A Level Physics Nuclear Physics Particle Accelerators Objectives

A –Level Physics: Nuclear Physics Particle Accelerators

Objectives:

FLASHBACK: Explain the meaning of constructive and destructive interference (6 marks)

Starter Activity Write rough notes on the video. Anything that is new information to you should be recorded. We will watch the documentary and then start filling in information https: //www. youtube. com/watch? v=MVW 247 LWDk

Types of Particle Accelerators Particle accelerators are machines that speed up particles using a combination of -400 BC electric and magnetic fields. There are two main types of particle accelerator, a linear particle accelerator and a cyclotron 1) Linear Particle Accelerator One of the easiest ways to accelerate a charged particle is to do so in a straight path. The problem with this is that this is limited to the maximum potential difference of the electrodes used to accelerate.
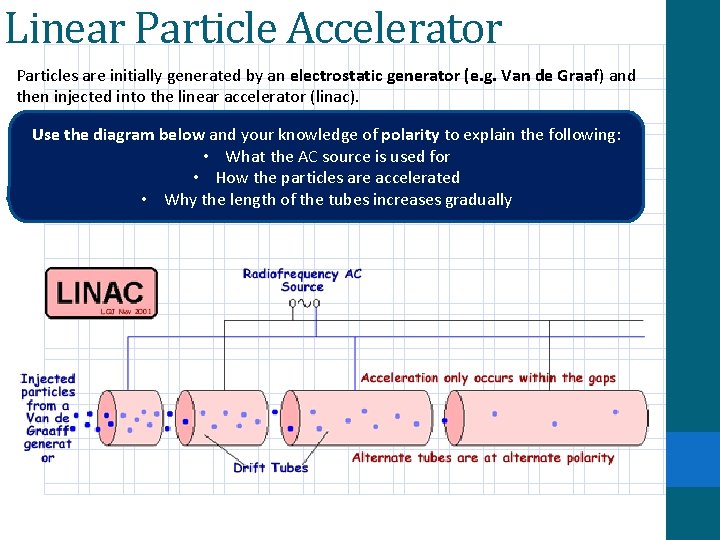
Linear Particle Accelerator Particles are initially generated by an electrostatic generator (e. g. Van de Graaf) and then injected into the linear accelerator (linac). -400 BC Use the diagram below and your knowledge of polarity to explain the following: • What the AC source is used for • How the particles are accelerated • Why the length of the tubes increases gradually

Cyclotrons -400 BC Centripetal Centre As Fleming’s left hand rule shows, the force on a moving charge in a magnetic field is perpendicular to its direction of travel. So, as it travels through and it gets pushed, it is put in the conditions (mathematically) for circular motion. The magnetic force acts a centripetal force Equate the equation for the magnetic force on an individual particle with centripetal force. Leave the combined formula with ‘r’ as the subject
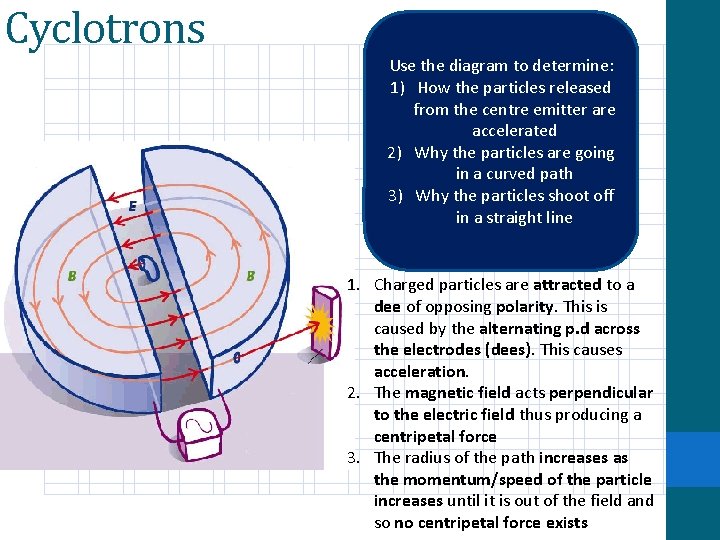
Cyclotrons Use the diagram to determine: 1) How the particles released from the centre emitter are accelerated 2) Why the particles are going in a curved path 3) Why the particles shoot off in a straight line 1. Charged particles are attracted to a dee of opposing polarity. This is caused by the alternating p. d across the electrodes (dees). This causes acceleration. 2. The magnetic field acts perpendicular to the electric field thus producing a centripetal force 3. The radius of the path increases as the momentum/speed of the particle increases until it is out of the field and so no centripetal force exists

