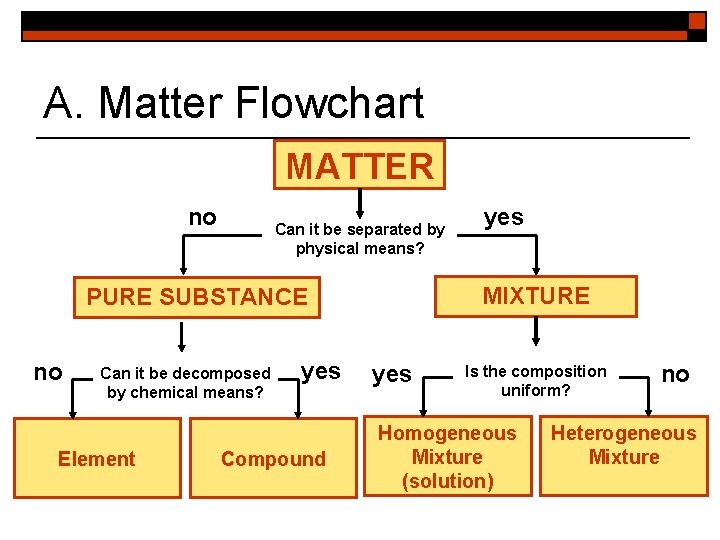
A. Matter Flowchart MATTER no Can it be separated by physical means? MIXTURE PURE SUBSTANCE no Can it be decomposed by chemical means? Element yes Compound yes Is the composition uniform? Homogeneous Mixture (solution) no Heterogeneous Mixture

B. Pure Substances o Element n n matter composed of identical atoms EX: copper

B. Pure Substances o Compound n n n matter composed of 2 or more elements in a fixed ratio properties differ from those of individual elements EX: salt (Na. Cl)

C. Mixtures Non-specific combination of 2 or more pure substances. Two Types of Mixtures Homogeneous Mixture Heterogeneous Mixture even distribution of components uneven distribution of components EX: saline solution EX: granite

A. Physical Property o A characteristic of matter that can be observed or measured without changing the identity of its matter or having to react it with another substance. n can be used to separate mixtures and identify substances n EX: color, state of matter, density, odor, ductility, malleability, conductivity, luster, magnetic, mass & volume

Density o Solids, liquids and gases are matter, so they all have density.

Determining Density o To find the density of a material, you need to know the mass and volume of a solid sample of the material. 1. 2. Mass is measured with a balance or scale. Use the displacement method or calculate the volume.

B. Physical Change o o o Physical changes occur when matter changes its property but not its chemical nature. Physical changes could include a change in: texture, shape, size, color, odor, volume, mass, state Ex. Boil, evaporate, dissolve, break, split, crack, grind, cut, crush, and bend.

Physical Change Remember! n properties remain the same n can be used to separate mixtures

Physical Change

C. Chemical Property o Describes how a material reacts with another substance. n EX: flammability, reactivity

D. Chemical Change n During a chemical change, properties change and new substances are formed. n Signs: color change, formation of a gas/solid, release of light/heat n Ex. Oxidation – tarnishing/rusting exploding, burning, fermenting, digesting, rotting

Chemical Change o A chemical change occurs when fireworks are used. Fireworks are made of metals such as magnesium and copper. These change chemically as they light up the sky.

Physical & Chemical Changes Game Che mic al

Physical & Chemical Changes Game y h P l a sic

Physical & Chemical Changes Game Che mic al

Physical & Chemical Changes Game y h P l a sic

Physical & Chemical Changes Game y h P l a sic

Physical & Chemical Changes Game Che mic al

Physical & Chemical Changes Game y h P l a sic

Physical & Chemical Changes Card Game y h P l a sic

Physical & Chemical Changes Game Che mic al

Physical & Chemical Changes Game y h P l a sic

Physical & Chemical Changes Game y h P l a sic

Physical & Chemical Changes Game Che mic al

Physical & Chemical Changes Game Che mic al

Physical & Chemical Changes Game y h P l a sic
Physical & Chemical Changes Game Che mic al

Physical & Chemical Changes Game y h P l a sic
- Slides: 29