A COMMERCIAL BACKLASH IN COVID 19 DIAGNOSTICS HISTORICAL

A COMMERCIAL BACKLASH IN COVID -19 DIAGNOSTICS? HISTORICAL LESSONS INCLUDING EBOLA November 10, 2020 HIV Counseling and Testing Center – Busia, Kenya Mickey Urdea, Founder and Partner Halteres Associates © 2020 Halteres Associates LLC

Outline q q q q Background on Halteres Brief history of SARS-Co. V-2 testing The major use cases, populations tested and settings Trends and issues Opportunities Lessons from Ebola and flu H 1 N 1 Potential futures for COVID-19 and implications for testing businesses © 2020 Halteres Associates LLC

Halteres Associates, LLC q Founded in 2002, Halteres has completed >300 projects for >200 clients in countries around the world q We are a small staff of employees, supported by over 150 diagnostic and testing industry veterans; our associates q We offer strategic and operational consulting • • Technology, market and competitive assessments Market needs, customer requirements, target product profiles Product, market and business development Commercial entry strategies Health economic and business modelling Strategic partner, OEM and due diligence assessments Assistance to find and engage potential investors Helping transform medical discovery and innovation into viable solutions that can impact the world q Our commercial clients range from small start-ups to large multinationals and funders in for-profit, not-for-profit and global health markets q We have broad technology, medical device and business expertise, including • • • Molecular diagnostics, NGS, immunoassays, clinical chemistry, hematology, cytology, pathology Systems and assay development for POC and central laboratories Infectious and chronic diseases and health and wellness applications Information and communications technologies (ICT) and health IT Human, veterinary, agriculture and environmental testing and surveillance © 2020 Halteres Associates LLC

Why Do Diagnostics Companies Fail? © 2020 Halteres Associates LLC Finally Published!

The Introduction of Diagnostics for SARS-Co. V-2 q q q q The diagnostics industry is running as fast as they can to provide systems and tests, but the pace and scale are unprecedented. RNA and immunoassay tests began appearing on the market within 45 days of the announcement of the outbreak in Wuhan. At that speed, all of the tests couldn’t possibly perform as well. It appears that some of the early immunoassays had been developed for SARS (SARS-Co. V) and were simply relabeled for COVID-19 (SARS-Co. V-2). Initial PCR tests from WHO and CDC were not as good as hoped, but they pushed to get tools to the lab community By and large, the labs and manufacturers lacked viral isolates, patient samples, standards and controls By and large, they still do Many tests are being used for applications they were not designed for (e. g. , antibody tests for surveillance, antigen tests for screening) © 2020 Halteres Associates LLC

