A Coordinated Registry Network Based on the Vascular

A Coordinated Registry Network Based on the Vascular Quality Initiative: VISION Vascular Implant Surveillance & Interventional Outcomes Network Philip P. Goodney, MS Chair, Research Advisory Committee, Society for Vascular Surgery Patient Safety Organization Art Sedrakyan, MD Ph. D, Cornell Infrastructure Center for MDEpi. Net (FDA 1 U 01 FD 005478 -01)

Launched by Society for Vascular Surgery in 2011 • Mission: To improve the quality, safety, effectiveness and cost of vascular health care by collecting and exchanging information. • 3 Components: – National Registries in a Patient Safety Organization – Regional Quality Improvement Groups • Based on Vascular Study Group of New England, 2002 – Web-based data collection - reporting system

National Registries for All Major Vascular Procedures • Carotid disease – Endarterectomy and stenting • Aortic disease – Open and endovascular abdominal aneurysm repair – Endovascular repair thoracic aorta • Lower extremity arterial disease – Bypass, interventional procedures, amputation • • Medical Management of PAD (in development) Dialysis access Vena cava filters Varicose veins
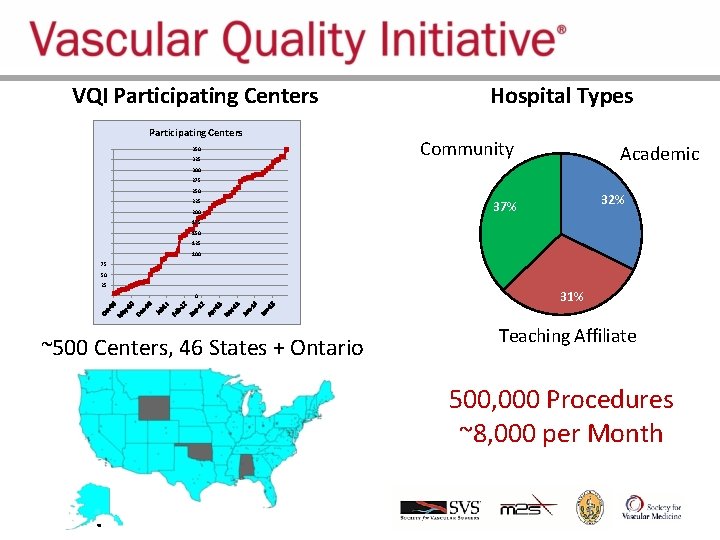
VQI Participating Centers 350 325 Hospital Types Community Academic 300 275 250 225 200 32% 37% 175 150 125 100 75 50 25 0 ~500 Centers, 46 States + Ontario 31% Teaching Affiliate 500, 000 Procedures ~8, 000 per Month .

Devices are a Key Aspect of Vascular Treatment Carotid Endarterectomy Carotid Artery Stent Endovascular AAA Repair Open AAA Repair Peripheral Vascular Intervention Infra-Inguinal Bypass Supra-Inguinal Bypass Thoracic and Complex EVAR Hemodialysis Access Lower Extremity Amputation IVC Filter Varicose Veins Devices Included Patch Type Stent, Protection Device Type Stent Graft Type Prosthetic Graft Type Balloon/Stent/Atherectomy Devices Prosthetic Graft Type Stent Graft Type Bandage, prosthesis type Device Type Ablation Device, Sclerosants
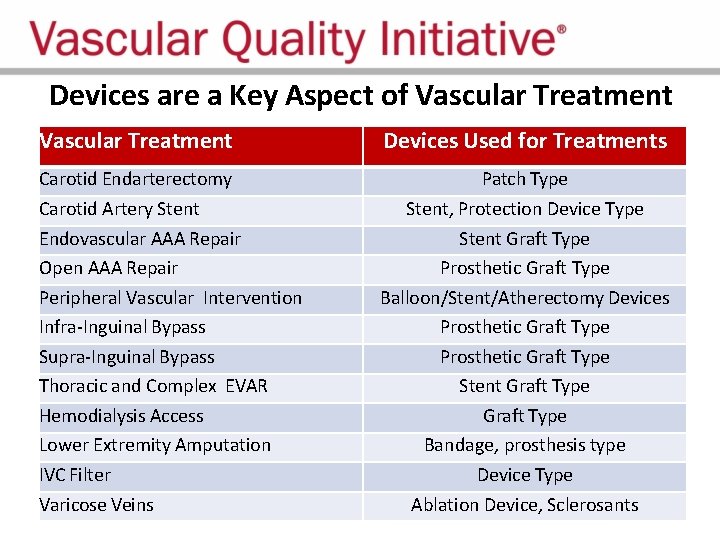
Devices are a Key Aspect of Vascular Treatment Devices Used for Treatments Carotid Endarterectomy Carotid Artery Stent Endovascular AAA Repair Open AAA Repair Peripheral Vascular Intervention Infra-Inguinal Bypass Supra-Inguinal Bypass Thoracic and Complex EVAR Hemodialysis Access Lower Extremity Amputation IVC Filter Varicose Veins Patch Type Stent, Protection Device Type Stent Graft Type Prosthetic Graft Type Balloon/Stent/Atherectomy Devices Prosthetic Graft Type Stent Graft Type Bandage, prosthesis type Device Type Ablation Device, Sclerosants

