A CRISPR Focus Into the Attitudes and Beliefs

A CRISPR Focus Into the Attitudes and Beliefs of Sickle Cell Patients, Parents, and Providers Towards Gene Editing VENCE L. BONHAM, J. D ANITRA PERSAUD, BA HEALTH DISPARITIES UNIT | SOCIAL AND BEHAVIORAL RESEARCH BRANCH DIVISION OF INTRAMURAL RESEARCH NATIONAL HUMAN GENOME RESEARCH INSTITUTE

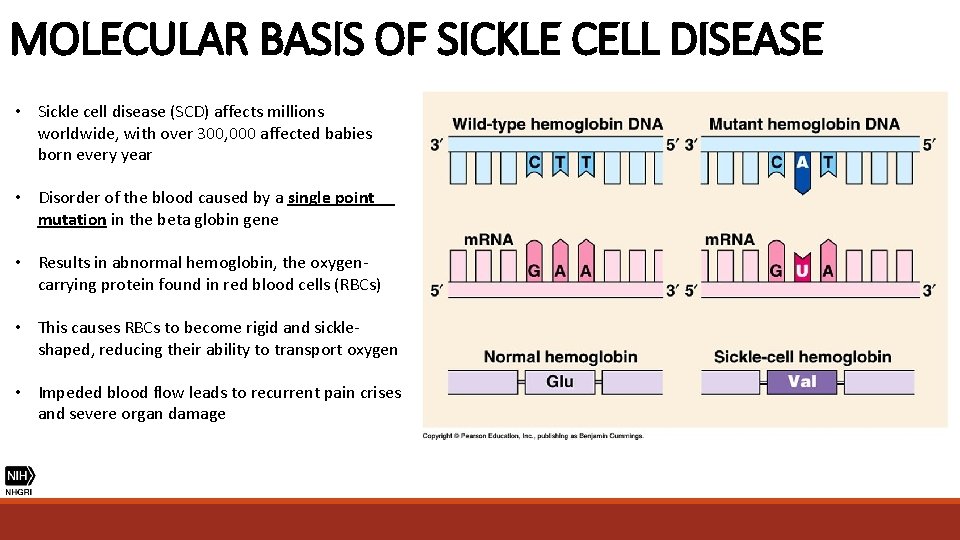
MOLECULAR BASIS OF SICKLE CELL DISEASE • Sickle cell disease (SCD) affects millions worldwide, with over 300, 000 affected babies born every year • Disorder of the blood caused by a single point mutation in the beta globin gene • Results in abnormal hemoglobin, the oxygencarrying protein found in red blood cells (RBCs) • This causes RBCs to become rigid and sickleshaped, reducing their ability to transport oxygen • Impeded blood flow leads to recurrent pain crises and severe organ damage
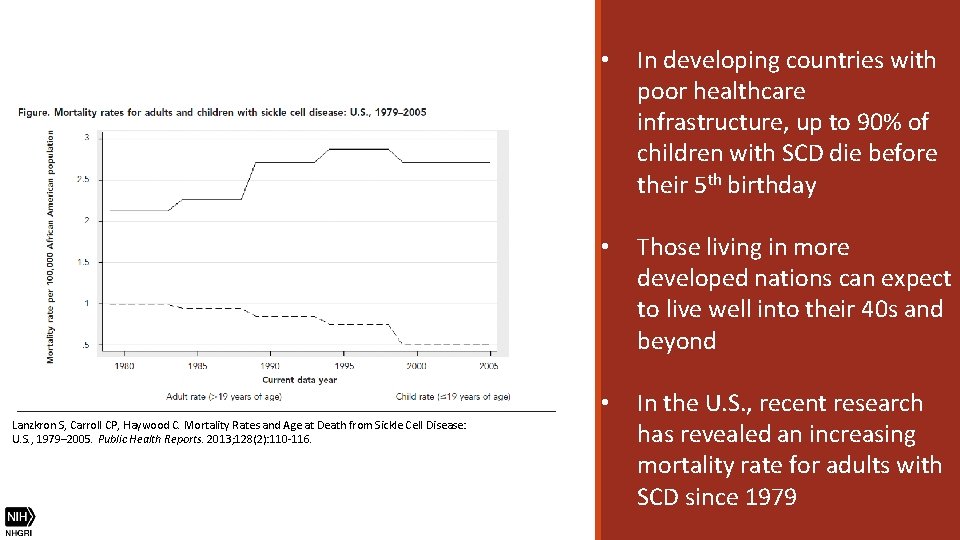
Lanzkron S, Carroll CP, Haywood C. Mortality Rates and Age at Death from Sickle Cell Disease: U. S. , 1979– 2005. Public Health Reports. 2013; 128(2): 110 -116. • In developing countries with poor healthcare infrastructure, up to 90% of children with SCD die before their 5 th birthday • Those living in more developed nations can expect to live well into their 40 s and beyond • In the U. S. , recent research has revealed an increasing mortality rate for adults with SCD since 1979
• Despite advances such as hydroxyurea, transfusion therapy, and penicillin prophylaxis, SCD patients still face significant hurdles to care • Stigma, discrimination, and clinical invisibility (e. g. labelled as drug seeking) • Less access to comprehensive team care

By 2050, the number of people with SCD is expected to increase by ~ 30% globally. Piel FB, Hay SI, Gupta S, Weatherall DJ, Williams TN (2013) Global Burden of Sickle Cell Anaemia in Children under Five, 2010– 2050: Modelling Based on Demographics, Excess Mortality, and Interventions. PLo. S Med 10(7): e 1001484. https: //doi. org/10. 1371/journal. pmed. 1001484