Nearly 2, 000 Tests for SARS-Co. V-2 Have Been Introduced https: //chs. asu. edu/diagnostics-commons/testing-commons © 2020 Halteres Associates LLC
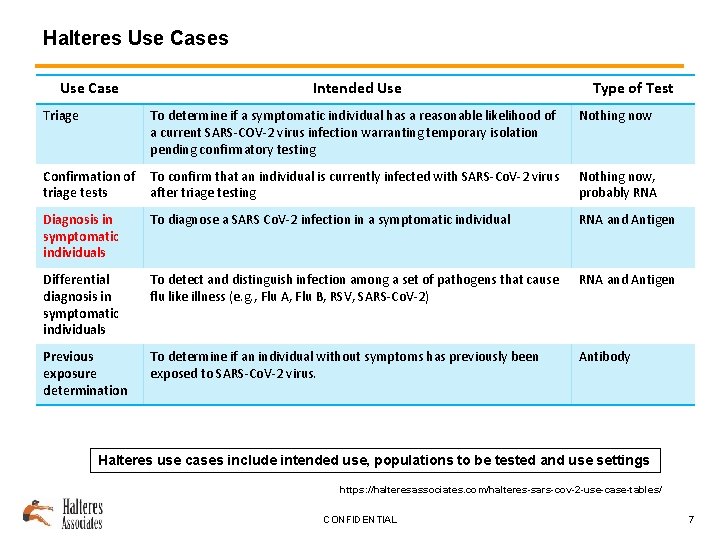
Halteres Use Case Intended Use Type of Test Triage To determine if a symptomatic individual has a reasonable likelihood of a current SARS-COV-2 virus infection warranting temporary isolation pending confirmatory testing Nothing now Confirmation of triage tests To confirm that an individual is currently infected with SARS-Co. V-2 virus after triage testing Nothing now, probably RNA Diagnosis in symptomatic individuals To diagnose a SARS Co. V-2 infection in a symptomatic individual RNA and Antigen Differential diagnosis in symptomatic individuals To detect and distinguish infection among a set of pathogens that cause flu like illness (e. g. , Flu A, Flu B, RSV, SARS-Co. V-2) RNA and Antigen Previous exposure determination To determine if an individual without symptoms has previously been exposed to SARS-Co. V-2 virus. Antibody Halteres use cases include intended use, populations to be tested and use settings https: //halteresassociates. com/halteres-sars-cov-2 -use-case-tables/ CONFIDENTIAL 7
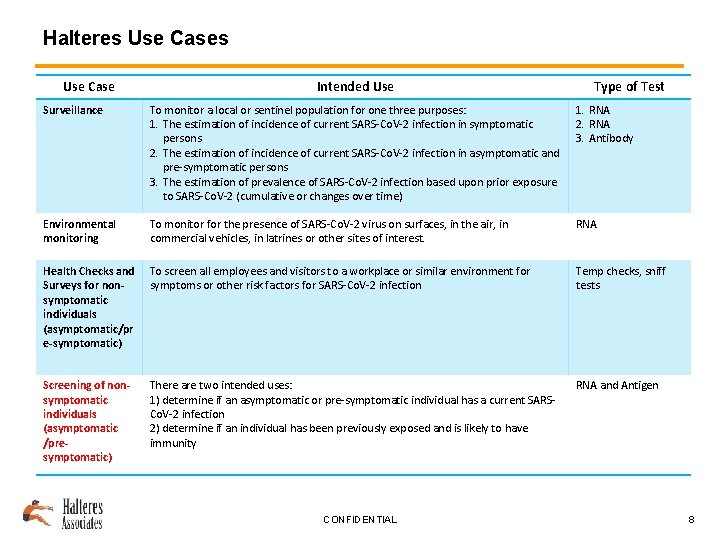
Halteres Use Case Intended Use Type of Test Surveillance To monitor a local or sentinel population for one three purposes: 1. The estimation of incidence of current SARS-Co. V-2 infection in symptomatic persons 2. The estimation of incidence of current SARS-Co. V-2 infection in asymptomatic and pre-symptomatic persons 3. The estimation of prevalence of SARS-Co. V-2 infection based upon prior exposure to SARS-Co. V-2 (cumulative or changes over time) 1. RNA 2. RNA 3. Antibody Environmental monitoring To monitor for the presence of SARS-Co. V-2 virus on surfaces, in the air, in commercial vehicles, in latrines or other sites of interest. RNA Health Checks and Surveys for nonsymptomatic individuals (asymptomatic/pr e-symptomatic) To screen all employees and visitors to a workplace or similar environment for symptoms or other risk factors for SARS-Co. V-2 infection Temp checks, sniff tests Screening of nonsymptomatic individuals (asymptomatic /presymptomatic) There are two intended uses: 1) determine if an asymptomatic or pre-symptomatic individual has a current SARSCo. V-2 infection 2) determine if an individual has been previously exposed and is likely to have immunity RNA and Antigen CONFIDENTIAL 8

