A Brief History of Chemistry Ancient Greece BCE

A Brief History of Chemistry • • • Ancient Greece ~_____ BCE ______ Atomos-_____ Thought matter was discreet

A Brief History of Chemistry • Aristotle • Thought matter was ________ • Credited with 4 Greek elements • Earth • Air • Fire • Water

A Brief History of Chemistry • The Middle Ages • ________ • Europe-Convert base metals to ____ • Asia- ______

A Brief History of Chemistry • Robert Boyle • The Skeptical Chemist • The beginnings of chemistry as an _________________

A Brief History of Chemistry • Antoine Lavoisier • The father of modern chemistry • Law of _______ of Mass • Helped develop the __________ • Made first comprehensive list of ________

A Brief History of Chemistry • John Dalton • _________ • 1803 -1807

A Brief History of Chemistry • 1867 • Dmitri Mendeleev • _________

A Brief History of Chemistry • 1897 • J. J. Thomson • Discovers _______

A Brief History of Chemistry • • 1911 Ernest Rutherford Gold Foil Experiment Discovered _________ • Discovered _____ in 1919 • James Chadwick discovered the ________ in 1932

A Brief History of Chemistry • The Manhattan Project • 1940’s • The Atomic Bomb

The Law of Conservation of Mass • Matter is neither _____ nor _____ during chemical reactions or during physical changes.
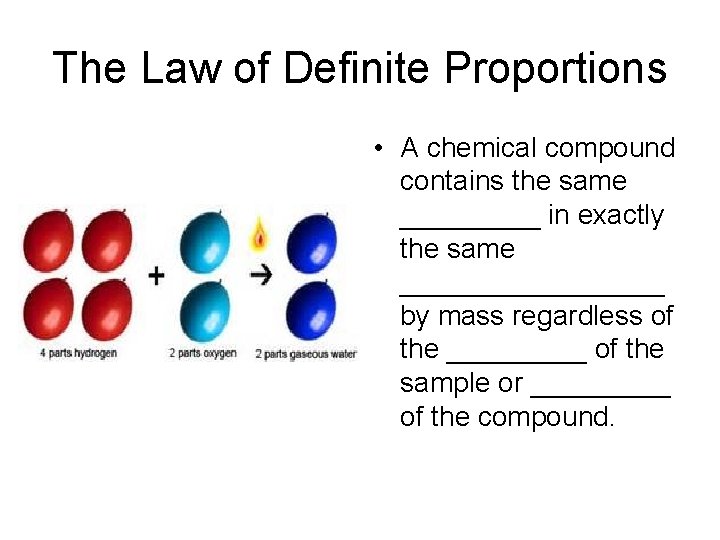
The Law of Definite Proportions • A chemical compound contains the same _____ in exactly the same _________ by mass regardless of the _____ of the sample or _____ of the compound.

The Law of multiple Proportions • If two or more different compounds are composed of the _________, then the ratio of the masses of the second element combined with a certain mass of the first element is always a ratio of _________.

Dalton’s Atomic Theory • All matter is made up of ________ called atoms. • Atoms of a given element are identical in ______, and other properties. • Atoms of different elements _______ in size, mass, and other properties.

Dalton’s Atomic Theory • Atoms cannot be _________, or _____. • Atoms of different elements can combine in simple, _________ to form chemical compounds.

Dalton’s Atomic Theory • In chemical reactions (rxns), atoms are _________, or _____.

Modern Atomic Theory • All matter is composed of _________particles called atoms. • Atoms of the same element have the same chemical properties, while atoms of different elements have different _________.

Modern Atomic Theory • While individual atoms of a given element may not all have the same _____ (due to differences in nuclear structure), any natural sample of the element will have a definite _________that is characteristic of that element as compared to any other element.

Modern Atomic Theory • _____ are formed when atoms of 2 or more elements unite, with each atom losing its characteristic _____ as a result of this combination. • Atoms are not _____ in physical or chemical rxns.

Discovery of the Electron • 1897 • JJ Thomson • Cathode Ray Tube Experiment • Discovered the electron

Discovery of the Electron • Air was evacuated from the tube and an electric current was passed through it. • A _____was seen. • When Thomson held a magnet to the glow, it was _________ the magnet.
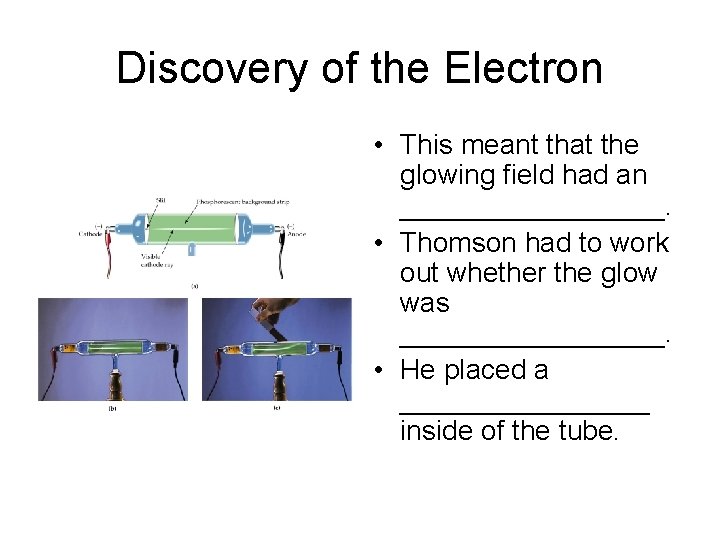
Discovery of the Electron • This meant that the glowing field had an _________. • Thomson had to work out whether the glow was _________. • He placed a ________ inside of the tube.