Electronvolts Amazing how many times this comes up? The kinetic energy that a charged particle gains when it is accelerated through a potential difference of V is given by the equation Ek=QV. As you know, that KE= ½ mv 2 and E=QV……then ½ mv 2= QV And if the ‘q’ is replaced with ‘e’ as it could be for a single electron or proton, we get… ½ mv 2= e. V So this makes sense as the KE gained by the particle is equivalent to the energy provided to it by the electric field. Remember that 1 e. V= 1. 6 x 10 -19 J

Cyclotron and Synchrotron Use pages 96 and 97 to answer the following: • How is the timing of the acceleration in a cyclotron maintained? • Use formulae to prove that the frequency of the cyclotron dees is independent of the radius • Include the relativistic effects of mass in the formula • What is a synchrotron? • Explain a situation whereby scientists were caught out for cheating
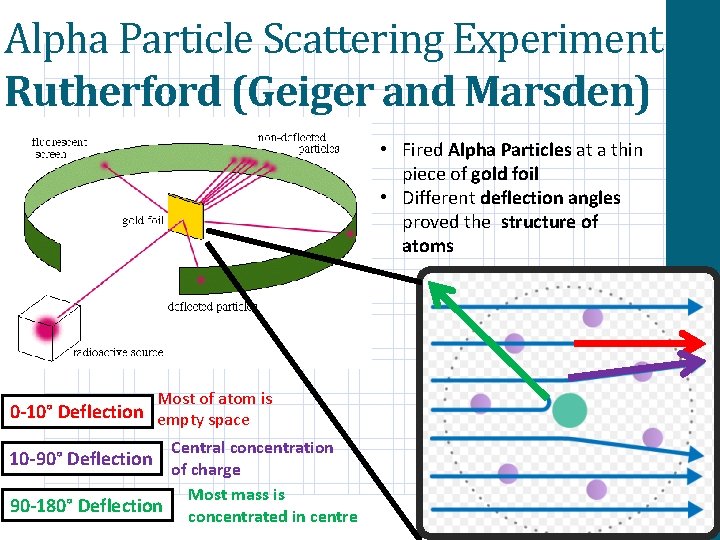
Alpha Particle Scattering Experiment Rutherford (Geiger and Marsden) • Fired Alpha Particles at a thin piece of gold foil • Different deflection angles proved the structure of atoms Most of atom is 0 -10° Deflection empty space Central concentration of charge Most mass is 90 -180° Deflection concentrated in centre 10 -90° Deflection

Chadwick’s Discovery of the Neutron The alpha particle experiment proved the existence of a centralised ball of mass and charge. However, this experiment did not explain why the mass number was so much greater than the proton number. In 1930, Irene Joliot-Curie and her husband found that alpha particles striking berryllium would cause it to give off unknown radiation. This radiation could then knock off protons out of paraffin. What does this observation tell us about this unknown radiation? They thought that the radiation could be gamma. Why could this not be the case? What law does this breach? By considering the conservation of momentum and kinetic energy in the collisions. They concluded that the radiation was a neutral particle with a mass 1% greater than a proton. A neutron

Nuclear Structure MINI quiz Label the Atomic Mass Label the Atomic/Proton Number Explain what an Isotope is Which quantity is labelled ‘Z’ and which is labelled ‘A’? • Explain why the atomic mass of chlorine is 35. 5 • •

Exam Question Discuss the experimental findings that lead to the modern understanding of nuclear structure (8 marks)

Exam Question Discuss the experimental findings that lead to the modern understanding of nuclear structure (6 marks) MARK SCHEME: • Dalton determined atoms can be distinguished by their mass • Reference to (Thompson’s) plum pudding model • Alpha Particle Scattering Experiment aimed alpha particles at gold nuclei/thin gold foil • Different deflection angles AND an explanation of the result (proved a centralised mass and charge) • Chadwick’s discovery of the neutron AND explanation of alpha particles striking berryllium

Electron Beams (Cathode Ray) Remember electrons can be accelerated. We often have to look at the acceleration by an electric field, just like in a particle accelerator! 2 Energy= ½ mv = e. V ½ mv 2 = e. V How fast would an electron move if accelerated from rest through a p. d. of 1200 V? NB: mass of electron= 9. 11 x 10^7 ms-1

Electron Probes Electron beams can be fired at crystal structures producing a scattering pattern which tells us about the structure of the crystal. Davisson and Germer proved that unlike the alpha particle scattering experiment, a beam through a crystal forms a diffraction pattern. This was experimental evidence to support De Broglie law as the beam (of particles) was behaving as a wave! The experiment proved that this diffraction pattern only gets produced when the beam’s electrons have a wavelength coinciding with the De. Broglie wavelength (wavelength=h/p)
- Slides: 17