9 DIAGNOSIS IN SYMPTOMATIC INDIVIDUALS • The Intended Use: § § To diagnose an active SARS Co. V-2 infection in an individual with symptoms. Note: could also be a differential diagnostics test with Flu A/B and RSV • Populations to Be Tested: § § Testing would primarily be conducted in persons with respiratory disease symptoms and typically with a physician’s prescription. In some locations, asymptomatic persons meeting certain risk criteria are permitted to be tested. – New York, Texas, Washington State, South Carolina, others • Use Settings: § § Testing sites would include locations where individuals commonly seek primary care, such as emergency rooms, urgent care clinics, hospitals and primary healthcare facilities or where individuals are referred for advanced care. There also drive-through and pop-up testing options © 2020 Halteres Associates LLC

1 0 SCREENING OF NON-SYMPTOMATIC INDIVIDUALS • The Intended Use: § § To determine if a non-symptomatic (asymptomatic or pre-symptomatic) individual has a current SARS-Co. V-2 infection. Note: could also be a differential screening test with Flu A/B and RSV • Populations to Be Tested: § § There a number of subsets of the general population currently being screened or under consideration for screening tests Healthcare staff, first responders (firemen, police), factory workers, food handlers, teachers, students, general population, athletes, others. • Use Settings: § Rapid test results enable testing to be conducted at or near the site of greatest need. – Healthcare staff at clinic or hospital – First responders at fire station or office – Factory workers, food handlers at site of business – Teachers, students at schools, colleges – Athletes at practice facilities – General population at pharmacy retail clinics, primary care clinics © 2020 Halteres Associates LLC
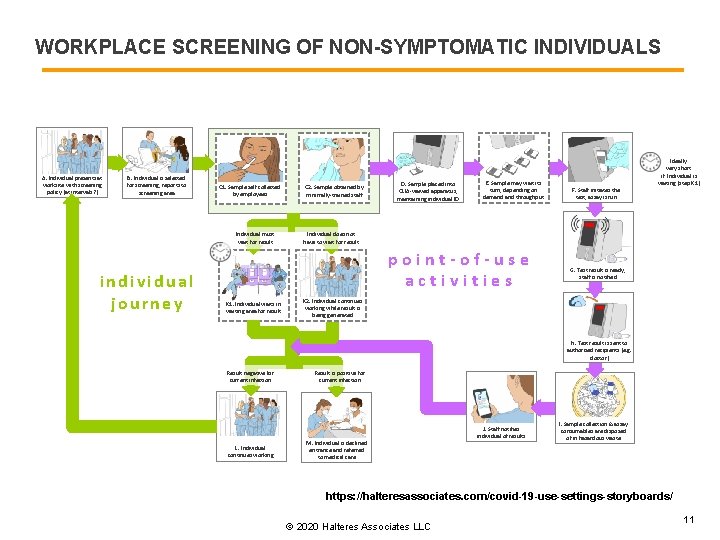
WORKPLACE SCREENING OF NON-SYMPTOMATIC INDIVIDUALS A. Individual presents at worksite with screening policy (at intervals? ) B. Individual is selected for screening, reports to screening area C 1. Sample self collected by employees Individual must wait for result individual journey C 2. Sample obtained by minimally-trained staff D. Sample placed into CLIA-waived apparatus, maintaining individual ID E. Sample may wait its turn, depending on demand throughput Individual does not have to wait for result point-of-use activities K 1. Individual waits in waiting area for result F. Staff initiates the test, assay is run Ideally very short if Individual is waiting (step K 1) G. Test result is ready, staff is notified K 2. Individual continues working while result is being generated H. Test result is sent to authorized recipients (e. g. doctor) Result negative for current infection L. Individual continues working Result is positive for current infection M. Individual is declined entrance and referred to medical care J. Staff notifies individual of results I. Sample collection & assay consumables are disposed of in hazardous waste https: //halteresassociates. com/covid-19 -use-settings-storyboards/ © 2020 Halteres Associates LLC 11