Opportunity for a Coordinated Research Network • Need for data comparing device, treatment types • Little consensus about best treatment methods – Carotid endarterectomy vs. stenting – Peripheral artery bypass vs. interventional treatment – Open vs endovascular aortic treatment • Lack of high-quality data about long-term outcomes • VQI data limited to 1 -year follow-up • Difficulty in systematic data collection

Opportunity for a Coordinated Research Network • Need for data comparing device, treatment types • Little consensus about best treatment methods – Carotid endarterectomy vs. stenting – Peripheral artery bypass vs. interventional treatment – Open vs endovascular aortic treatment • Lack of high-quality data about long-term outcomes • VQI data limited to 1 -year follow-up • Difficulty in systematic data collection

Opportunity for a Coordinated Research Network • Need for data comparing device, treatment types • Little consensus about best treatment methods – Carotid endarterectomy vs. stenting – Peripheral artery bypass vs. interventional treatment – Open vs endovascular aortic treatment • Lack of high-quality data about long-term outcomes • VQI data limited to 1 -year follow-up • Difficulty in systematic data collection

Our “VISION“ for the Data In VISION Clinical and Technical Data Available in Registries about procedures, patients, and devices Long-term, generalizable follow-up using linkages to Medicare Claims National repository of Linked Clinical. Claims Analytic Datasets for VISION Low – Cost, but High Value Data for generalizable, real-world effectiveness research

Our “VISION“ for the Data In VISION Clinical and Technical Data Available in Registries about procedures, patients, and devices Long-term, generalizable follow-up using linkages to Medicare Claims National repository of Linked Clinical. Claims Analytic Datasets for VISION Low – Cost, but High Value Data for generalizable, real-world effectiveness research

Our “VISION“ for the Data In VISION Clinical and Technical Data Available in Registries about procedures, patients, and devices Long-term, generalizable follow-up using linkages to Medicare Claims National repository of Linked Clinical. Claims Analytic Datasets for VISION Low – Cost, but High Value Data for generalizable, real-world effectiveness research

Our “VISION“ for the Data In VISION Clinical and Technical Data Available in Registries about procedures, patients, and devices Long-term, generalizable follow-up using linkages to Medicare Claims National repository of Linked Clinical. Claims Analytic Datasets for VISION Low – Cost, but High Value Data for generalizable, real-world effectiveness research