GENE THERAPY & SICKLE CELL DISEASE • In October 2014, a 13 -year old patient in France received a drug product called Lenti. Globin BB 305 (lentiviral-mediated gene addition of an anti-sickling variant) • Patient had a history of numerous pain crises, two episodes of acute chest syndrome, and bilateral hip osteonecrosis. He had undergone cholecystectomy and splenectomy. • After procedure, hemolysis was suppressed, hemoglobin levels returned to normal, and SCDrelated complications discontinued
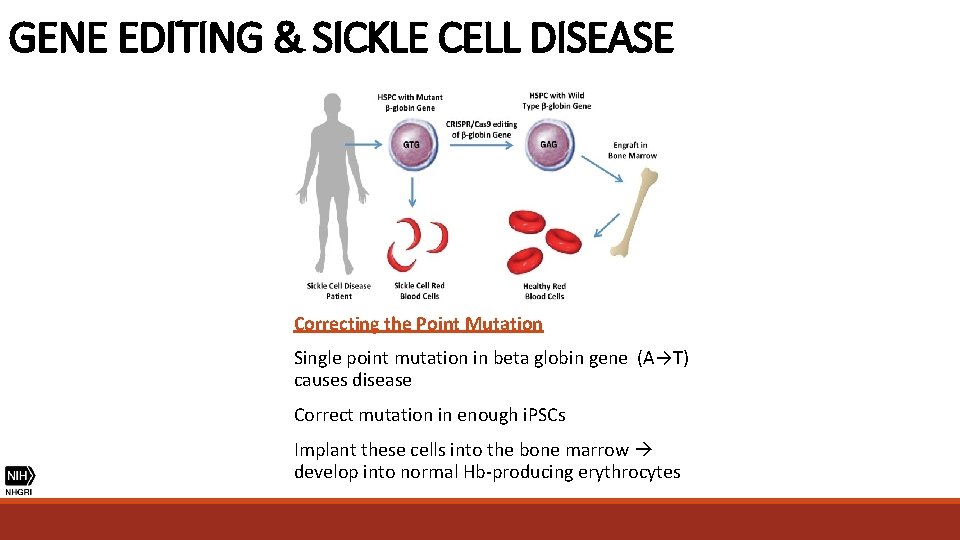
GENE EDITING & SICKLE CELL DISEASE Correcting the Point Mutation Single point mutation in beta globin gene (A→T) causes disease Correct mutation in enough i. PSCs Implant these cells into the bone marrow develop into normal Hb-producing erythrocytes

GENE EDITING & SICKLE CELL DISEASE • In 2016, scientists from UC Berkeley found a way to edit the beta globin gene in stem cells and deliver the corrected sequence via CRISPR Cas 9 • Corrected stem cells were found to be intact 16 weeks post-transplant in the mouse models
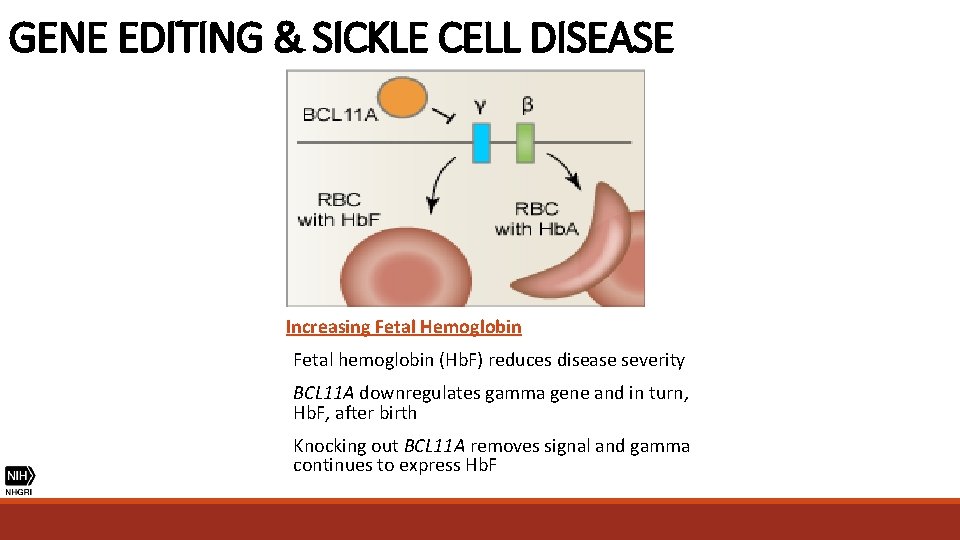
GENE EDITING & SICKLE CELL DISEASE Increasing Fetal Hemoglobin Fetal hemoglobin (Hb. F) reduces disease severity BCL 11 A downregulates gamma gene and in turn, Hb. F, after birth Knocking out BCL 11 A removes signal and gamma continues to express Hb. F

GENE EDITING & SICKLE CELL DISEASE • Sickle cell disease is milder in individuals whose RBCs carry more fetal hemoglobin (Hb. F), a type of hemoglobin resistant to sickling • In 2015, a team from MIT and Harvard discovered that deactivating the molecular switch BCLIIA enables higher levels of Hb. F
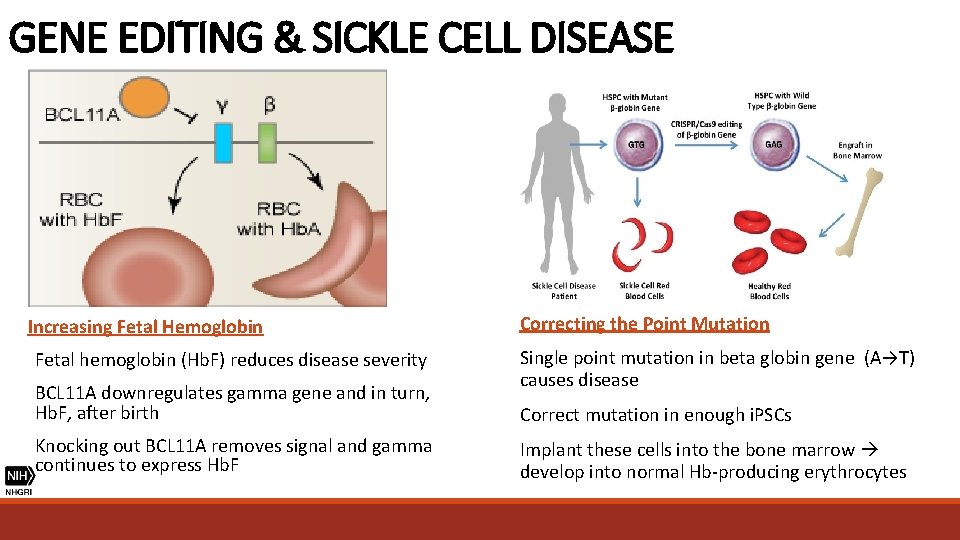
GENE EDITING & SICKLE CELL DISEASE Increasing Fetal Hemoglobin Correcting the Point Mutation Fetal hemoglobin (Hb. F) reduces disease severity Single point mutation in beta globin gene (A→T) causes disease BCL 11 A downregulates gamma gene and in turn, Hb. F, after birth Knocking out BCL 11 A removes signal and gamma continues to express Hb. F Correct mutation in enough i. PSCs Implant these cells into the bone marrow develop into normal Hb-producing erythrocytes
https: //directorsblog. nih. gov/2016/10/25/sickle-cell-disease-gene-editing-tools-point-to-possible-ultimate-cure/

