A Determine the oxidation number for each atom

A Determine the oxidation number for each atom in the following molecules 1. H 2 S 2. P 2 O 5 3. S 8 4. SCl 2 5. Na 2 SO 3 6. SO 4 -2 7. Na. H 8. Cr 2 O 7 -2 9. Sn. Br 4 10. Ba(OH)2

A Intersection 14 12/05/06 Electrochemistry 19. 9 -19. 13 p 941 -955

A December in Studio S M 12/11 Poster session, paper due 12/17 Review session 79 pm Tu W 12/5 Exam 3 12/6 Studio 12/12 final 12/13 In. IS class assignment 12/19 Final exam 8 -10 am Th F 12/8 Polymers; check out S

A Watershed Poster Session • • Monday, December 11 in USB 2165 Board (4 ft x 4 ft), easel, pins Set up by 1: 10 and 3: 10 One person stationed at poster; others evaluate • Rubric available • Paper due same time

A Last In-Class Assignment • Wednesday, December 13 th in studio • Available on-line • Read papers before coming to class; bring them with you. • May make any notes you like on the papers • Goal: to evaluate scientific method and data

A Outline • Ed’s demos • Balancing Redox Reactions • Electrochemistry – Electrochemical cells and Standard Hydrogen Electrodes – Nernst – Quantifying current

A Ed’s Demos

A Oxidation States of Vanadium: Reduction of V 5+ to V+2 • Reaction 1 – Zn (s) + 2 VO 3 - (aq) + 8 H 3 O+ (aq) ↔ 2 VO 2+ (aq) + Zn+2 (aq) + 12 H 2 O (l) • Reaction 2 – Zn (s) + 2 VO 2+ (aq) + 8 H 3 O+ (aq) ↔ 2 V 3+ (aq) + Zn+2 (aq) + 6 H 2 O (l) • Reaction 3 – Zn (s) + 2 V 3+ (aq) ↔ 2 V 2+ + Zn+2 (aq) V+5 (aq) → V+4 (aq) yellow to green V+4 (aq) → V+3 (aq) green to blue V+3 (aq) → V+2 (aq) blue to violet

A Oxidation States of Manganese: Mn+7, Mn+6, Mn+4, and Mn+2 • +7 (purple) to +2 (colorless) – 2 Mn. O 4 - (aq) + H+ (aq) + 5 HSO 3 - (aq) ↔ 2 Mn+2 (aq) + 5 SO 4 -2 (aq) + 3 H 2 O(l) • + 7 (purple) to +4 (brown) – OH- + 2 Mn. O 4 - (aq) + 3 HSO 3 - (aq) ↔ 2 Mn. O 2 (s) + 3 SO 4 -2 (aq) + 2 H 2 O(l) • + 7 (purple) to + 6 (green) – 2 Mn. O 4 - (aq) + 3 OH- + HSO 3 - (aq) ↔ 2 Mn. O 4 -2(aq) + SO 4 -2 (aq) + 2 H 2 O(l)
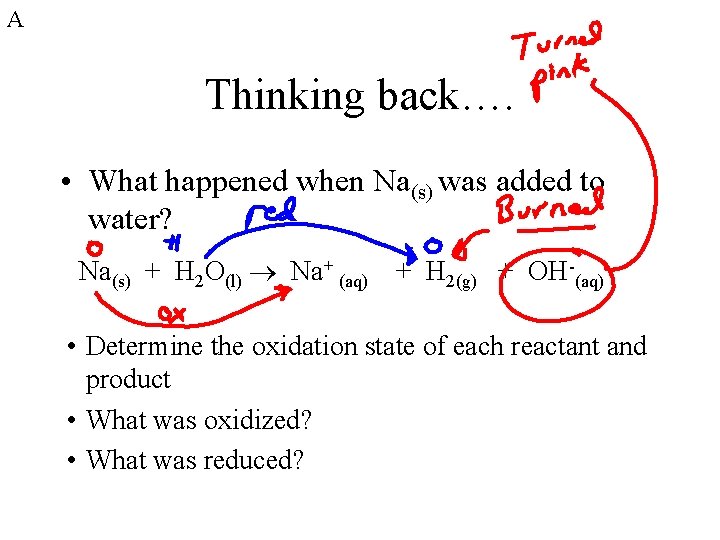
A Thinking back…. • What happened when Na(s) was added to water? Na(s) + H 2 O(l) Na+ (aq) + H 2(g) + OH-(aq) • Determine the oxidation state of each reactant and product • What was oxidized? • What was reduced?

