A Hydrocarbon Typical petroleum product Nonpolar CH 2
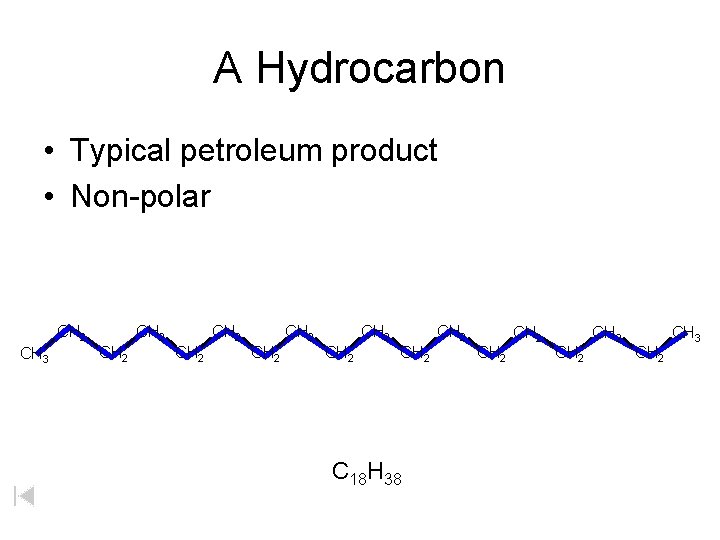
A Hydrocarbon • Typical petroleum product • Non-polar CH 2 CH 3 CH 2 CH 2 CH 2 C 18 H 38 CH 2 CH 2 CH 3

Oil and Water Don’t Mix • Oil is nonpolar • Water is polar “Like dissolves like” Zumdahl, De. Coste, World of Chemistry 2002, page 470
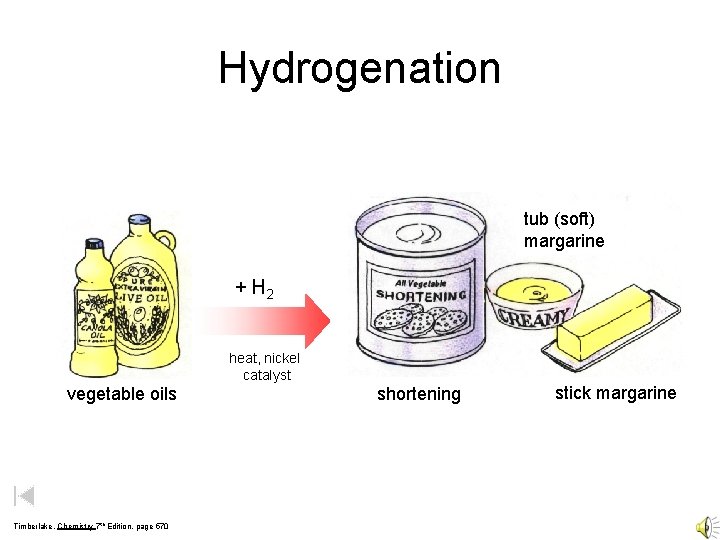
Hydrogenation tub (soft) margarine + H 2 heat, nickel catalyst vegetable oils Timberlake, Chemistry 7 th Edition, page 570 shortening stick margarine

Molecular Polarity H nonpolar molecules: -- e– are shared equally -- tend to be symmetric e. g. , fats and oils H–C–H H H H O polar molecules: -- e– NOT shared equally e. g. , water “Like dissolves like. ” like polar + polar = solution nonpolar + nonpolar = solution polar + nonpolar = suspension (won’t mix evenly)

Using Solubility Principles Chemicals used by body obey solubility principles. -- water-soluble vitamins: e. g. , vit. C -- fat-soluble vitamins: e. g. , vits. A, D Dry cleaning employs nonpolar liquids. -- polar liquids damage wool, silk -- also, dry clean for stubborn stains (ink, rust, grease) -- (tetra) perchloroethylene is in common use Cl Cl C=C Cl Cl
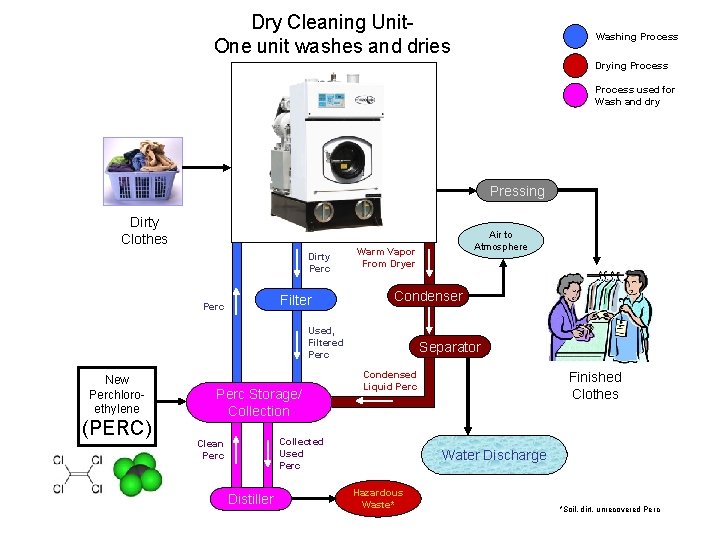
Dry Cleaning Unit. One unit washes and dries Washing Process Drying Process used for Wash and dry Pressing Dirty Clothes Dirty Perc Filter Perc Warm Vapor From Dryer Condenser Used, Filtered Perc New Perchloroethylene (PERC) Perc Storage/ Collection Separator Condensed Liquid Perc Collected Used Perc Clean Perc Distiller Air to Atmosphere Finished Clothes Water Discharge Hazardous Waste* *Soil, dirt, unrecovered Perc
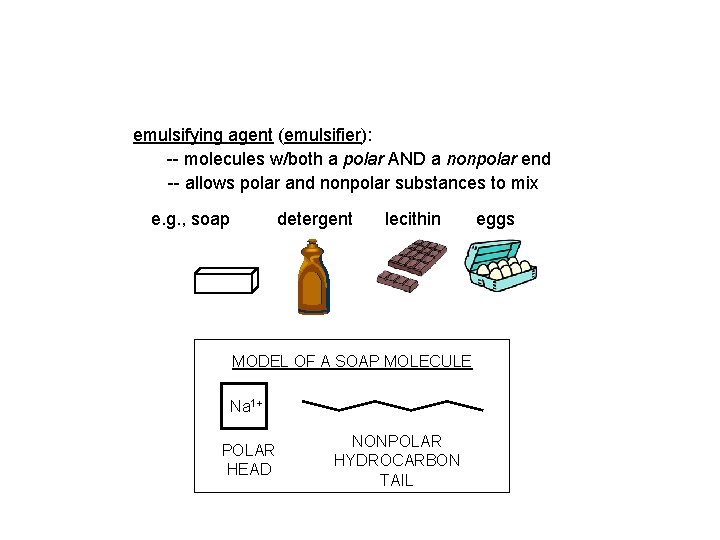
emulsifying agent (emulsifier): -- molecules w/both a polar AND a nonpolar end -- allows polar and nonpolar substances to mix e. g. , soap detergent lecithin MODEL OF A SOAP MOLECULE Na 1+ POLAR HEAD NONPOLAR HYDROCARBON TAIL eggs
- Slides: 7