How this works…. Start With VQI Data Linkages to Medicare Claims The Dartmouth Institute Mr. Jones (name, SS#) Clinical Factors (comorbidities) Implant Data (Graft XYZ) Surgical Details (how it was placed) Surgeon Details Hospital Information Short term complications Measure Long-Term Events: • • Survival Effectiveness of the Procedure Long-Term Device Failures/Revisions Cost

time VISION is a System in Evolution Year Funding 2014 NIH U 01 (Skinner) Multi-procedure outcomes Bekelis et al, BMJ 2016 2015 NIH U 01 (Skinner) FDA U 01 (Sedrakyan) Direct Linkages Hoel et al, JVS 2017 NIH U 01 (Skinner) FDA U 01 (Sedrakyan) PCORI (O’Malley) Validation with clinical data (single site), 5 -year outcomes Columbo et al, JAMA Open 2018, JVS 2018 FDA U 01 (Sedrakyan) PCORI (O’Malley) AHA SFRN (Goodney) P 01 (Skinner) Device-specific outcomes, cost data, under-65 populations, validation with trials data (multi-site) via EDUCATE Multiple authors, in preparation 2018 Key Step Key Publication

time VISION is a System in Evolution Year Funding 2012 ARHQ R 21 (Goodney) Probabilistic Linkages, 2 -year outcomes Wallaert et al, JVS 2016 2014 NIH U 01 (Skinner) Multi-procedure outcomes Bekelis et al, BMJ 2016 2015 NIH U 01 (Skinner) FDA U 01 (Sedrakyan) Direct Linkages Hoel et al, JVS 2017 NIH U 01 (Skinner) FDA U 01 (Sedrakyan) PCORI (O’Malley) Validation with clinical data (single site), 5 -year outcomes Columbo et al, JAMA Open 2018, JVS 2018 FDA U 01 (Sedrakyan) PCORI (O’Malley) AHA SFRN (Goodney) P 01 (Skinner) Device-specific outcomes, cost data, under-65 populations, validation with trials data (multi-site) via EDUCATE Multiple authors, in preparation 2018 Key Step Key Publication

VISION: First Steps
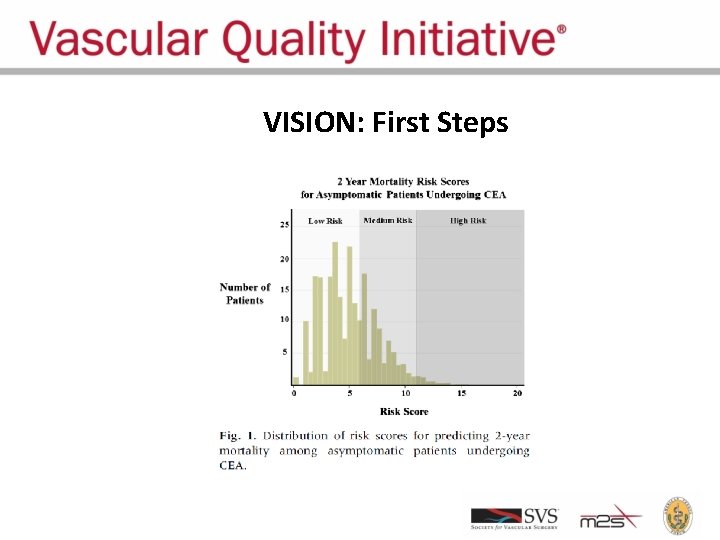
VISION: First Steps

time VISION is a System in Evolution Year Funding 2012 ARHQ R 21 (Goodney) Probabilistic Linkages, 2 -year outcomes Wallaert et al, JVS 2016 2014 NIH U 01 (Skinner) Multi-procedure outcomes Bekelis et al, BMJ 2016 2015 NIH U 01 (Skinner) FDA U 01 (Sedrakyan) Direct Linkages Hoel et al, JVS 2017 NIH U 01 (Skinner) FDA U 01 (Sedrakyan) PCORI (O’Malley) Validation with clinical data (single site), 5 -year outcomes Columbo et al, JAMA Open 2018, JVS 2018 FDA U 01 (Sedrakyan) PCORI (O’Malley) AHA SFRN (Goodney) P 01 (Skinner) Device-specific outcomes, cost data, under-65 populations, validation with trials data (multi-site) via EDUCATE Multiple authors, in preparation 2018 Key Step Key Publication