RESEARCH QUESTIONS What is the genetic literacy of SCD patients and parents? What do physicians perceive the genetic literacy of their patients to be? How effective is a video-based educational tool on improving one’s understanding of CRISPR Cas 9? What are the attitudes and beliefs of SCD patients, parents, and physicians towards participation in gene editing clinical trials?
STUDY POPULATION Adults living with sickle cell disease (N=46) ◦ Must have a diagnosis of sickle cell disease ◦ 18 years of age or older ◦ Must be English speaking Parents of individuals with sickle cell disease (N=40) ◦ Must have a child (pediatric or adult) with sickle cell disease ◦ Child may be living or deceased ◦ Must be English speaking Physicians that care for individuals living with sickle cell disease (N=23) ◦ Must currently care for patients with sickle cell disease (minimum of 12 months) ◦ Must have been caregiver for at least five patients with SCD ◦ Must be English speaking
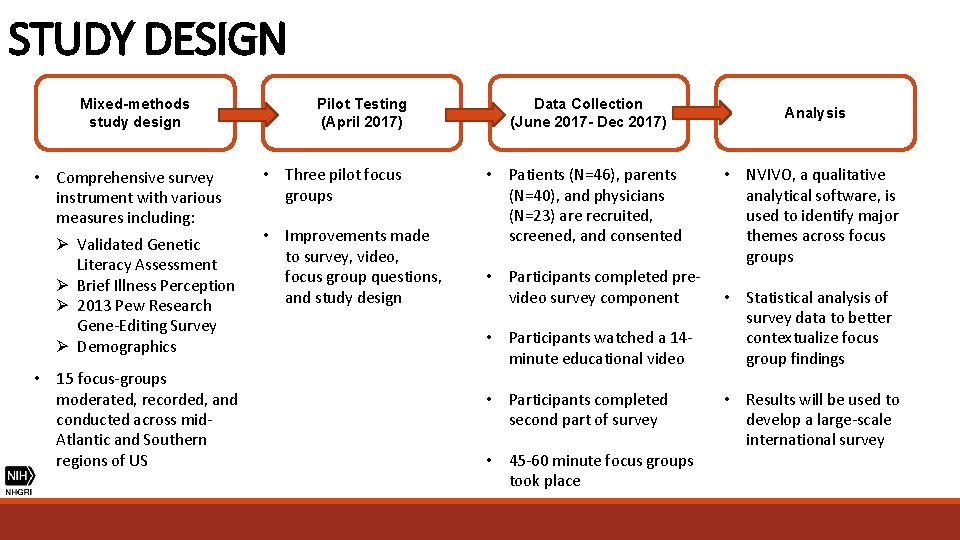
STUDY DESIGN Mixed-methods study design • Comprehensive survey instrument with various measures including: Ø Validated Genetic Literacy Assessment Ø Brief Illness Perception Ø 2013 Pew Research Gene-Editing Survey Ø Demographics • 15 focus-groups moderated, recorded, and conducted across mid. Atlantic and Southern regions of US Pilot Testing (April 2017) • Three pilot focus groups • Improvements made to survey, video, focus group questions, and study design Data Collection (June 2017 - Dec 2017) • Patients (N=46), parents (N=40), and physicians (N=23) are recruited, screened, and consented • Participants completed prevideo survey component • Participants watched a 14 - minute educational video • Participants completed second part of survey • 45 -60 minute focus groups took place Analysis • NVIVO, a qualitative analytical software, is used to identify major themes across focus groups • Statistical analysis of survey data to better contextualize focus group findings • Results will be used to develop a large-scale international survey

RECRUITMENT PROCESS Identified actively engaged stakeholders (patients, parents and providers) to work with in recruitment Partnered with established advocacy, research and education groups Invited (advocacy, support, professional) organizations to host focus groups ◦ Sent out recruitment materials to members of organizations Hosted focus groups at national conferences ◦ Sent out recruitment materials to conference attendees

EDUCATIONAL VIDEO TOOL Development • Video content based upon literature review • Collaboration with ECIB, DPCE, and other researchers Objectives • To create and evaluate an informative tool for the public • To provide study participants with relevant information to discuss Next Stages • Consolidate feedback from focus groups to inform changes

Results
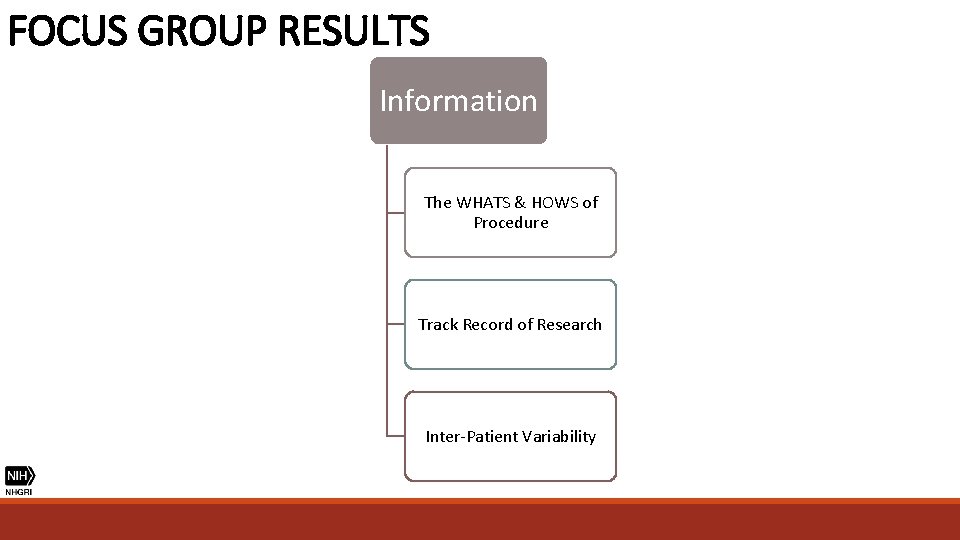
FOCUS GROUP RESULTS Information The WHATS & HOWS of Procedure Track Record of Research Inter-Patient Variability

FOCUS GROUP RESULTS “How are they going to determine who qualifies and who doesn’t? ” (Parent) “Are success rates different for the different types of sickle cell? ” (Patient) “Every sickle cell is complicated because no patient is the same. ” (Patient) INTER-PATIENT VARIABILITY