Discovery of the Electron • If the wheel moved, the glow was _________. • If the wheel did not move, the glow was _____. • The wheel moved, so Thomson knew that __________were produced by the current.
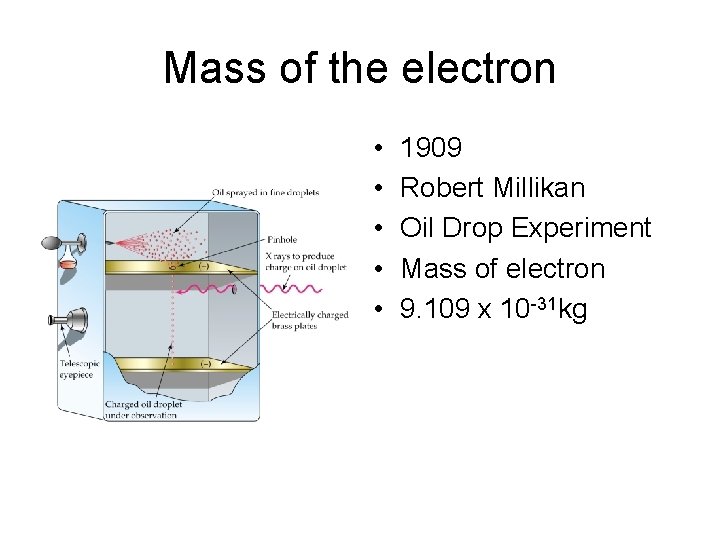
Mass of the electron • • • 1909 Robert Millikan Oil Drop Experiment Mass of electron 9. 109 x 10 -31 kg

Plum Pudding Model

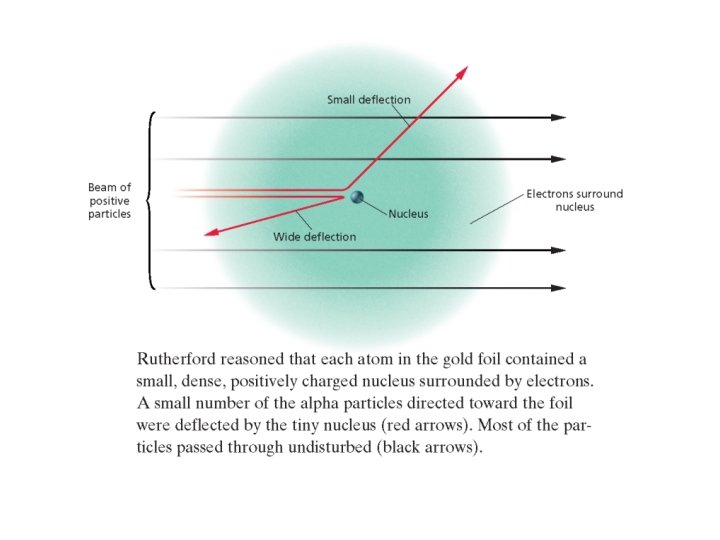
Rutherford’s Gold Foil Experiment • • 1911 Ernest Rutherford Gold Foil Experiment Discovered the atomic nucleus

The atom • An atom is the smallest particle of an element that retains the ________ of that element. • The ______ is a very small region located at the center of an atom. • The nucleus is made up of at least one positively charged particle called a ______ and usually one or more neutral particles called ______.

The atom • Surrounding the nucleus is a region occupied by negatively charged particles called _______. • Protons, neutrons, and electrons are often referred to as ____________.
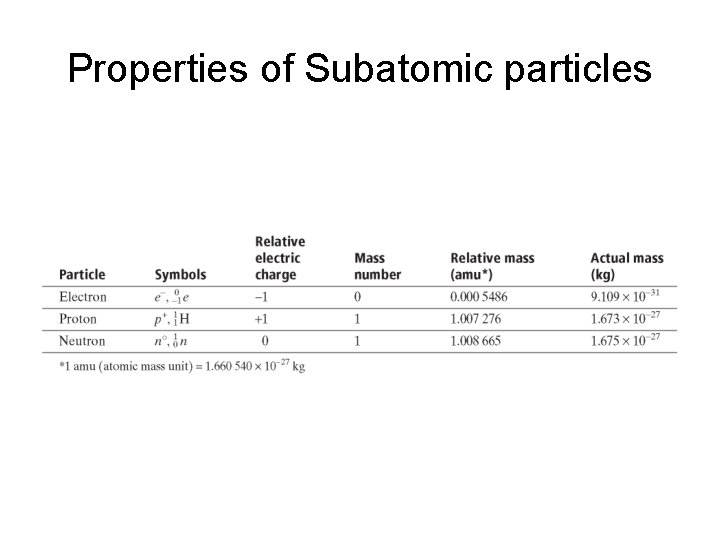
Properties of Subatomic particles