VISION: Infrastructure with Claims

time VISION is a System in Evolution Year Funding 2012 ARHQ R 21 (Goodney) Probabilistic Linkages, 2 -year outcomes Wallaert et al, JVS 2016 2014 NIH U 01 (Skinner) Multi-procedure outcomes Bekelis et al, BMJ 2016 2015 NIH U 01 (Skinner) FDA U 01 (Sedrakyan) Direct linkages Hoel et al, JVS 2017 NIH U 01 (Skinner) FDA U 01 (Sedrakyan) PCORI (O’Malley) Validation with clinical data (single site), 5 -year outcomes Columbo et al, JAMA Open 2018, JVS 2018 FDA U 01 (Sedrakyan) PCORI (O’Malley) AHA SFRN (Goodney) P 01 (Skinner) Device-specific outcomes, cost data, under-65 populations, validation with trials data (multi-site) via EDUCATE Multiple authors, in preparation 2018 Key Step Key Publication

VISION: First Matching Report
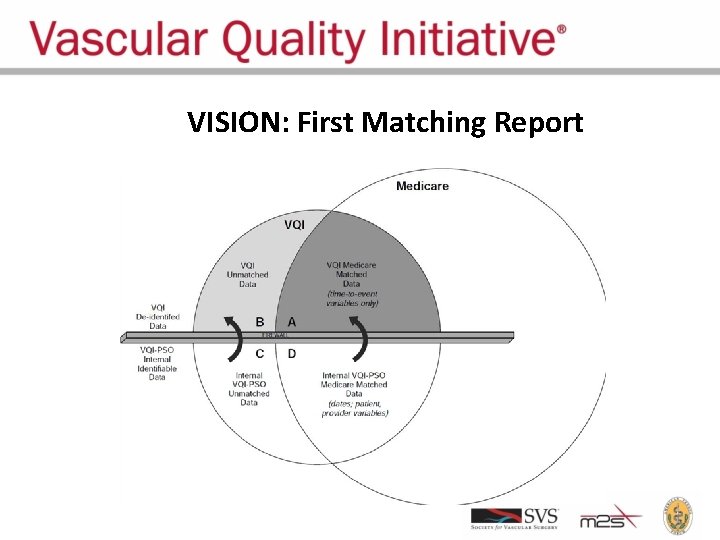
VISION: First Matching Report

time VISION is a System in Evolution Year Funding 2012 ARHQ R 21 (Goodney) Probabilistic Linkages, 2 -year outcomes Wallaert et al, JVS 2016 2014 NIH U 01 (Skinner) Multi-procedure outcomes Bekelis et al, BMJ 2016 2015 NIH U 01 (Skinner) FDA U 01 (Sedrakyan) Direct Linkages Hoel et al, JVS 2017 NIH U 01 (Skinner) FDA U 01 (Sedrakyan) PCORI (O’Malley) Validation with clinical data (single site), 5 -year outcomes Columbo et al, JAMA Open 2018, JVS 2018 FDA U 01 (Sedrakyan) PCORI (O’Malley) AHA SFRN (Goodney) P 01 (Skinner) Device-specific outcomes, cost data, under-65 populations, validation with trials data (multi-site) via EDUCATE Multiple authors, in preparation 2018 Key Step Key Publication

