a Edible fats and oils Learning intention Learn

a) Edible fats and oils Learning intention Learn about the characteristic properties of fats and oils and study how they are formed by a condensation reaction of glycerol with fatty acids.

Fats in the Diet Fats provide more energy per gram than carbohydrates. Fat molecules are insoluble, and tend to group together and form a large droplet. We store our extra energy as fat. The type of fat we eat is important. Animal fats contain important fat soluble vitamins. Oils, are thought to be healthier than solid fats, as they are less likely to be deposited inside our arteries.

Fats and oils Naturally occuring Animal fat lard suet Marine oil Vegetable oil sunflower oil coconut oil cod liver oil whale oil

Fats and Oils 50% of your brain is fat. Fats and oils are a range of substances all based on glycerol, propane-1, 2, 3 -triol. Natural fats and oils are a mixture of triglyceride compounds. Each OH group can combine chemically with one carboxylic acid molecule. The resulting molecules are fats and oils. They are described as triglycerides. The hydrocarbon chain in each can be from 4 to 24 C’s long. The C’s can be single bonded (saturated) or double bonded (unsaturated). Glycerol propane-1, 2, 3 -triol a trihydric acid
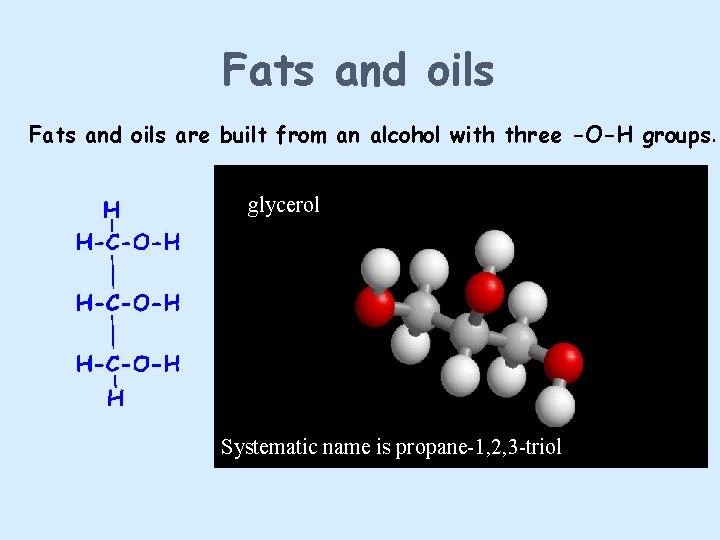
Fats and oils are built from an alcohol with three -O-H groups. glycerol Systematic name is propane-1, 2, 3 -triol
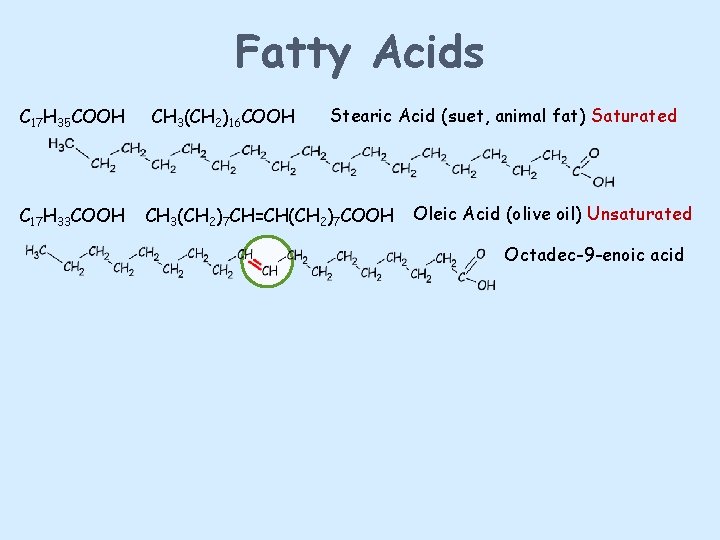
Fatty Acids C 17 H 35 COOH C 17 H 33 COOH CH 3(CH 2)16 COOH Stearic Acid (suet, animal fat) Saturated CH 3(CH 2)7 CH=CH(CH 2)7 COOH Oleic Acid (olive oil) Unsaturated Octadec-9 -enoic acid
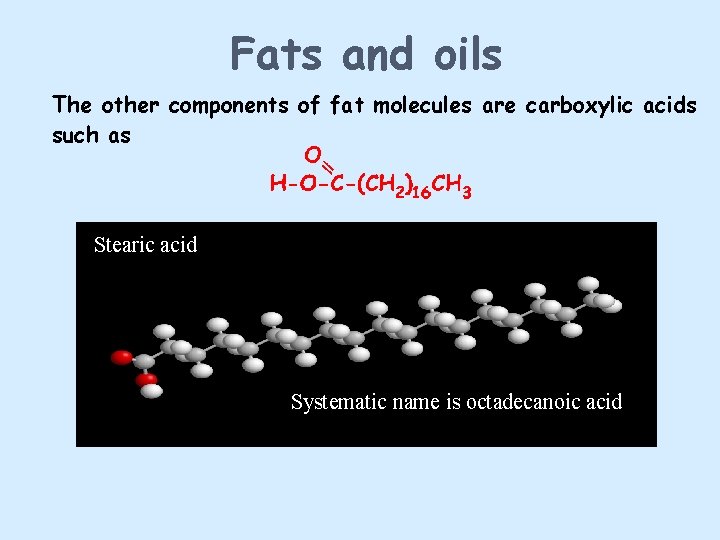
Fats and oils The other components of fat molecules are carboxylic acids such as Stearic acid Systematic name is octadecanoic acid
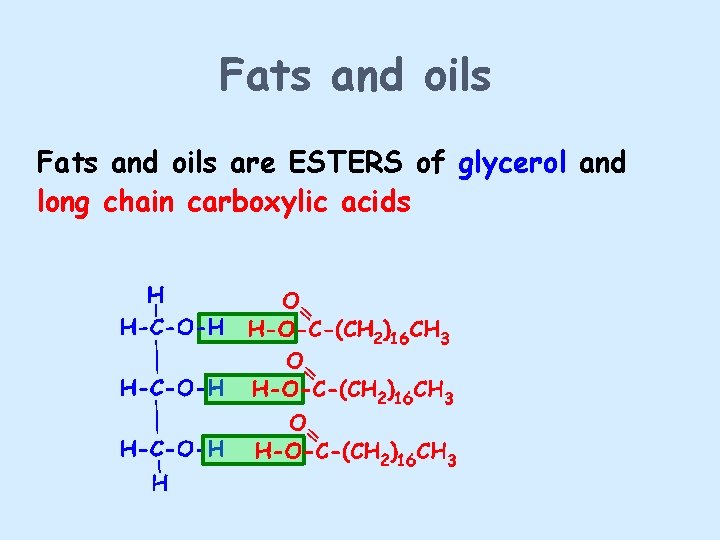
Fats and oils are ESTERS of glycerol and long chain carboxylic acids