FOCUS GROUP RESULTS “Show me every animal that died and why. ” (Patient) "There’s a saying, there’s proof in the pudding. I want you to show me evidence and your findings and your results, whether it's 25 percent, 50 percent. " (Parent) "Chances of success, chances of failure, chance of death, chance of irreversible complications, known possible things that could go wrong. " (Physician) TRACK RECORD OF RESEARCH
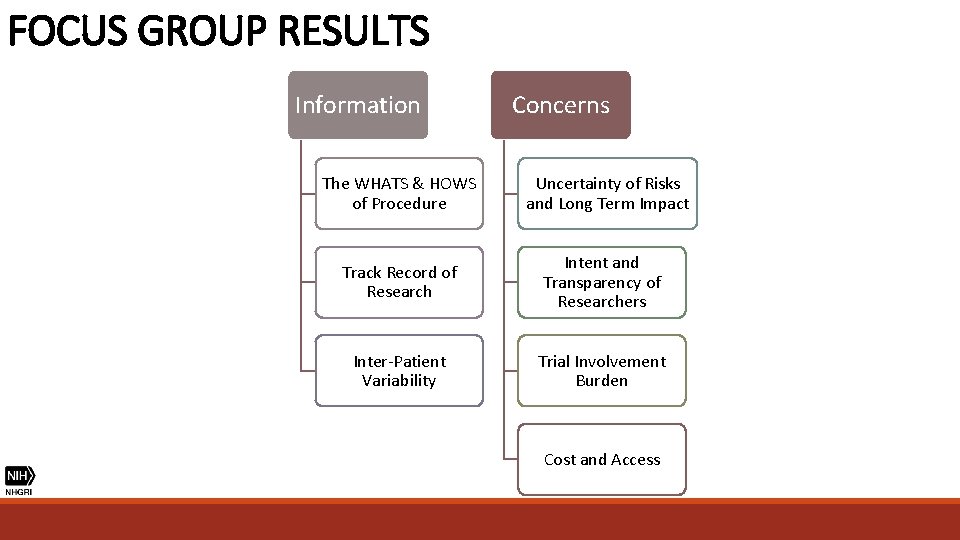
FOCUS GROUP RESULTS Information Concerns The WHATS & HOWS of Procedure Uncertainty of Risks and Long Term Impact Track Record of Research Intent and Transparency of Researchers Inter-Patient Variability Trial Involvement Burden Cost and Access

FOCUS GROUP RESULTS "If the schedule is intense then that may not work with my lifestyle because I wouldn’t want to have to miss days of work and things like that. . . because I feel like with trials you can’t like miss things. " (Patient) "Getting to and from…how much time is it going to take? Even though right now she's not working, but I am. I'm the only person working in our household. " (Parent) LEVEL OF TRIAL BURDEN
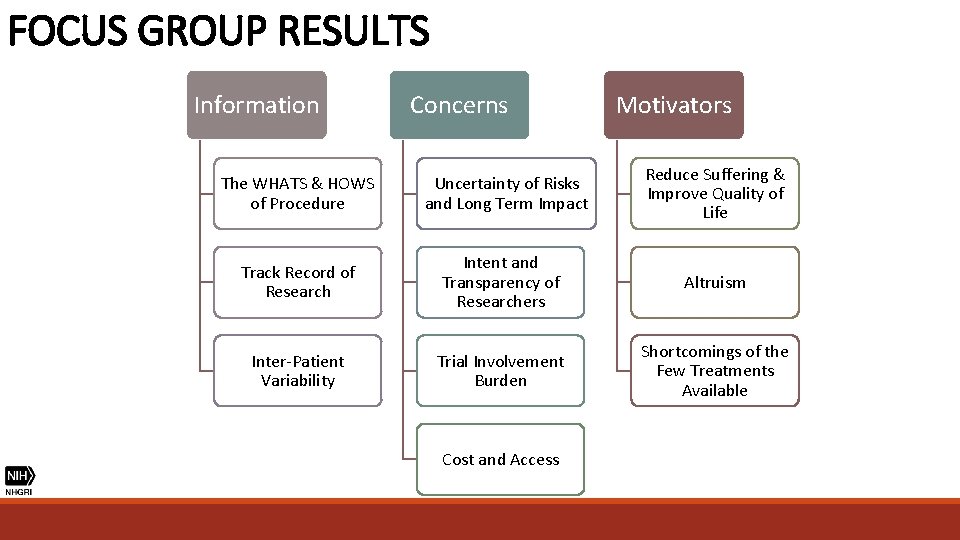
FOCUS GROUP RESULTS Information Concerns Motivators The WHATS & HOWS of Procedure Uncertainty of Risks and Long Term Impact Reduce Suffering & Improve Quality of Life Track Record of Research Intent and Transparency of Researchers Altruism Inter-Patient Variability Trial Involvement Burden Shortcomings of the Few Treatments Available Cost and Access

FOCUS GROUP RESULTS “I met somebody who was in the BABY HUGs study and their daughter was one of the first children in BABY HUGs. . . I really thanked him, because I don’t know if I would have done what he did. . . So every time I think that, I have to give myself pause and say, well, if not me than who? Because it is so important and we all want healthy children and somebody did that…There is a thing on Discovery Channel called “First In Human”. Because somebody is the first. Somebody took that penicillin that now we take for granted and you give your child at two months. ” (Parent) ALTRIUSM
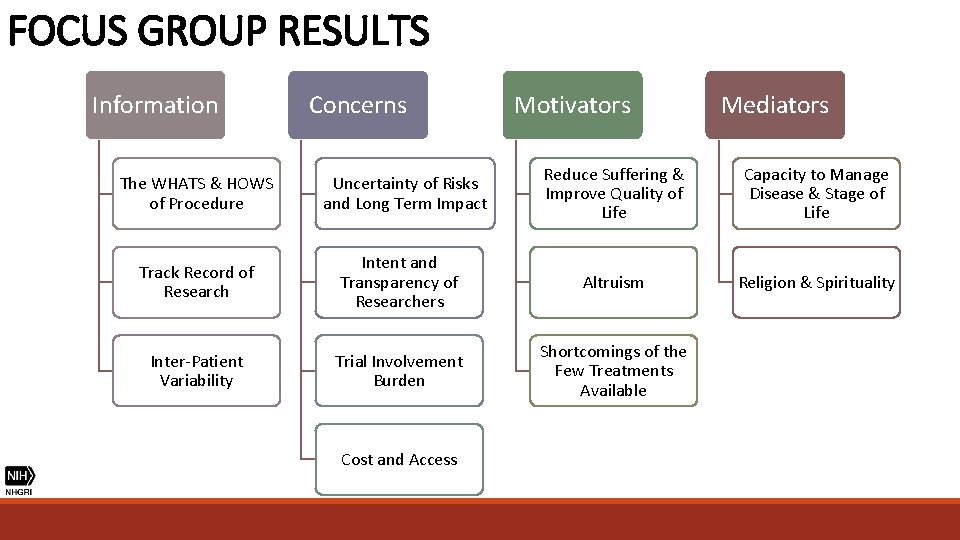
FOCUS GROUP RESULTS Information Concerns Motivators Mediators The WHATS & HOWS of Procedure Uncertainty of Risks and Long Term Impact Reduce Suffering & Improve Quality of Life Capacity to Manage Disease & Stage of Life Track Record of Research Intent and Transparency of Researchers Altruism Religion & Spirituality Inter-Patient Variability Trial Involvement Burden Shortcomings of the Few Treatments Available Cost and Access