VISION: Validation Start With VQI Data Linkages to Medicare Claims The Dartmouth Institute
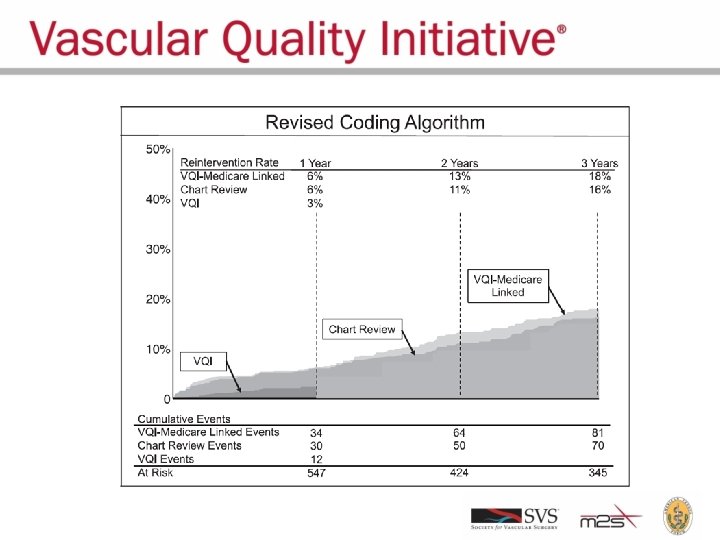
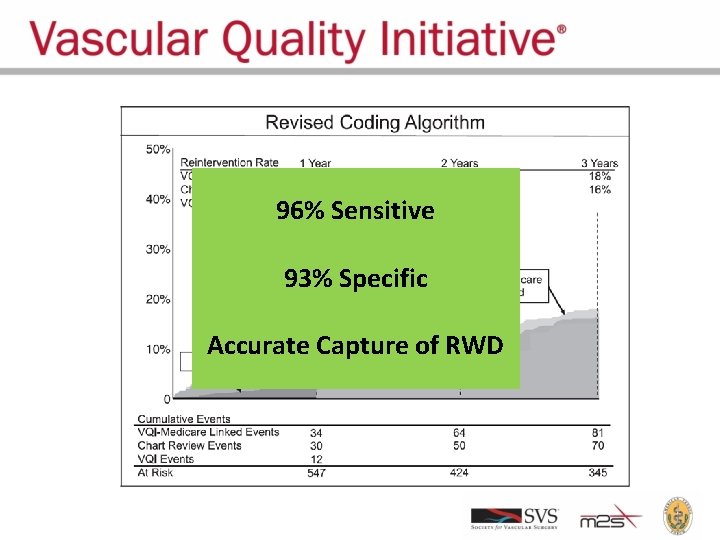
96% Sensitive 93% Specific Accurate Capture of RWD

VISION: Long-Term Survival
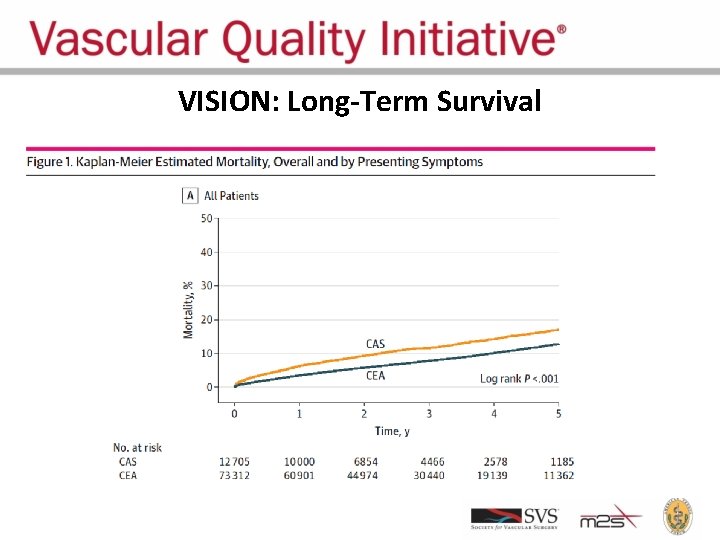
VISION: Long-Term Survival