L Screening: Random and Serial • Random testing: § § § No history of a previous SARS-Co. V-2 test No predictable or scheduled repeat testing is expected A tested individual could be at any point in an infection – therefore a low limit of detection is critical • Serial testing: § § Predictable and repeated testing on a predetermined schedule If the test interval is ~3 to 5 days or less, it is likely that this strategy will detect individuals early in their infection when they have a high viral load. Technically, the first time an individual is tested counts as a random test, and then all subsequent tests would be serial A poorer limit of detection can be acceptable, because if the virus is not detected on one day because its level is too low, there is the opportunity to detect it on the next testing day when the viral load has increased and still prevent transmission. © 2020 Halteres Associates LLC
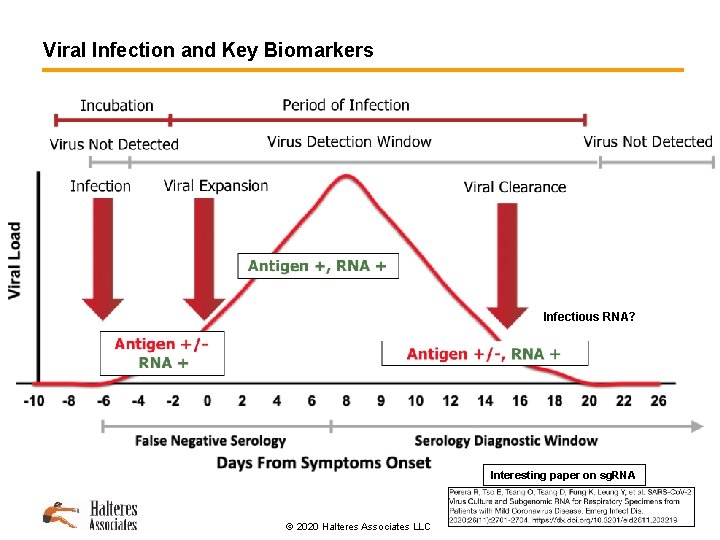
Viral Infection and Key Biomarkers Infectious RNA? Interesting paper on sg. RNA © 2020 Halteres Associates LLC

Issues with RNA vs Antigen Tests: False Negative Problem © 2020 Halteres Associates LLC
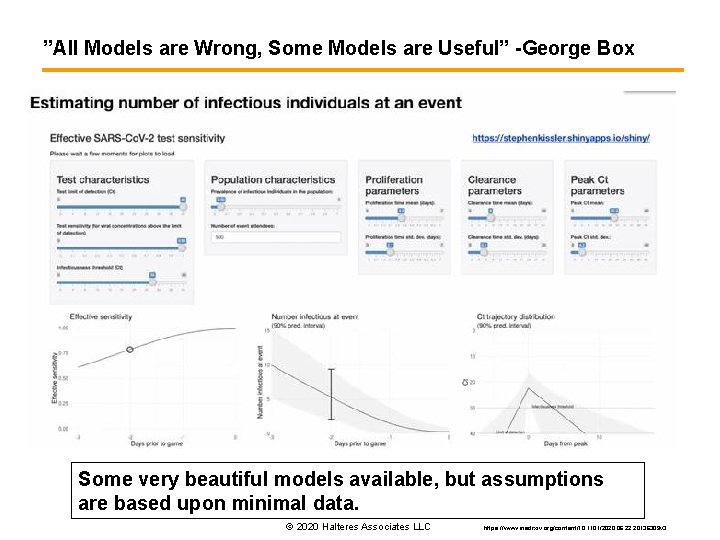
”All Models are Wrong, Some Models are Useful” -George Box Some very beautiful models available, but assumptions are based upon minimal data. © 2020 Halteres Associates LLC https: //www. medrxiv. org/content/10. 1101/2020. 06. 22. 20136309 v 3