FOCUS GROUP RESULTS “I would say that… in my 20 s it was hard. I think that if this came up in my 20 s, my husband I would have said well maybe let’s try it. … You’re a little more fearless at that age than at my age. I’ve been dealing with this for 45 years, I can deal with it for 45 more. ” (Patient) CAPACITY TO MANAGE DISEASE & STAGE OF LIFE

Who do patients and parents want to consult with most? Least? Family Physicians Researchers Other Patients Autonomous Decision

PHYSICIANS “And I think that relationships are extremely important and when you build that trust with someone, they could tell you to ride this rocket ship to the moon and you believe that they have your child’s best interests at heart. You genuinely believe that, that speaks volumes for me. ” (Parent)

RECOMMENDATIONS TO RESEARCHERS Engage and empower the community Directions Forward “Do what you can do. But maybe supporting and educating more community-based programs, being able to go into the communities. . . so that you can provide information, the pamphlets, the educational tools. . . so that the information is out there. ” (Patient) “I think for me the education component of it is really big. Everybody who has sickle cell knows somebody with sickle cell. . . The more educated we are, the more powerful we become. And then we don’t have to worry about our community being underserved because they’ll be able to advocate for themselves. . . ” (Parent)

RECOMMENDATIONS TO RESEARCHERS Engage and empower the community Directions Forward Continue to commit resources to SCD and keep going “Dedicate the resources because we as a community, deserve it. Not because we are special but because it is a compelling scientific argument. . . Sickle cell should be the first. . . No excuses. We don’t have treatments. We have one treatment that is not the best – it is what we have. We have another one, which I am not really hearing much about. ” (Parent) “Because if we could get rid of sicklecell, that would be great. . . I don’t want us to be the lowest on the totem pole. I feel like we’ve been neglected when it comes to a lot of research. . . ” (Patient)

RECOMMENDATIONS TO RESEARCHERS Engage and empower the community “Scientists and researchers should want to do this to help patients, to help people, to see them as people and not as science experiments. . . keep the patient first. ” (Parent) Directions Forward Continue to commit resources to SCD and keep going Keep all aspects of the approach patientcentric “We need a seat at the table. When this clinical trial is going on and you’ve got the researchers. . . who are above in setting up protocols, setting up how it is going to work – advocacy, CBO, patient, medical researcher, people that have sickle cell, need to be involved in every aspect of the trial. ” (Patient)

RECOMMENDATIONS TO RESEARCHERS Engage and empower the community Commit resources to SCD and move forward Directions Forward Keep all aspects of the approach patientcentric Transparency and Open Communication “It should perhaps be set up a little bit different than what scientific communications have been before, which is all through these kinds of channels or journals or NYT science page. It should be on the talk shows and on more ordinary communication. ” (Physician) “Make it accessible. Not so hidden that you have to go through hoops and back doors to find it because oftentimes that is a problem. We know the research is out there. We know the information is out there. But accessing that information is sometimes very, very difficult. ” (Parent)