Fats and oils Removal of water in the condensation reaction gives - The molecular formula shown above suggests that the fat molecule is shaped like an E, but the molecule is actually shaped more like this:

b) The melting point of fats and oils Learning intention Learn how differences in structure of fats and oils lead to differences in strength of intermolecular forces, and therefore to differences in melting points. .

Fats and oils Fats are mainly built from carboxylic acids with C-C single bonds. Stearic acid in beef fat Oils have some C=C bonds in the carboxylic acids from which they are made. Oleic acid in olive oil

Fats and oils Oil Double bonds in oil make the molecule less compact. Less tightly packed molecules make oils liquid. Fat molecules pack together more tightly, making fats solid at room temperature.
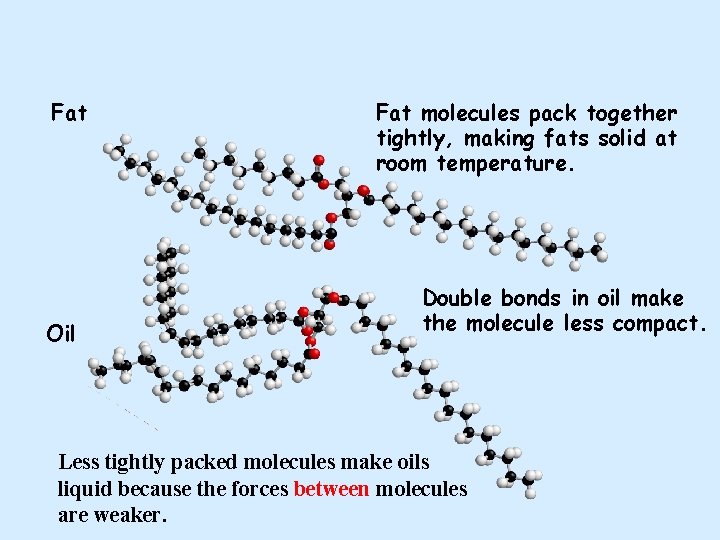
Fat Oil Fat molecules pack together tightly, making fats solid at room temperature. Double bonds in oil make the molecule less compact. Less tightly packed molecules make oils liquid because the forces between molecules are weaker.

Hydrogenation The addition of hydrogen to an unsaturated oil will ‘harden’ the oil. Increase it’s m. p. The hydrogen is added across the double bond. Used with margarine, otherwise margarine would be a liquid when taken out of the fridge.

In practice both fats and oils are mixtures of esters containing both saturated and unsaturated compounds. Beef Fat Olive oil In general oils have a higher proportion of unsaturated molecules.
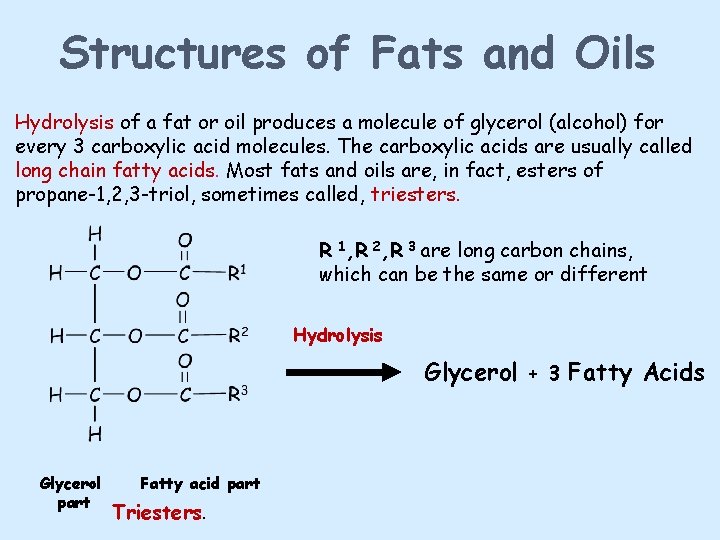
Structures of Fats and Oils Hydrolysis of a fat or oil produces a molecule of glycerol (alcohol) for every 3 carboxylic acid molecules. The carboxylic acids are usually called long chain fatty acids. Most fats and oils are, in fact, esters of propane-1, 2, 3 -triol, sometimes called, triesters. R 1, R 2, R 3 are long carbon chains, which can be the same or different Hydrolysis Glycerol + 3 Fatty Acids Glycerol part Fatty acid part Triesters.
- Slides: 17