a beta gamma alpha and RADIATION Marie Curie

a beta gamma alpha α, and RADIATION Marie Curie Antoine-Henri Becquerel (1852 – 1908) © JP 1

1896: Becquerel accidentally discovered that uranyl crystals emitted invisible radiation which hit even an enclosed photographic plate uranyl salt © JP Becquerel’s Notes Photographic film 2

90 thorium Marie Curie discovered that thorium, Z=90, was a radioactive element 1867 -1934 1898: Marie and Pierre Curie discovered polonium, Z=84 and radium, Z = 88, two new radioactive elements. 84 polonium © JP radium 88 as paint 3
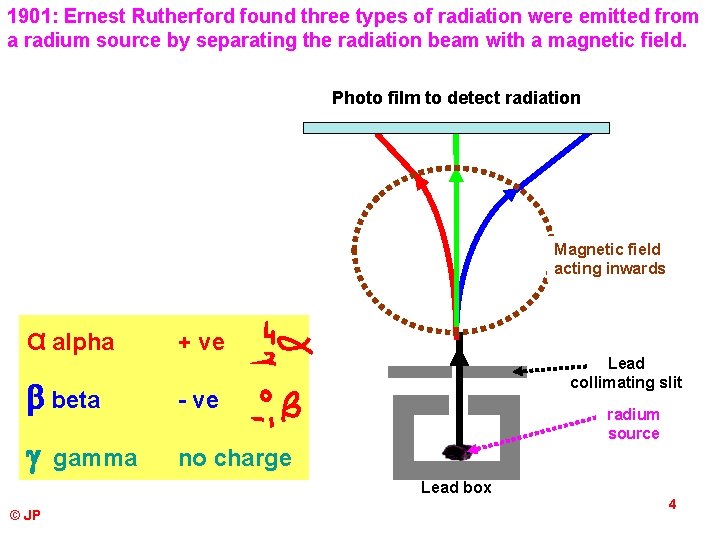
1901: Ernest Rutherford found three types of radiation were emitted from a radium source by separating the radiation beam with a magnetic field. Photo film to detect radiation Magnetic field acting inwards α alpha + ve beta - ve no charge gamma Lead collimating slit radium source Lead box © JP 4

Alpha particles are helium nuclei. Typical speed 0. 1 c. They have a 2+ charge. Beta particles come from the nucleus and are high speed electrons. How do electrons come from the nucleus? Typical speed 0. 9 c They have a 1 - charge. Gamma rays are high frequency, high energy electromagnetic radiation. speed = c. They have no charge. © JP 5

Radioactive atoms have: • • • too many neutrons or too many protons or are just too big An unstable NUCLEUS can emit radioactive particles in order to reach stability: – Beta Particle Production – Alpha Particle Production – Gamma Ray Emission © JP 6