EDUCATIONAL CONTENT AND COMMUNICATION What should be communicated? • Requirements of participation • Selection Criteria • Inter-Patient Variability • What Does Involvement Look Like • Details on the procedure specifics • Risks vs. Benefits • Explain how Gene Editing Works • Comparison of Gene Therapy to BMT and other pre-existing options • Breakdown of Previous Research Conducted and Results from Research How should it be communicated?
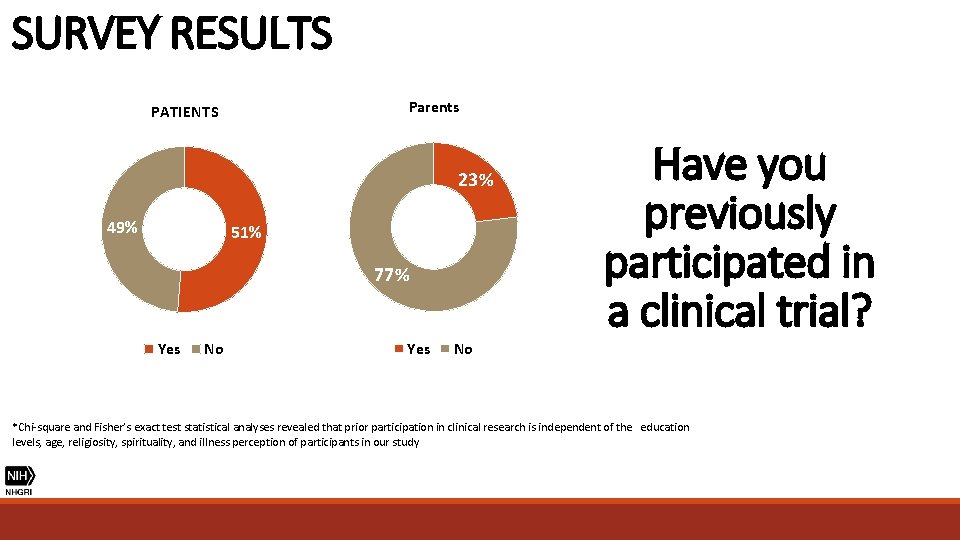
SURVEY RESULTS Parents PATIENTS 23% N= 36 N= 41 49% 51% 77% Yes No Have you previously participated in a clinical trial? Yes No *Chi-square and Fisher’s exact test statistical analyses revealed that prior participation in clinical research is independent of the education levels, age, religiosity, spirituality, and illness perception of participants in our study
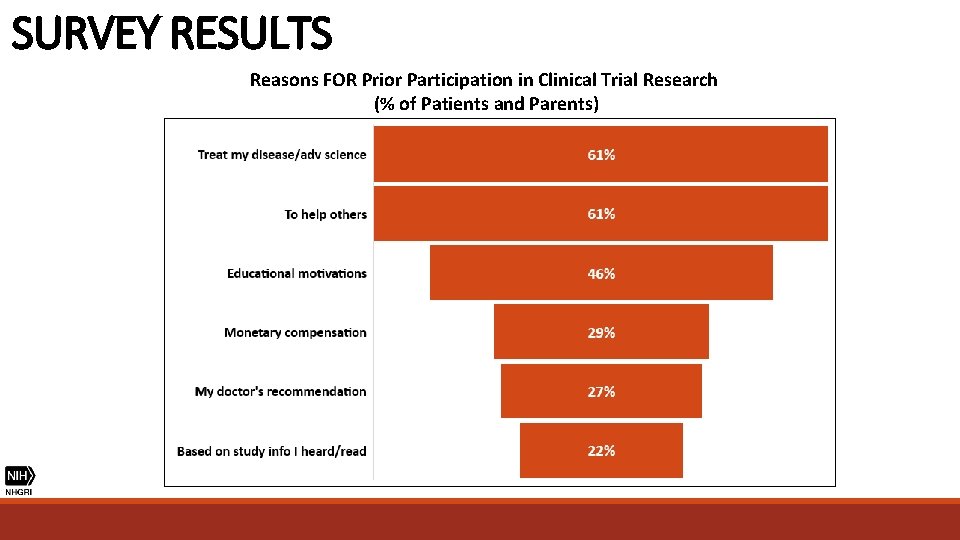
SURVEY RESULTS Reasons FOR Prior Participation in Clinical Trial Research (% of Patients and Parents)
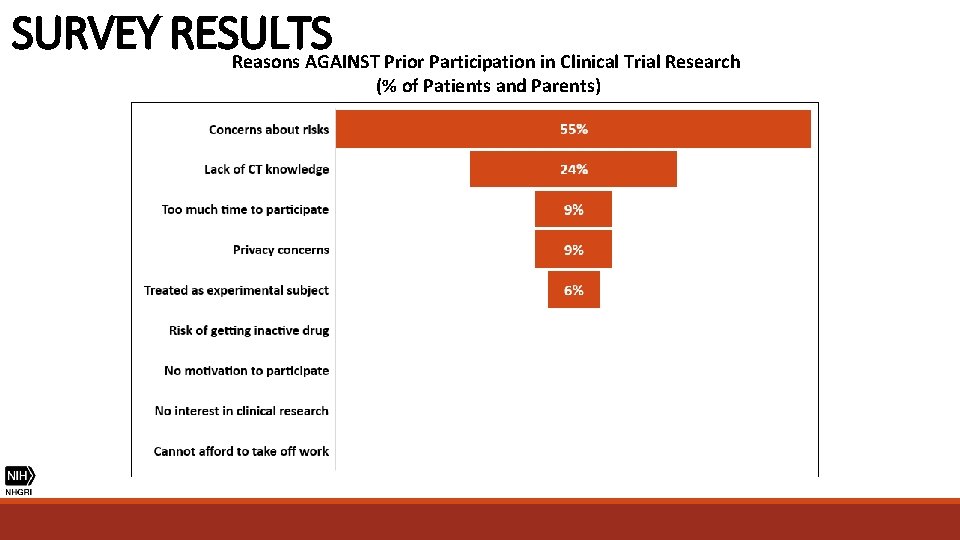
SURVEY RESULTS Reasons AGAINST Prior Participation in Clinical Trial Research (% of Patients and Parents)
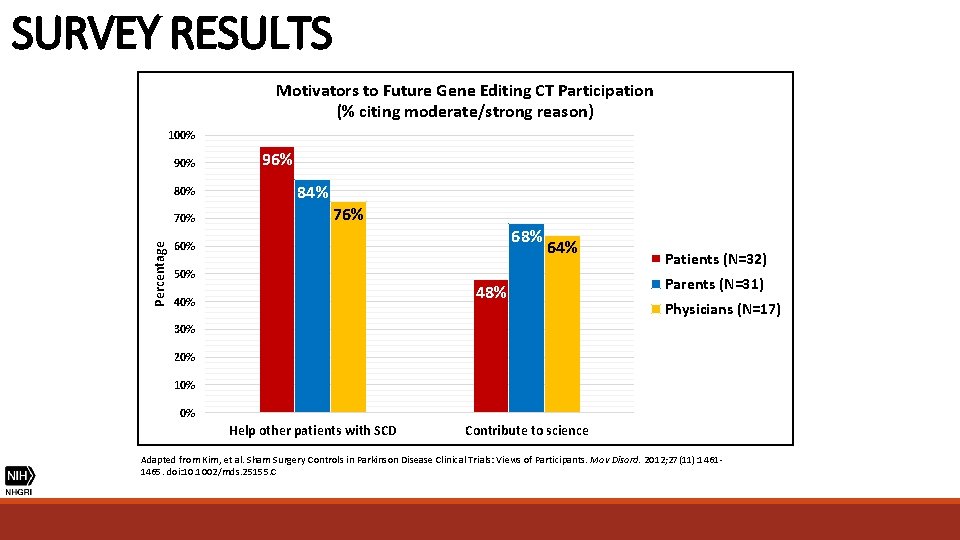
SURVEY RESULTS Motivators to Future Gene Editing CT Participation (% citing moderate/strong reason) 100% 90% 80% Percentage 70% 96% 84% 76% 68% 60% 50% 64% 48% 40% Patients (N=32) Parents (N=31) Physicians (N=17) 30% 20% 10% 0% Help other patients with SCD Contribute to science Adapted from Kim, et al. Sham Surgery Controls in Parkinson Disease Clinical Trials: Views of Participants. Mov Disord. 2012; 27(11): 14611465. doi: 10. 1002/mds. 25155. C
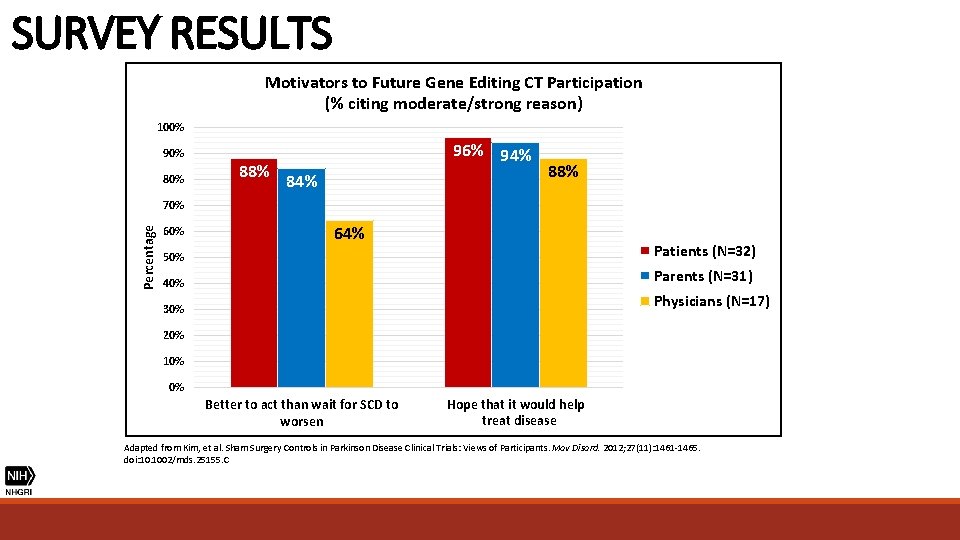