Then Reality Bites • When Notre Dame’s football team hosted South Florida on Saturday, • • • Sept. 19, the PCR tests taken by the team 3 p. m. Friday were negative. On Monday, Notre Dame had nine positives. "Those guys didn't contract that virus between Friday and Monday, " athletic director said. "In all likelihood, it was building in their system at the time. It just hadn't Jack Swarbrick reached the level yet where it was detectable, and it's probably less transmissible during that same time, though it can be transmitted. It's both of those dynamics -- how reliable is your testing, and where are you in the virus' development in your body? ”* Were they infected Friday but the in vivo proliferation of virus was too low for detection? Did they get infected Saturday at the game and it took 2 days to reach detectable levels? Or would they have been detectable Sunday? Were they infected on Sunday? No samples were taken between Friday and Monday, so we don’t know * https: //www. espn. com/college-football/story/_/id/30154998/how-college-football-handling-covid-19 -protocols-one-month-season © 2020 Halteres Associates LLC

Antigen Test False Positive Problem: FDA Letter to Clinical Laboratory Staff and Health Care Providers “Remember that positive predictive value (PPV) varies with disease prevalence when interpreting results from diagnostic tests. PPV is the percent of positive test results that are true positives. As disease prevalence decreases, the percent of test results that are false positives increase. • For example, a test with 98% specificity would have a PPV of just over 80% in a population with 10% prevalence, meaning 20 out of 100 positive results would be false positives. • The same test would only have a PPV of approximately 30% in a population with 1% prevalence, meaning 70 out of 100 positive results would be false positives. • At 0. 1% prevalence, the PPV would only be 4%, meaning that 96 out of 100 positive results would be false positives. • Health care providers should take the local prevalence into consideration when interpreting diagnostic test results. ” FDA Website 11 -3 -20 © 2020 Halteres Associates LLC
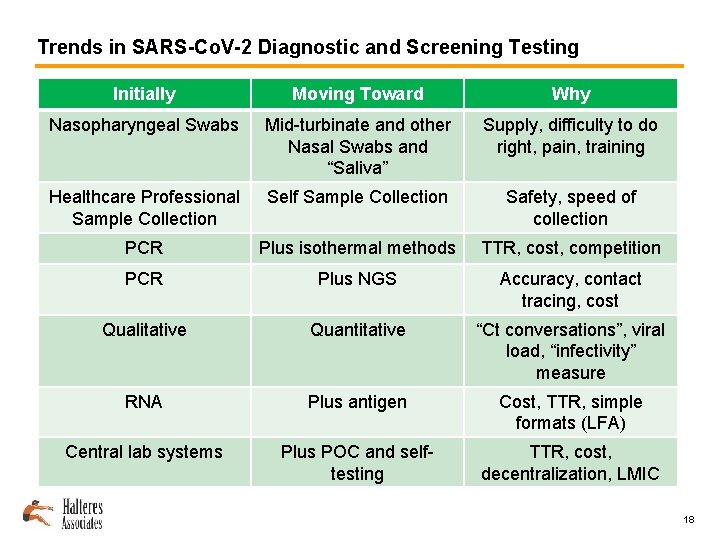
Trends in SARS-Co. V-2 Diagnostic and Screening Testing Initially Moving Toward Why Nasopharyngeal Swabs Mid-turbinate and other Nasal Swabs and “Saliva” Supply, difficulty to do right, pain, training Healthcare Professional Sample Collection Self Sample Collection Safety, speed of collection PCR Plus isothermal methods TTR, cost, competition PCR Plus NGS Accuracy, contact tracing, cost Qualitative Quantitative “Ct conversations”, viral load, “infectivity” measure RNA Plus antigen Cost, TTR, simple formats (LFA) Central lab systems Plus POC and selftesting TTR, cost, decentralization, LMIC 18

How Much Testing for SARS-Co. V-2? Rockefeller Foundation Projected US Test Needs (May 2020): 1 M/day to 30 M/day https: //covidtracking. com/data/national © 2020 Halteres Associates LLC