time VISION is a System in Evolution Year Funding 2012 ARHQ R 21 (Goodney) Probabilistic Linkages, 2 -year outcomes Wallaert et al, JVS 2016 2014 NIH U 01 (Skinner) Multi-procedure outcomes Bekelis et al, BMJ 2016 2015 NIH U 01 (Skinner) FDA U 01 (Sedrakyan) Direct Linkages Hoel et al, JVS 2017 NIH U 01 (Skinner) FDA U 01 (Sedrakyan) PCORI (O’Malley) Validation with clinical data (single site), 5 -year outcomes Columbo et al, JAMA Open 2018, JVS 2018 FDA U 01 (Sedrakyan) PCORI (O’Malley) AHA SFRN (Goodney) P 01 (Skinner) Manufacturer-specific outcomes, cost data, under-65 populations, validation with trials data (multi-site) via EDUCATE Multiple authors, in preparation 2018 Key Step Key Publication
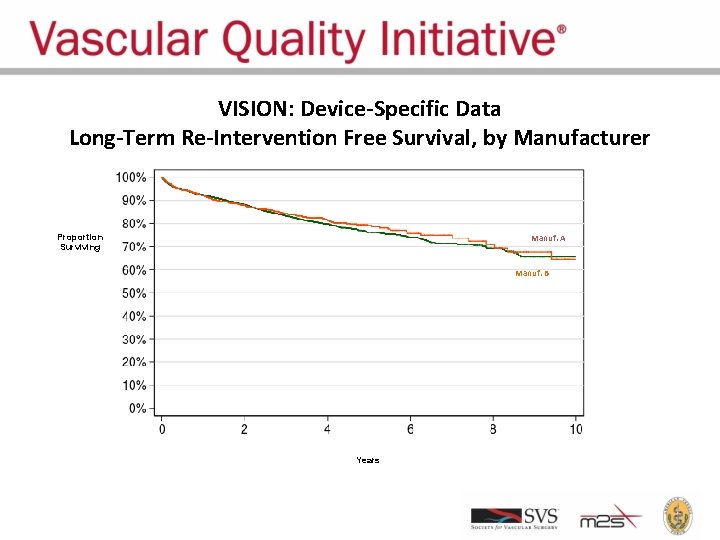
VISION: Device-Specific Data Long-Term Re-Intervention Free Survival, by Manufacturer Proportion Surviving Manuf. A Manuf. B Years
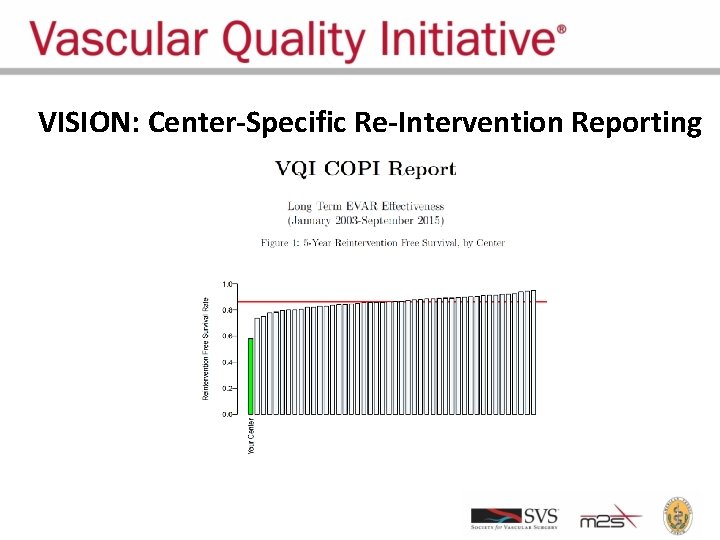
VISION: Center-Specific Re-Intervention Reporting
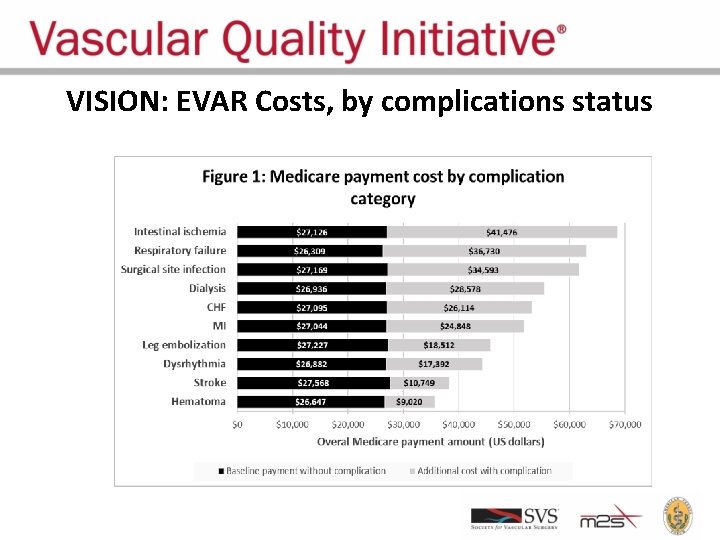
VISION: EVAR Costs, by complications status
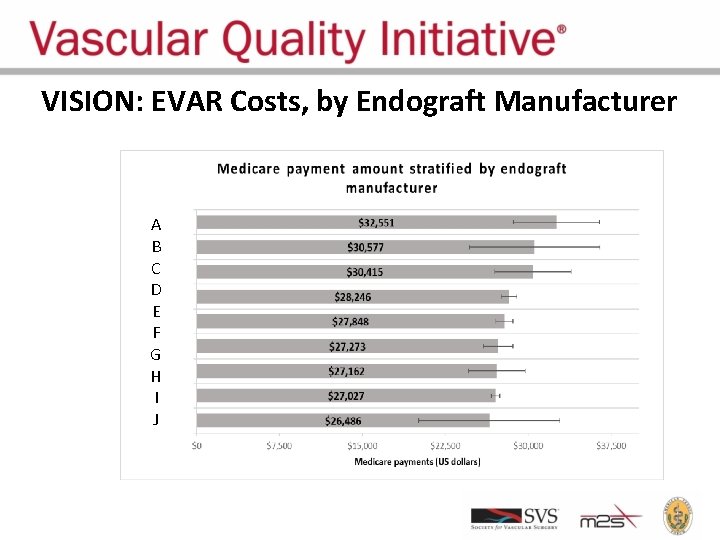
VISION: EVAR Costs, by Endograft Manufacturer A B C D E F G H I J
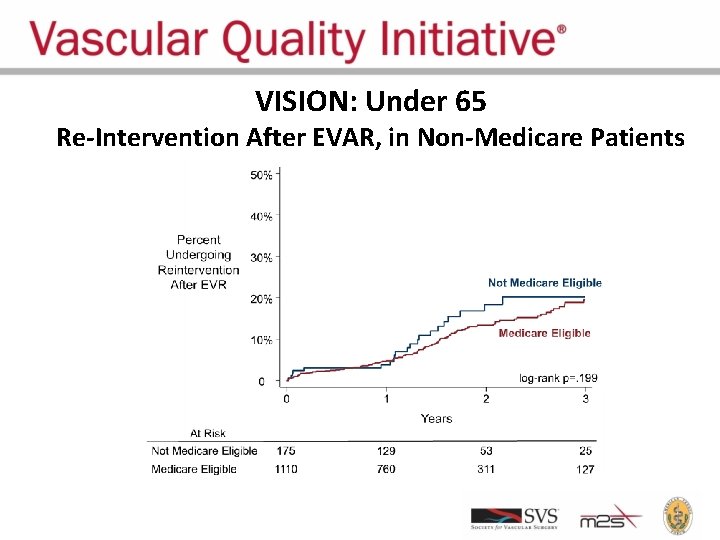
VISION: Under 65 Re-Intervention After EVAR, in Non-Medicare Patients

