A COPY OF CREATIVE RESEARCH ENTERPRISES PRESENTATTION by

A COPY OF CREATIVE RESEARCH ENTERPRISES’ PRESENTATTION by Sheo S. Prasad At the FALL 2001 AGU MEETING 12 Dec 2001 Creative Research Enterprises’ Presentation at AGU 2001 Fall Meeting 1

TABLE OF CONTENTS • ABSTRACT • BACKGROUND • RECENT DEVELOPMENTS • EVEN MORE RECENT DEVELOPMENTS (since the abstract was submitted) • SUMMARY & CONCLUSIONS • URGENT TASKS AHED 12 Dec 2001 Creative Research Enterprises’ Presentation at AGU 2001 Fall Meeting 2

ABSTRACT • O 3(X 1 A 1) with v high enough to produce N 2 O is not generated in O(3 P), O 2 recombination • The possibility that some form of excited O 3, resulting either directly or indirectly from O(1 D), O 2 recombination, produced N 2 O in Zipf-Prasad experiment cannot be ruled out at least for now. More experiments are needed. • Pending those experiments, from an analysis of four experiments it is concluded that N 2 O is a product in the reactions of electronically excited O 3(1 B 2) and O 3(2 1 A 1) with N 2 and in the photolysis of O 3 N 2 dimer 12 Dec 2001 Creative Research Enterprises’ Presentation at AGU 2001 Fall Meeting 3

IMPORTANCE OF N 2 O - O 3 CONNECTION • O 3 & N 2 O are two very important constituents of the atmosphere. Both are greenhouse gases and O 3 shields the biosphere from harmful UV-A and UV-B. There is also a destructive relationship between the two since N 2 O is the dominant in situ stratospheric source of NO. • While the sources of the two gases are currently thought to be quite different, analyses of several experiments have suggested that some types of excited O 3 might form N 2 O. • An O 3 -N 2 O connection can be potentially important due to the stated destructive relationship. 12 Dec 2001 Creative Research Enterprises’ Presentation at AGU 2001 Fall Meeting 4

EVOLUTION OF N 2 O-O 3 CONNECTION Has twists & turns very familiar in path to progress! • Prasad, 1981; Electronically excited metastable triplet O 3 was thought to be a possible source of N 2 O based on experiments that appeared to support formation of triplet O 3 in O, O 2 recombination • Prasad, 1994; 10 experiments further support N 2 O formation via some type of excited O 3. But, by this time the prospect of metastable excited triplet was lost. So, excited O 3(X 1 A 1, very high v) from O, O 2 recombination was proposed as N 2 O source • Zipf & Prasad, 1998; O 3(X 1 A 1, very high v) appeared to explain the high yield of N 2 O in Zipf-Prasad experiment • 2001; Role of O 3(X 1 A 1) eliminated (Estupinan). But, electronically excited O 3 may be forming N 2 O after all ! (Prasad) 12 Dec 2001 Creative Research Enterprises’ Presentation at AGU 2001 Fall Meeting 5

THE RECENT DEVELOPMENT: ESTUPINAN’S NON-DETECTION OF N 2 O IN N 2/O 3 PHOTOLYSIS AT 532 NM • Estupinan’s non-detection in this experiment does not mean that N 2 O is not produced by O 3(X 1 A 1, sufficiently high v). Instead, it means that the high vibration needed to produce N 2 O is not generated when O 3(X) from O(3 P) , O 2 recombination. • Thus, the origin and identity of the species that produced N 2 O in Zipf-Prasad experiment remains an important chemical physics and atmospheric chemistry problem to be solved by more experiments. At least for now, that species may still be some form of excited O 3 resulting either directly or indirectly from O(1 D), O 2 recombination. 12 Dec 2001 Creative Research Enterprises’ Presentation at AGU 2001 Fall Meeting 6

MORE RECENT DEVELOPMENT: AN ANALYSIS OF THE SECOND SET OF ESTUPINAN’S EXPERIMENT AND THREE OTHER PREVIOUS EXPERIMENTS Pending experiments needed to better understand Zipf-Prasad’s, the rest of the presentation will dwell on an analysis of experiments by – Estupinan on N 2/O 3 photolysis at 266 nm – Gaedtke et al on on N 2/O 3 photolysis at 254 nm – Kajimoto & Cvetanovic on N 2/O 3 photolysis at 254 nm and at N 2 pressures from 27 to 113 atmospheres – De. More & Raper in liquid phase and 200 to 350 nm using a model of N 2 O quantum yield that explains the yield observed in all these experiments encompassing a very wide range of pressures and radiation wavelength and relative N 2, O 2 & O 3 amount 12 Dec 2001 Creative Research Enterprises’ Presentation at AGU 2001 Fall Meeting 7