Testing in Some Locations Not Going in the Right Direction https: //www. nbcnews. com/health-news/covid-19 -testinglags-cases-increase-across-united-states-n 1243946 oct 19 © 2020 Halteres Associates LLC
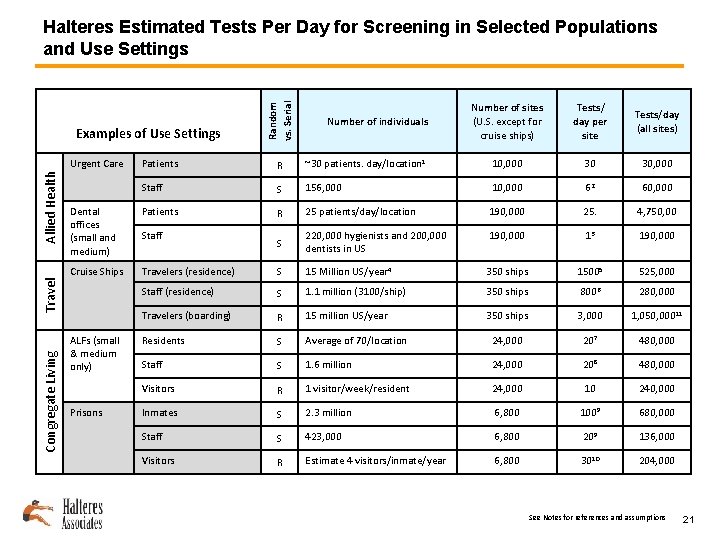
Examples of Use Settings Congregate Living Number of individuals Number of sites (U. S. except for cruise ships) Tests/ day per site Tests/day (all sites) Patients R ~30 patients. day/location 1 10, 000 30 30, 000 Staff S 156, 000 10, 000 62 60, 000 Dental offices (small and medium) Patients R 25 patients/day/location 190, 000 25. 4, 750, 00 S 220, 000 hygienists and 200, 000 dentists in US 190, 000 13 190, 000 Cruise Ships Travelers (residence) S 15 Million US/year 4 350 ships 15005 525, 000 Staff (residence) S 1. 1 million (3100/ship) 350 ships 8006 280, 000 Travelers (boarding) R 15 million US/year 350 ships 3, 000 1, 050, 000 11 Residents S Average of 70/location 24, 000 207 480, 000 Staff S 1. 6 million 24, 000 208 480, 000 Visitors R 1 visitor/week/resident 24, 000 10 240, 000 Inmates S 2. 3 million 6, 800 1009 680, 000 Staff S 423, 000 6, 800 209 136, 000 Visitors R Estimate 4 visitors/inmate/year 6, 800 3010 204, 000 Travel Allied Health Urgent Care Random vs. Serial Halteres Estimated Tests Per Day for Screening in Selected Populations and Use Settings ALFs (small & medium only) Prisons Staff See Notes for references and assumptions 21

What Happened to Diagnostic Tests in Previous Pandemics? q In the pandemic (H 1 N 1) 2009, there were 16 influenza tests given an EUA by the US FDA l l q Similarly, nearly all EUA-authorized Ebola tests were no longer available two years after the 2014 West African epidemic l l q By 2010 there were only 4 remaining on the market The EUAs were eliminated and the more stringent FDA 510(k) path to clearance was invoked Almost none of the immunoassays remained on the market due to poor sensitivity One new test approved in 2019 from Ora. Sure In the current SARS-Co. V-2 pandemic, initially in the US a large number of immunoassay tests were given EUA status, but since then more than 150 of the products have been removed from the market due to poor performance. l This was due to the effort by the FDA and NCI to develop validation panels Existing RNA tests have and will likely continue to be reclassified q FDA/NIST standard used to test and compare many RNA tests q l l q Abbott IDNow EUA changed from broad use claim to “first 5 days after symptoms” Others likely to be changed as well What happens when the EUAs for SARS-Co. V-2 go away? © 2020 Halteres Associates LLC