VISION: Multiple authors/investigators Dr. Grace Wang, UPenn Late TEVAR Outcomes Dr. Sarah Deery, Harvard/BI Deaconess Long-term Open AAA Outcomes Dr. Jessica Simons, UMass Peripheral Vascular Intervention Outcomes

Integration in other realms

VISION: What is next? • That is why we are here today. • Clinical Investigators • Industry • Regulators • Discuss and brainstorm how RWE from VISION can provide value in vascular care.

VISION: What is next? • That is why we are here today. • Clinical Investigators • Industry • Regulators • Discuss and brainstorm how RWE from VISION can provide value in vascular care.

VISION: What is next? • That is why we are here today. • Clinical Investigators • Industry • Regulators • Discuss and brainstorm how RWE from VISION can provide value in vascular care.

VISION: What is next? • That is why we are here today. • Clinical Investigators • Industry • Regulators • Discuss and brainstorm how RWE from VISION can provide value and help improve vascular care.

Thank You All For Coming !!! FDA and other Stakeholders Including Patients Device Companies for PAS and Surveillance VQI-Based Link to PCORI CDRN VISION Link to Medicare, Commercial Claims and All. Payer NY Data Inter-National Registries Thank you FDA for hosting! Let’s have a great day of discussion!
- Slides: 43