GAEDTKE ET AL EXPERIMENT SUGGEST N 2 O FORMATION FROM PROCESS OTHER THAN O(1 D), N 2 ASSOCIATION • Gaedtke et al (1973) found N 2 O formation in photolysis of N 2/O 3 mixture at 254 nm and 1 atm pressure and determined 2. 7 x 10 -36 as the rate coefficient for the O(1 D) + N 2 + M -> N 2 O + M, if the observed N 2 O is attributed to that reaction. • Later work by Kajimoto & Cvetanovic (1975) suggested a much smaller (by factor of 7. 7) rate coefficient (or, 3. 5 x 10 -37) and this smaller coefficient is currently recommended by NASA Panel. • In retrospect, Gaedtke et al experiment imply that at lower pressures N 2 O may form more efficiently by process (es) other than O(1 D)+N 2 12 Dec 2001 Creative Research Enterprises’ Presentation at AGU 2001 Fall Meeting 8

EFFICIENT FORMATION OF N 2 O BY PROCESSES OTHER THAN O(1 D), N 2 ASSOCIATION IS CONFIRMED BY ESTUPINAN EXPERIMENT • 28 year later, Estupinan et al found a linear variation of the quantum yield N 2 O in 100 to 1000 torr pressure range, N 2 O/ PN 2 = 2. 1 x 10 -6 when PN 2 is in atm • If the observed N 2 O is attributed to O(1 D), N 2 association (as Estupinan et al did), the rate coefficient for the association reaction at 1 atm again turns out to be too large by a factor of almost 8 compared to current NASA Panel recommendation based on Kajimoto & Cvetanovic experiment. • Thus, it is urgent to search for the process(es) that could produce N 2 O more efficiently than the O(1 D), N 2 association at 1 atm. 12 Dec 2001 Creative Research Enterprises’ Presentation at AGU 2001 Fall Meeting 9

PRODUCTION FROM ELECTRONICALLY EXCITED O 3 IS A LOGICAL CHOICE • None of the other species expected in N 2/O 3 photolysis (like O 2(1 g) , O 2(b 1 )) has enough energy to produce N 2 O • In principle photolysis of O 3 with Hartley band photons can produce vibrationally excited O 2 with vibrational energy needed to possibly generate N 2 O. However, at 266 nm this too is not possible. • O 3(X, 1 B 1) with high v attainable in O, O 2 recombination has already been eliminated • Thus, O 3(1 B 2) + N 2 -> N 2 O# + O 2*# (where superscripts * and # represent, respectively, electronic and combined vibrational and translational excitations) may be the logical process. 12 Dec 2001 Creative Research Enterprises’ Presentation at AGU 2001 Fall Meeting 10

THE FACT THAT ELECTRONICALLY EXCITED O 3 HAS LIFETIME OF MOSTLY FEMTOSECONDS SHOULD NOT CAUSE MUCH CONCERN • Possibly, when a N 2 comes so close to a O 3(1 B 2) that it might react then the close proximity perturbs the dissociation dynamics to an extent that there is time to form the transition state. • The "net" reaction may involve a hitherto unrecognized, electronically excited, O 3 with shallow minimum in its potential energy surface into which a fraction of O 3(1 B 2) may change by curve crossing • Also, after all short lived O 2(B 3 ) with lifetime of ps or less is known to react 12 Dec 2001 Creative Research Enterprises’ Presentation at AGU 2001 Fall Meeting 11

RATE CONSTANT FOR O 3(1 B 2) + N 2 -> N 2 O# + O 2*# DERIVED FROM ESTUPINAN’S N 2 O IS VERY REASONABLE • At any point in the irradiated region the very small but finite number density of O 3(1 B 2) that have not as yet lost their identity is n(O 3(1 B 2)) = J n(O 3) / kdiss ; kdiss = (lifetime)-1 • The corresponding quantum yield is : (k/kdiss )n(N 2) where k is the rate constant of the title reaction • With Estupinan’s N 2 O & lifetime = 10 fs, k = 8 x 10 -12 cm 3 s 1 12 Dec 2001 Creative Research Enterprises’ Presentation at AGU 2001 Fall Meeting 12

IF O 3(1 B 2) FORMS N 2 O, THENTHE FOLLOWING SHOULD ALSO HOLD • There should be considerable N 2 O formation when O 3 is excited to the secondary minima of the 2 1 A 1 or to the quasi-bound portion of the 1 B 2 potential energy surface that are responsible for the Huggins bands (despite the negligible yield of O(1 D) in this region). • The N 2 O derived in from Estupinan’s data should be consistent with that from the high pressure data of Kajimoto and Cvetanovic. From a reinterpretation of Kajimoto & Cvetanovic and De. More & Raper experiments, using a more complete model of N 2 O, both constrains are found to be fulfilled (as will be now explained). 12 Dec 2001 Creative Research Enterprises’ Presentation at AGU 2001 Fall Meeting 13