Many Tests Have EUA or CE-IVD, but The Rules Are Changing https: //chs. asu. edu/diagnostics-commons/testing-commons © 2020 Halteres Associates LLC

What happens when the EUAs for SARS-Co. V-2 go away? Manufacturers will likely be given a period of time to resubmit a full 510(k) filing q Performance requirements could become more difficult q l l l q Typical processes and documentation for IVD tests will be required l q Iso certification, GMP, QMS, design controlled product development, verification, validation, etc. Companies with previous 510(k) cleared/approved tests will be prepared l l q Clinical sensitivity and specificity improvement (? ) Larger number of clinical samples (? ) Closer alignment of trials and actual use (e. g. , asymptomatic, screening vs diagnosis) (? ) Large multinationals Small companies that planned for this eventuality If companies do not resubmit, their products must be withdrawn from the market © 2020 Halteres Associates LLC

But How Long Will the SARS-Co. V-2 Test Market Last? What ever I say here will be wrong! q Most optimistic view of the path to end the pandemic q l l l Vaccines work as well as for smallpox or polio, last a life time, become available quickly to most of the global population and people seek vaccination And/or very safe and effective antiviral drugs will become available as prophylactics The virus disappears as did H 1 N 1 flu after the 2009 outbreak Most experts interviewed do not seem to believe this scenario q But if true, some tests will still be used q l Diagnostics will be needed for a significant amount of time (RNA and Antigen) – COVID-19 will still be a significant part of potential ILI etiology – Likely to be part of a panel with flu A/B and RSV (differential diagnosis use case) l Surveillance will still be used (Antibody) – How well are vaccination programs doing? (Prevalence and titer changes over time) – Will need updated serology tests based on vaccine types (spike vaccine, nucleocapsid test? ) l Screening and other use cases likely would decrease over time (RNA and Antigen) – When the incidence of COVID-19 dips to non-detectable in locations screening might cease in many places – But all regions will not have done as well and when travel opens there will be new outbreaks – Health checks and surveys could still be important © 2020 Halteres Associates LLC

But How Long Will the SARS-Co. V-2 Test Market Last? q Less optimistic view l l Vaccines work about as well as flu vaccines (~40 -60% effective, decrease severity) Antiviral drugs used mostly in hospitalized persons (remdesivir) Virus is endemic for the foreseeable future with continued and scattered peak outbreaks However, physicians continue to improve their skills at dealing with COVID-19, decreasing mortality This is view is not widely held, but what happens is probably somewhere in between the two q However, if true, many tests will likely continue for a long time q l l l Diagnostics would continue, probably in context of ILI Surveillance would continue to be used Broad screening programs would still be used © 2020 Halteres Associates LLC

What Should Companies Do in This Uncertainty? q Realistically assess the value of the SARS-Co. V-2 offering relative to current and future competition l l q The US government isn’t going to subsidize the market forever l l l q Already reimbursement changed from $100 to $75 for RNA tests if over 2 day TTR Perkin-Elmer lab has quoted RNA tests for $30 (down from usual $150) Find a way to get paid for the mid- to long term Think of this much more like flu testing l l q Faster, cheaper, fewer false negatives, fewer false positives? Higher throughput, easier to collect samples, home tests? Unpredictable market in waves of infection (but not necessarily seasonal) Be sure to have other product options (e. g. , differential diagnostic with other influenza like disease pathogens, expand menu on same platform) Take advantage of the opportunity quickly l l l Hone the value proposition Find the funds to scale, if possible If scaling is a problem, find a niche that the offering is well suited to Recognize that the US is not the only market q Have a backup plan q © 2020 Halteres Associates LLC

Thanks for listening © 2020 Halteres Associates LLC
- Slides: 28