ALPHA DECAY + ? energy e. g. © JP 7

A neutron in the nucleus turns into a proton, an electron, an antineutrino and energy. The nucleus now has one more proton than it started with. BETA DECAY + + + During a beta + decay, a proton in an atom's nucleus turns into a neutron, a positron, a neutrino and energy. © JP 8
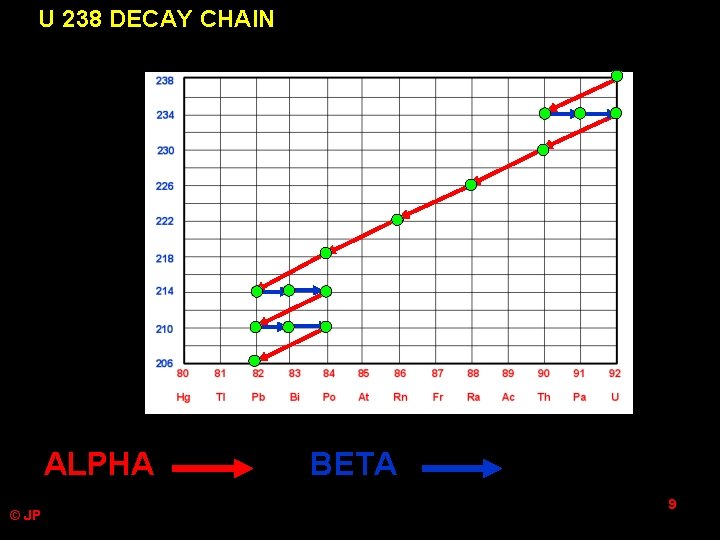
U 238 DECAY CHAIN ALPHA © JP BETA 9
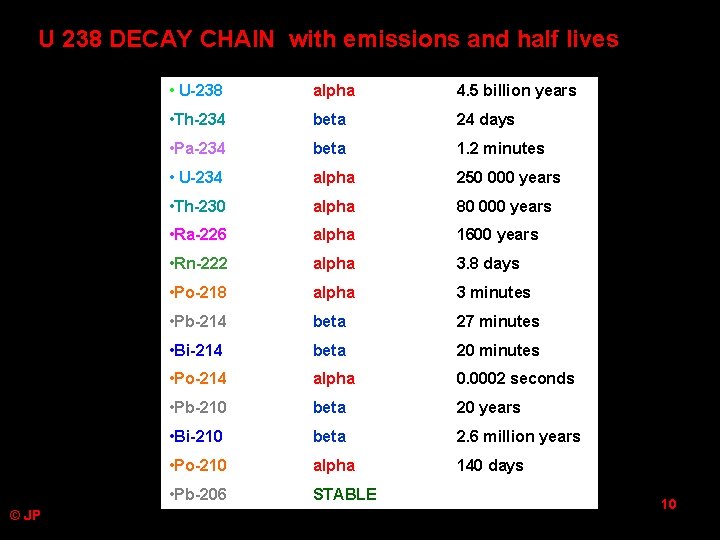
U 238 DECAY CHAIN with emissions and half lives © JP • U-238 alpha 4. 5 billion years • Th-234 beta 24 days • Pa-234 beta 1. 2 minutes • U-234 alpha 250 000 years • Th-230 alpha 80 000 years • Ra-226 alpha 1600 years • Rn-222 alpha 3. 8 days • Po-218 alpha 3 minutes • Pb-214 beta 27 minutes • Bi-214 beta 20 minutes • Po-214 alpha 0. 0002 seconds • Pb-210 beta 20 years • Bi-210 beta 2. 6 million years • Po-210 alpha 140 days • Pb-206 STABLE 10
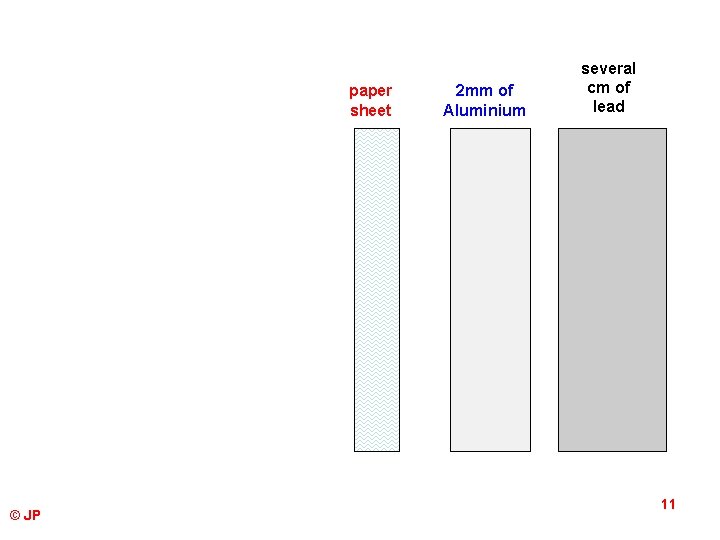
paper sheet © JP 2 mm of Aluminium several cm of lead 11

RANGE OF PARTICLES IN AIR ALPHA RADIATION BETA RADIATION GAMMA RADIATION © JP Travels a few centimetres in air because they pick up electrons as they pass through air becoming helium atoms. This makes ions from the air particles (ionisation). They are stopped by a sheet of paper and are no danger to humans unless inhaled or ingested – then cancer or genetic defects. Because they travel faster, they do not pick up electrons (ionise the air) as much as alpha particles and so travel further. They travel about 1 m in air, 1 cm into body tissue and are stopped be a sheet of metal a few millimetres thick. Not dangerous unless the source is inside the body. Has an unlimited range in air. It is always given off by a radioactive substance and is very harmful – kills living tissue (cancer). It will penetrate 30 cm into steel or several centimetres into lead. 12

Bubble Chambers • Because the decay products have different charges and speeds, their paths in magnetic fields vary. • This means we can tell them apart. © JP 13
- Slides: 13