SURVEY RESULTS Motivators to Future Gene Editing CT Participation (% citing moderate/strong reason) 100% 90% 88% 96% 94% 88% Percentage 70% 64% 50% Patients (N=32) 40% Parents (N=31) Physicians (N=17) 30% 20% 10% 0% Better to act than wait for SCD to worsen Hope that it would help treat disease Adapted from Kim, et al. Sham Surgery Controls in Parkinson Disease Clinical Trials: Views of Participants. Mov Disord. 2012; 27(11): 1461 -1465. doi: 10. 1002/mds. 25155. C
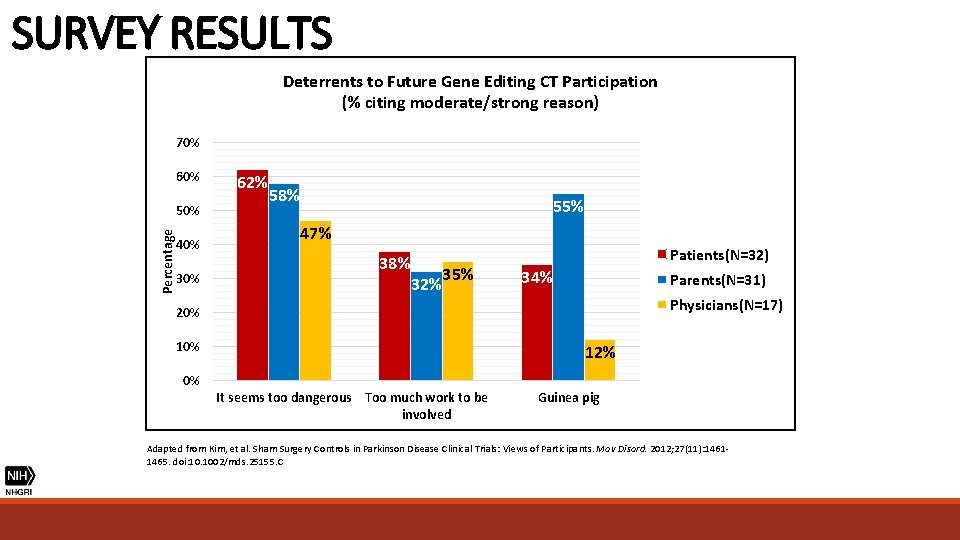
SURVEY RESULTS Deterrents to Future Gene Editing CT Participation (% citing moderate/strong reason) 70% 60% Percentage 50% 40% 30% 62% 58% 55% 47% 38% 32% 35% Patients(N=32) 34% Parents(N=31) Physicians(N=17) 20% 12% 0% It seems too dangerous Too much work to be involved Guinea pig Adapted from Kim, et al. Sham Surgery Controls in Parkinson Disease Clinical Trials: Views of Participants. Mov Disord. 2012; 27(11): 14611465. doi: 10. 1002/mds. 25155. C
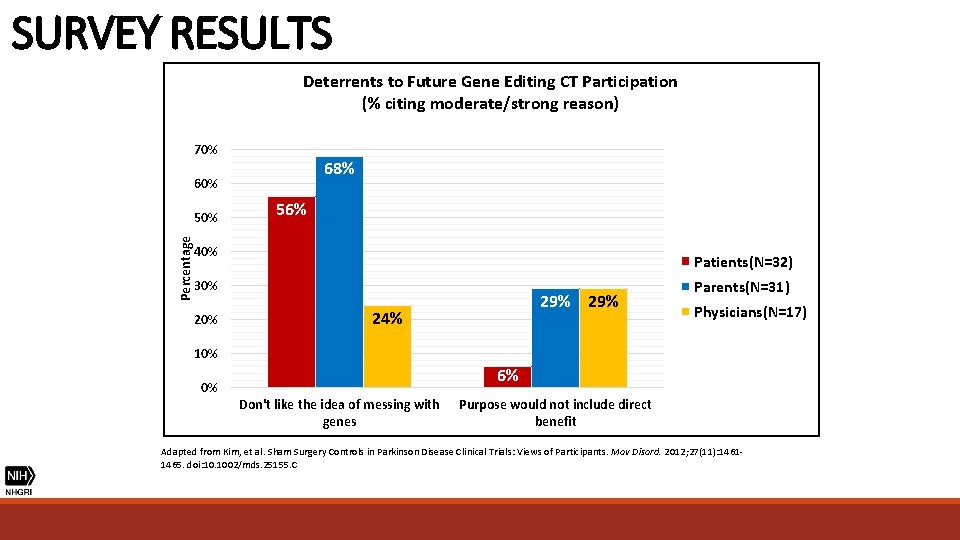
SURVEY RESULTS Deterrents to Future Gene Editing CT Participation (% citing moderate/strong reason) 70% 68% 60% Percentage 50% 56% 40% Patients(N=32) 30% 29% 24% Parents(N=31) Physicians(N=17) 10% 6% 0% Don't like the idea of messing with genes Purpose would not include direct benefit Adapted from Kim, et al. Sham Surgery Controls in Parkinson Disease Clinical Trials: Views of Participants. Mov Disord. 2012; 27(11): 14611465. doi: 10. 1002/mds. 25155. C
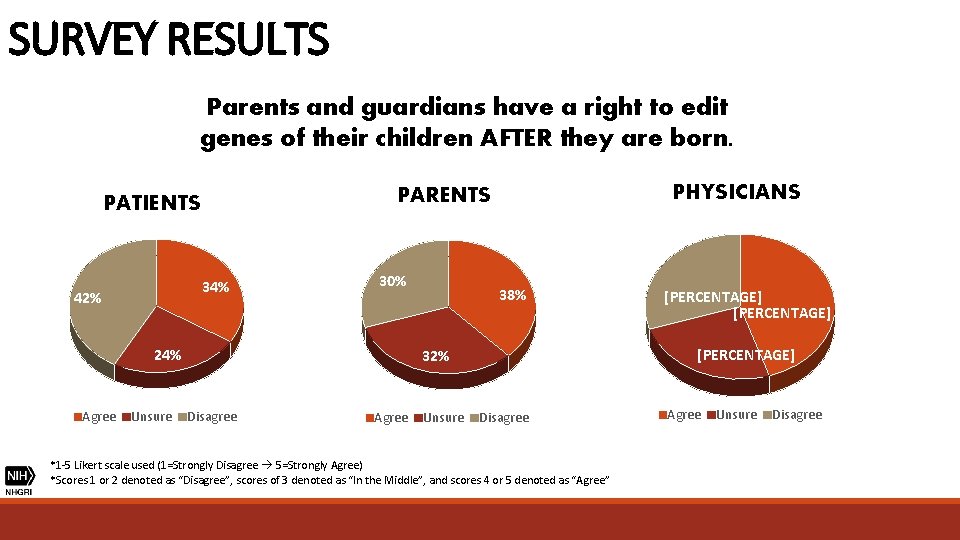
SURVEY RESULTS Parents and guardians have a right to edit genes of their children AFTER they are born. 34% 42% 30% 24% Agree Unsure PHYSICIANS PARENTS PATIENTS 38% [PERCENTAGE] 32% Disagree Agree Unsure [PERCENTAGE] Disagree *1 -5 Likert scale used (1=Strongly Disagree 5=Strongly Agree) *Scores 1 or 2 denoted as “Disagree”, scores of 3 denoted as “In the Middle”, and scores 4 or 5 denoted as “Agree” Agree Unsure Disagree
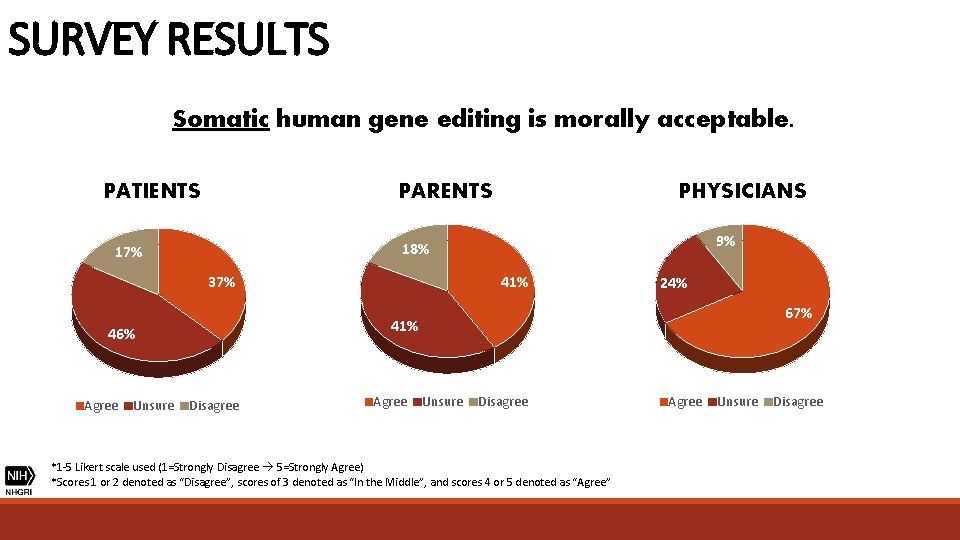