M Balancing Redox Reactions When KMn. O 4 (potassium permanganate) is mixed with Na 2 C 2 O 4 (sodium oxalate) under acidic conditions, Mn+2(aq) ions and CO 2(g) form. The unbalanced chemical equation is: KMn. O 4(aq) + Na 2 C 2 O 4(aq) Mn+2(aq) + CO 2(g) + K+(aq) + Na+(aq) K+ and Na+ are spectator ions, so we can ignore them at this point. Mn. O 4 - (aq) + C 2 O 4 -2(aq) Mn+2(aq) + CO 2(g)

M Half-Reactions Mn. O 4 - (aq) + C 2 O 4 -2(aq) Mn+2(aq) + CO 2(g) • Reduction reaction • Oxidation reaction

M Reduction reaction Mn. O 4 - Mn+2 Step 1: Balance all elements other than oxygen and hydrogen. Step 2: Balance the oxygens by adding water. Step 3: Balance the hydrogens using H+ Step 4: Balance the electrons Mn+7 on reactants side Mn+2 on products side Step 5: Check charge balance and elemental balance

M Oxidation reaction C 2 O 4 -2 CO 2

M Combine Half Reactions 5 e- + 8 H+ + Mn. O 4 - Mn+2 + 4 H 2 O C 2 O 4 -2 2 CO 2 + 2 e- We are assuming the reaction takes place under acidic conditions!

M Balancing in Base Change H+ to water by adding OH- to each side 5 e- + 8 H+ + Mn. O 4 - Mn+2 + 4 H 2 O C 2 O 4 -2 2 CO 2 + 2 e-
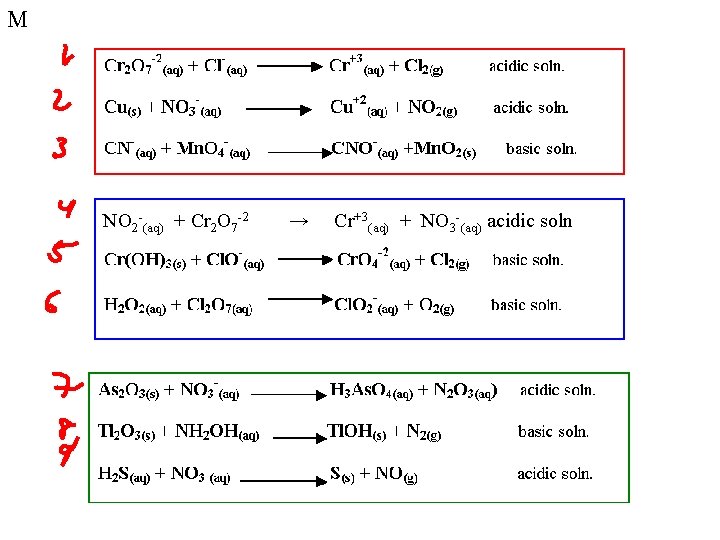
M NO 2 -(aq) + Cr 2 O 7 -2 → Cr+3(aq) + NO 3 -(aq) acidic soln

A Electrochemical Cells

A Definitions • Electrochemical cell: A combination of anode, cathode, and other materials arranged so that a product-favored redox reaction cause a current to flow or an electric current can cause a reactantfavored redox reaction to occur • Voltaic cell (battery): An electrochemical cell or group of cells in which a product-favored redox reaction is used to produce an electric current. • Galvanic cell: A cell in which an irreversible chemical reaction produces electrical current • Electrolytic cell: electrochemical reactions are produced by applying electrical energy

A A Copper-Zinc battery – What Matters? Consider reduction potentials: Cu+2 + 2 e- → Cu(s) Zn+2 + 2 e- → Zn(s) 0. 3419 V -0. 7618 V Place Zn electrode in copper sulfate solution – What happens? Copper is plated on Zn electrode Cu+2 + 2 e- → Cu(s) Zn(s) → Zn+2 + 2 e- 0. 3419 V 0. 7618 V Cu+2 + Zn(s) → Zn+2 + Cu(s) 1. 1 V E > 0, spontaneous Note, no need for electron to flow external to cell for reaction to occur!!