Forces in the Nucleus • When two protons are extremely close to each other, there is a ________ between them. • A similar attraction exists when _______ are very close to each other or when protons and neutrons are very close together. • The short-range proton-neutron, proton, and neutron-neutron forces that hold the nuclear particles together are referred to as _________.
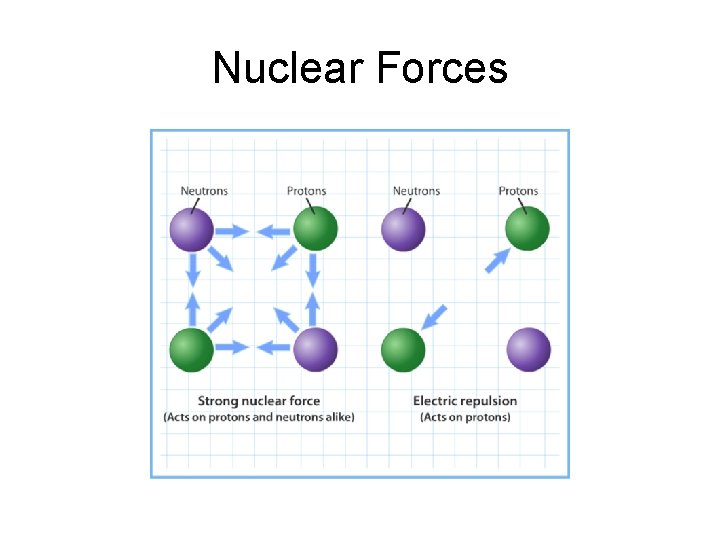
Nuclear Forces

Atomic Number • Atoms of different elements have different numbers of _____. • Atoms of the same element all have the _______ number of protons. • The atomic number (Z) of an element is the number of _____ of each atom of that element.
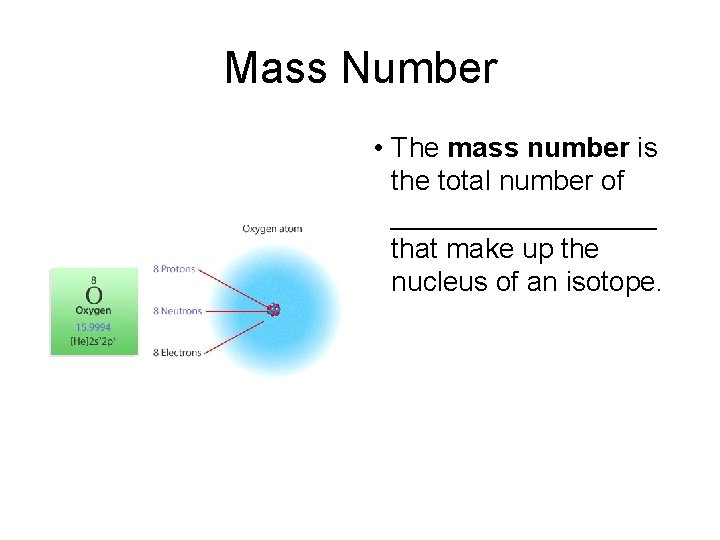
Mass Number • The mass number is the total number of _________ that make up the nucleus of an isotope.

Isotopes • Isotopes are atoms of the same element that have different _____. • The isotopes of a particular element all have the ____ of protons and electrons but _____ numbers of neutrons. • Most of the elements consist of ____ of isotopes.

Relative Atomic Mass • The standard used by scientists to compare units of atomic mass is the _______ atom, which has been arbitrarily assigned a mass of exactly 12 atomic mass units, or 12 amu. • One _________, or 1 _____, is exactly 1/12 the mass of a carbon-12 atom. • The atomic mass of any atom is determined by comparing it with the mass of the ______ atom.

Average Atomic Mass • Average atomic mass is the weighted average of the atomic masses of the naturally occurring _____ of an element. • Calculating Average Atomic Mass • The average atomic mass of an element depends on both the _______ and the __________of each of the element’s isotopes.

The mole • A mole (abbreviated mol) is the amount of a substance that contains as many particles as there atoms in exactly _______ of carbon-12. • The mole is the SI unit for amount of ______. • Avogadro’s number— 6. 022 1415 × 1023—is the number of particles in ______ of a pure substance.

Molar Mass • The mass of _____ of a pure substance is called the molar mass of that substance. • Molar mass is usually written in units of _______. • The molar mass of an element is numerically equal to the atomic mass of the element in _________.

Molar Conversions • What is the mass in grams of 3. 50 mol of the element copper, Cu?

Molar Conversions • How many moles of silver, Ag, are in 3. 01 1023 atoms of silver?
- Slides: 41