SURVEY RESULTS Somatic human gene editing is morally acceptable. PATIENTS 37% 41% 24% 67% 41% 46% Unsure 9% 18% 17% Agree PHYSICIANS PARENTS Disagree Agree Unsure Disagree *1 -5 Likert scale used (1=Strongly Disagree 5=Strongly Agree) *Scores 1 or 2 denoted as “Disagree”, scores of 3 denoted as “In the Middle”, and scores 4 or 5 denoted as “Agree” Agree Unsure Disagree
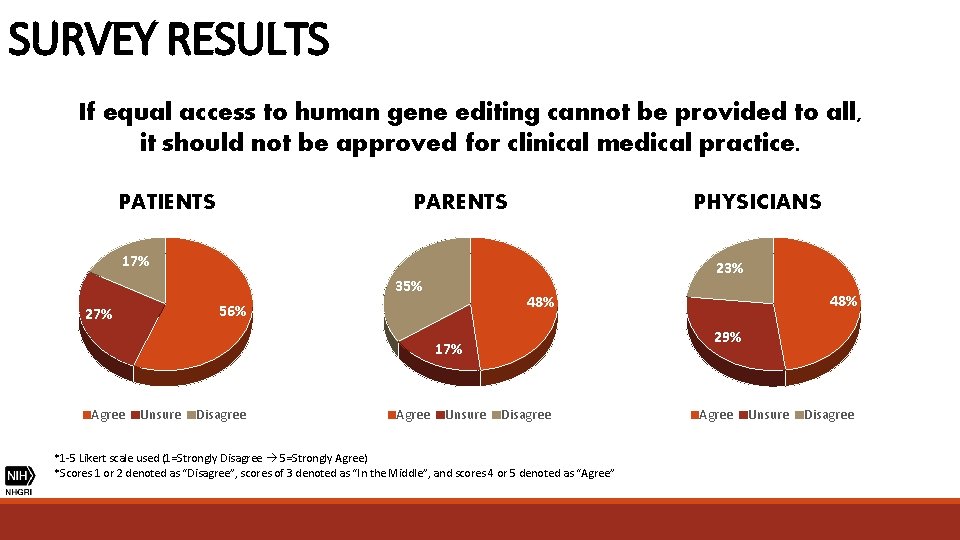
SURVEY RESULTS If equal access to human gene editing cannot be provided to all, it should not be approved for clinical medical practice. PATIENTS PHYSICIANS PARENTS 17% 23% 35% 29% 17% Agree Unsure Disagree Agree 48% 56% 27% Unsure Disagree *1 -5 Likert scale used (1=Strongly Disagree 5=Strongly Agree) *Scores 1 or 2 denoted as “Disagree”, scores of 3 denoted as “In the Middle”, and scores 4 or 5 denoted as “Agree” Agree Unsure Disagree

SUMMARY • Optimism towards a gene editing curative treatment was expressed across stakeholders due to debilitating nature of SCD and lack of existing treatments • Reservations related to the permanency of editing DNA, uncertain nature of risks and long term impact, trial burden, future access and equity, and the transparency of research enterprise surfaced • However, stakeholders provided recommendations that can inform the development of a model of engagement whereby BOTH researchers and patients benefit

ACKNOWLEGEMENTS All of the patients, parents, and physicians who graciously offered to participate. Bonham Lab Social and Behavioral Research Branch Division of Policy, Communications, and Education (NHGRI) Education and Community Involvement Branch ◦ Carla Easter, Ph. D ◦ Elizabeth Tuck Medical Arts Team ◦ Alvaro Encinas, Videographer ◦ Darryl Leja, Graphic Artist ◦ Ernesto de Aguila, Photographer

Questions
- Slides: 48