A A Copper-Zinc battery – What Matters? Consider reduction potentials: Cu+2 + 2 e- → Cu(s) Zn+2 + 2 e- → Zn(s) 0. 3419 V -0. 7618 V Place Cu electrode in zinc sulfate solution – What happens? Zn doesn’t plate on copper electrode? ! Cu(s) → Cu+2 + 2 e- -0. 3419 V Zn+2 + 2 e- → Zn(s) -0. 7618 V Zn+2 + Cu(s) → Cu+2 + Zn(s) -1. 1 V E < 0, not spontaneous No reaction occurs !!
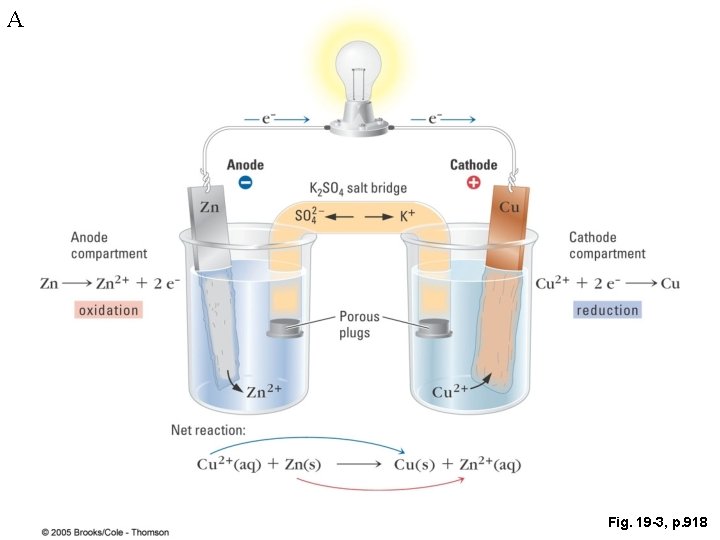
A Fig. 19 -3, p. 918
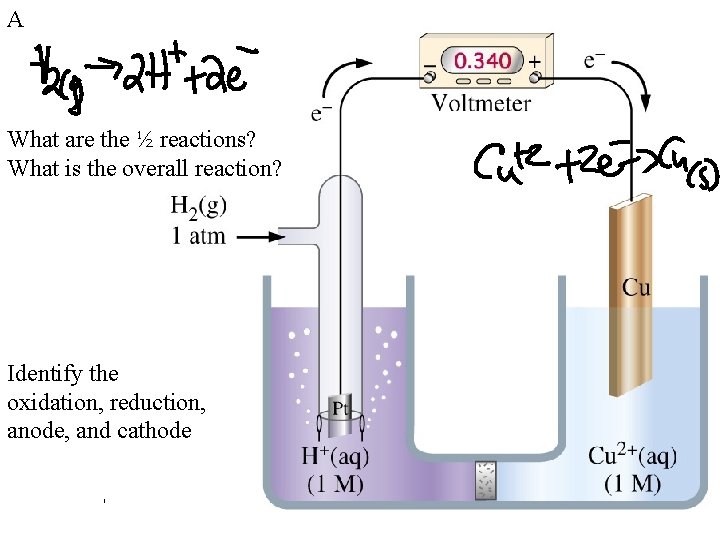
A What are the ½ reactions? What is the overall reaction? Identify the oxidation, reduction, anode, and cathode
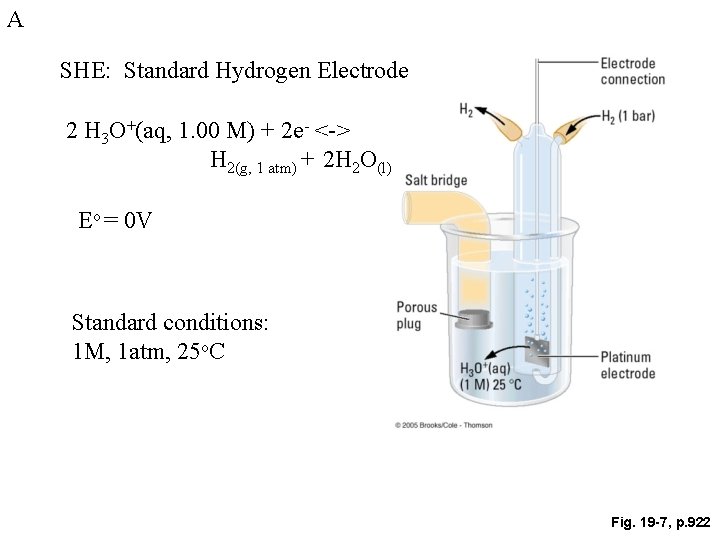