THE MODEL OF N 2 O USED TO REINTERPRET KAJIMOTO & CVETANOVIC AND DEMORE & RAPER DATA HAS FOLLOWING FEATURES • Quantum yield from O 3(1 B 2) and O 3(2 1 A 1) [Linear in pressure p] • All elements of Kajimoto & Cvetanovic’s model of N 2 O from O(1 D), N 2 association [p 2 variation] • Contribution of O 3 N 2 + hv N 2 O+ O 2 that represents the photolysis of O 3 component of the O 3 N 2 and the O(1 D), N 2 association inside the dimer. [Also, p 2 variation] • Details are in a preprint available for distribution to those interested. 12 Dec 2001 Creative Research Enterprises’ Presentation at AGU 2001 Fall Meeting 14

THE N 2 O FOR THE O 3(1 B 2) COMPONENT FROM KAJIMOTO & CVETANOVIC AND N 2 O FROM ESTUPINAN DATA AGREE EXCELLENTLY 12 Dec 2001 Creative Research Enterprises’ Presentation at AGU 2001 Fall Meeting 15

THE N 2 O FROM O 3 EXCITED BY HUGGINS BAND DIMER EFFECT ARE ALSO EXPERIMENTAL REALITIES 12 Dec 2001 Creative Research Enterprises’ Presentation at AGU 2001 Fall Meeting 16
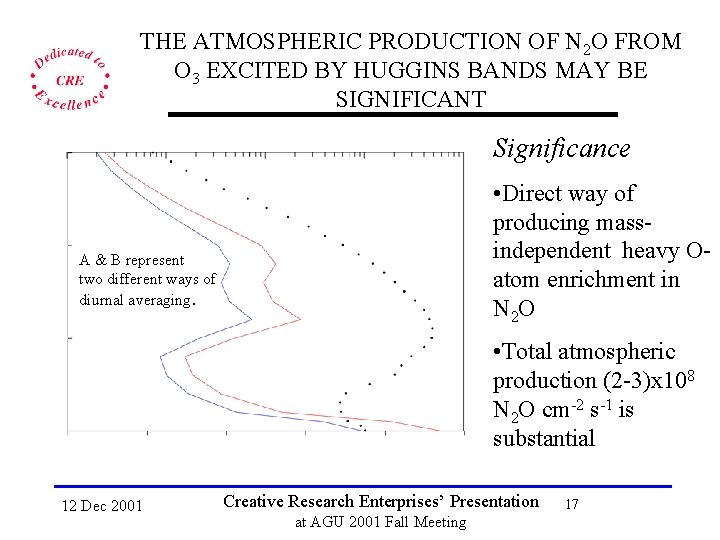
THE ATMOSPHERIC PRODUCTION OF N 2 O FROM O 3 EXCITED BY HUGGINS BANDS MAY BE SIGNIFICANT Significance • Direct way of producing massindependent heavy Oatom enrichment in N 2 O A & B represent two different ways of diurnal averaging. • Total atmospheric production (2 -3)x 108 N 2 O cm-2 s-1 is substantial 12 Dec 2001 Creative Research Enterprises’ Presentation at AGU 2001 Fall Meeting 17

SUMMARY & CONCLUDING REMARKS (#1) N 2 O is a quite possible and atmospherically significant product when O 3 is excited by Huggins band (310 -340 nm) photons in air (#2) Since this production may occur in the stratosphere, missing sinks of N 2 O are implied, if the possibility in (1) is upheld by experiments (#3) O 3(X 1 A 1) with so high v that they might produce N 2 O are not generated in O(3 P), O 2 recombination. But, this does not preclude its formation in other ways such as fluorescence from O 3(1 B 2) (#4) The identification of the species responsible for N 2 O observed by Zipf-Prasad is an important chemical physics and atmospheric chemistry problem. For now at least, that species may still be some form of excited O 3 resulting either directly or indirectly from O(1 D), O 2 recombination. 12 Dec 2001 Creative Research Enterprises’ Presentation at AGU 2001 Fall Meeting 18

EXPERIMENTAL RESEARCH TASKS THAT NEED URGENT ATTENTION • Since De. More & Raper experiment was done in condensed phase, it is most important to study the production of N 2 O with high spectral resolution when gas phase mixtures of air and O 3 are irradiated by Huggins band photons at various atmospherically significant temperatures and pressures, simultaneously examining the isotopic composition of the product N 2 O • It is also important to repeat Zipf-Prasad (ZP) and Estupinan experiments; the former with spectrally finely resolved light source spanning the range of wavelengths covered by ZP’s lamp, and the latter with O 3/air ratio tending to zero. 12 Dec 2001 Creative Research Enterprises’ Presentation at AGU 2001 Fall Meeting 19

EXPERIMENTAL RESEARCH TASKS (Cont. ) • The fact that Estupinan et al experiments done with the marvels of modern laboratory techniques gave the same answer as was obtained 28 years ago with much simpler techniques available at that time shows that • the needed set of experiments can be done with relatively simple techniques available at even moderately equipped laboratories. • It is therefore hoped that this presentation will enthuse many others to experimentally check the interpretations presented here. 12 Dec 2001 Creative Research Enterprises’ Presentation at AGU 2001 Fall Meeting 20
- Slides: 20