A Kinetic Study of an Iodine Clock Reaction











- Slides: 13

A Kinetic Study of an Iodine Clock Reaction

A Kinetic Study of an Iodine Clock Reaction S 2 O 82 - + 3 I- 2 SO 42 - + I 3 n n (1) determine the rate law (2) determine the numerical value of the rate constant at room temperature (3) examine impact of temperature on the reaction & determine the activation energy (Ea) (4) investigate catalytic activity of selected metal ions on the reaction

Method of Initial Rates n n n Measure the rate of reaction near the start of the reaction time Alter concentration(s) of reactants and examine the impact on the initial rate Allows one to determine the exponents in the rate law m rate of reaction = k [S 2 O 82 -] [I-] n

Measuring the rate S 2 O 82 - + 3 I- 2 SO 42 - + I 3 n n Must monitor consumption of reactant and / or production of product Can detect I 3 - with starch indicator I 3 - + starch blue-black starch complex n n Problem = color appears “instantly” Can delay detection for known amount of reaction

The trick = a molecular reaction “clock”. . . n Delay the appearance of the blue-black color for a defined AMOUNT of reaction. fast n I 2 + I - I 3 - Consume I 2 BEFORE it can become I 3 -. very fast I 2 + 2 S 2 O 32 - S 4 O 62 - + 2 In Use constant amount of delaying reagent (S 2 O 32 -) every trial

A proposed mechanism

Finding the rate of the reaction S 2 O 82 - + 3 I- 2 SO 42 - + I 3 - n But we are “timing” the reaction by adding the thiosulfate (S 2 O 32 -) and delaying the appearance of I 3 rate of reaction = rate of consumption of I = 2

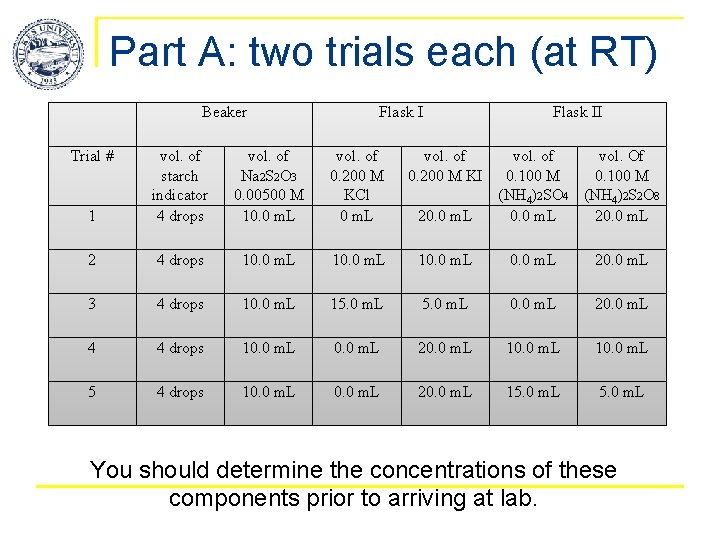
Part A: two trials each (at RT) Beaker Trial # Flask II vol. of Na 2 S 2 O 3 0. 00500 M 10. 0 m. L vol. of 0. 200 M KCl 0 m. L vol. of 0. 200 M KI 1 vol. of starch indicator 4 drops 2 4 drops 10. 0 m. L 20. 0 m. L 3 4 drops 10. 0 m. L 15. 0 m. L 0. 0 m. L 20. 0 m. L 4 4 drops 10. 0 m. L 20. 0 m. L 10. 0 m. L 5 4 drops 10. 0 m. L 20. 0 m. L 15. 0 m. L 20. 0 m. L vol. of vol. Of 0. 100 M (NH 4)2 SO 4 (NH 4)2 S 2 O 8 0. 0 m. L 20. 0 m. L You should determine the concentrations of these components prior to arriving at lab.

Finding the Rate Law Exponents – a graphical m n approach rate of reaction = k [S 2 O 82 -] [I-] can also be rewritten in a logarithmic form to get an equation for a straight line: log(rate) = log(k) + m log[S 2 O 82 -] + n log[I-] Cleverly keeping one concentration constant while varying the other allows us to determine “m” and “n”. . . and ultimately the value of k.

Part B – Temperature Variation n Repeat Trial 1 composition from Part A q q n Two separate trials done at a temperature close to 0°C Two separate trials done at a temperature near 50°C This data will be combined with the room temperature data to determine the activation energy (Ea) and the pre-exponential constant (A, the “collisional” constant)

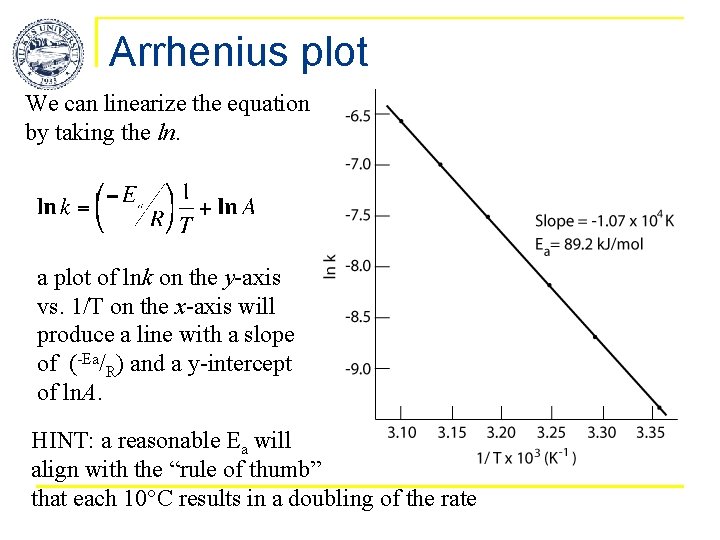
Arrhenius plot We can linearize the equation by taking the ln. a plot of lnk on the y-axis vs. 1/T on the x-axis will produce a line with a slope of (-Ea/R) and a y-intercept of ln. A. HINT: a reasonable Ea will align with the “rule of thumb” that each 10°C results in a doubling of the rate

Part C – Catalytic Ion Activity n Repeat Trial 1 composition from Part A q q Add metal ion as possible catalyst One trial each. . . If time or reagents are in short supply, work may be divided across the lab and results “pooled” For the best catalyst (shortest reaction time); repeat once again with the metal ion solution added along with five drops of an EDTA solution EDTA is known to effectively bind 2+ metal ions: it is used in food preservation

TIPS for the lab and report n Pay attention to chemical labels q n n n thiosulfate and persulfate are not the same and neither are potassium iodide / potassium chloride, etc. Calculate your rates IN LAB Graph your work ASAP (i. e. not the night before or day your report is due) Allow time for the report to develop as you attempt to understand the experiment