A SHE: Standard Hydrogen Electrode 2 H 3 O+(aq, 1. 00 M) + 2 e- <-> H 2(g, 1 atm) + 2 H 2 O(l) Eo = 0 V Standard conditions: 1 M, 1 atm, 25 o. C Fig. 19 -7, p. 922
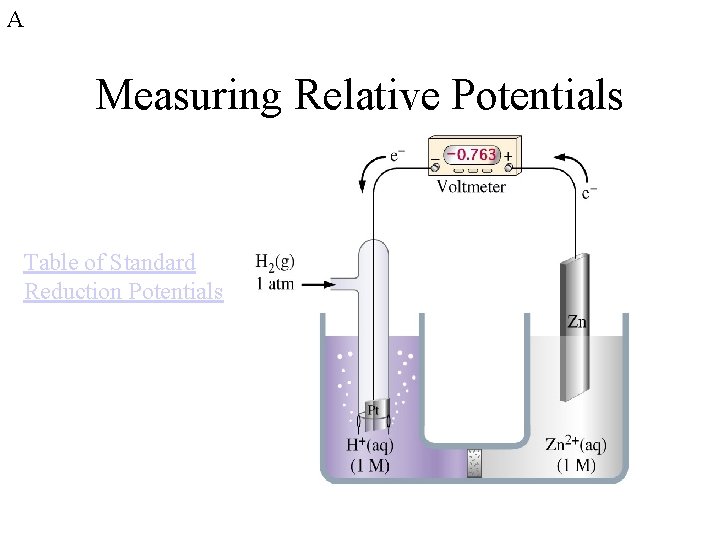
A Measuring Relative Potentials Table of Standard Reduction Potentials
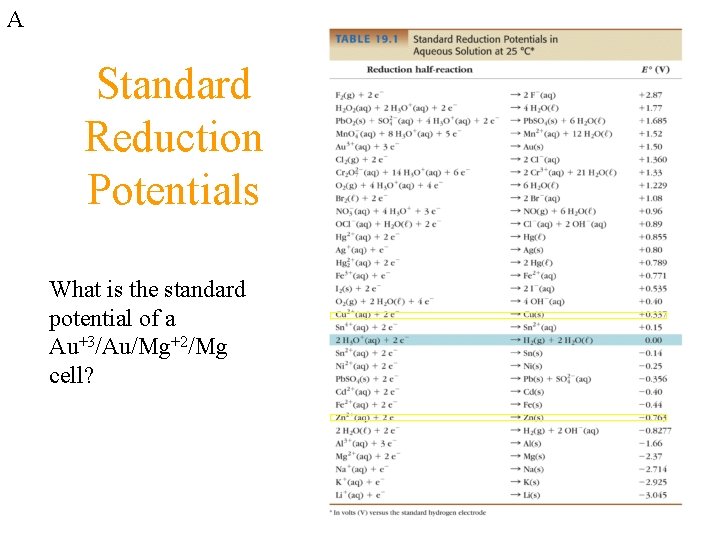
A Standard Reduction Potentials What is the standard potential of a Au+3/Au/Mg+2/Mg cell?

A The half-reaction with the more positive standard reduction potential occurs at the cathode as reduction. The half-reaction with the more negative standard reduction potential occurs at the anode as oxidation.
- Slides: 27