9172018 DO NOW Write question answer in notebook

9/17/2018 DO NOW • (Write question & answer in notebook) • How many electrons can go into: • The first energy level? • The 2 nd energy level? • The 3 rd energy level? th • The 4 energy level?

The Atom Protons and electrons are attracted to each other because of opposite charges Define electrostatics the branch of physics dealing with electric phenomena not associated with electricity in motion. Electrostatic Basics:

Electrostatic Basics: - - repel - + attract + + repel


Bohr Model of Atom

Bohr Model of Atom • Planetary Model of atom – put electrons into fixed paths around nucleus called orbits • Assigned electrons to fixed energy levels • Moving from one energy level to another = requires a fixed quantum of energy • Quantum Leap – jump from one step to another
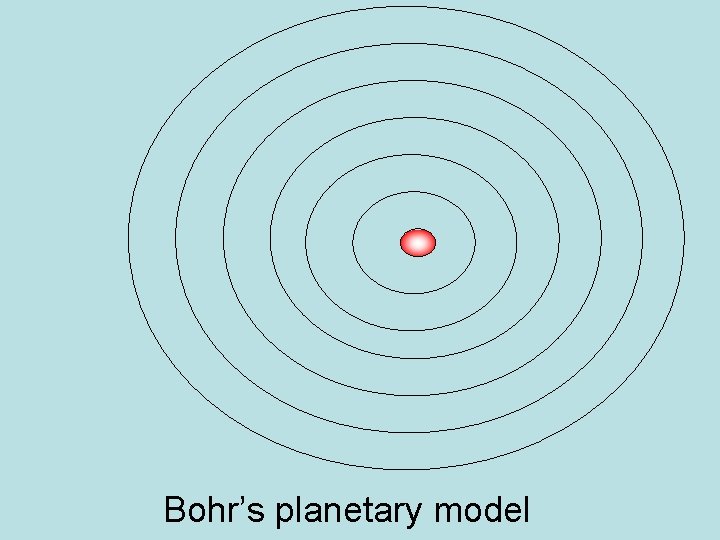
Bohr’s planetary model

The Wave-like Electron The electron propagates through space as an energy wave. To understand the atom, one must understand the behavior of electromagnetic waves. Louis de. Broglie

The Nature of Light

Light • The study of light led to the development of the quantum mechanical model. • Light is a kind of electromagnetic radiation. • Electromagnetic radiation includes many kinds of waves • All move at 2. 998 x 108 m/s (symbol = c) • c = 2. 998 x 108 m/s
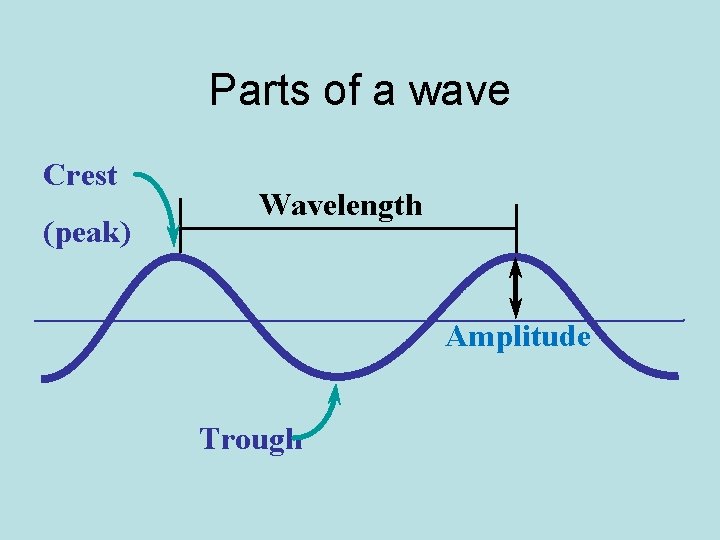
Parts of a wave Crest (peak) Wavelength Amplitude Trough

• • • Parts of Wave Crest - high point on a wave Trough - Low point on a wave Amplitude - distance from origin to crest Wavelength - distance from crest to crest Wavelength - is abbreviated l Greek letter lambda.

Atomic Spectrum How color tells us about atoms

Prism • White light is made up of all the colors of the visible spectrum. • Passing it through a prism separates it.
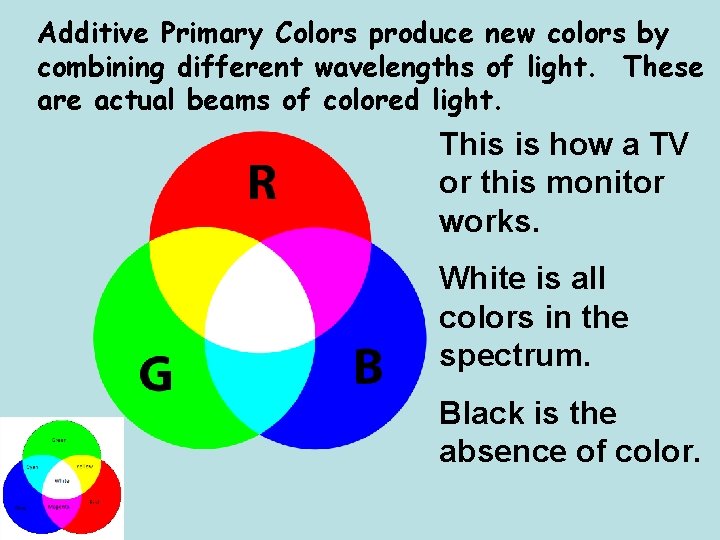
Additive Primary Colors produce new colors by combining different wavelengths of light. These are actual beams of colored light. This is how a TV or this monitor works. White is all colors in the spectrum. Black is the absence of color.

Subtractive Primary Colors are used in printing and art. These colors are not produced from light sources. White: absence of color Black: all colors

If the light is not white • By heating a gas with electricity we can get it to give off colors. • Passing this light through a prism does something different.
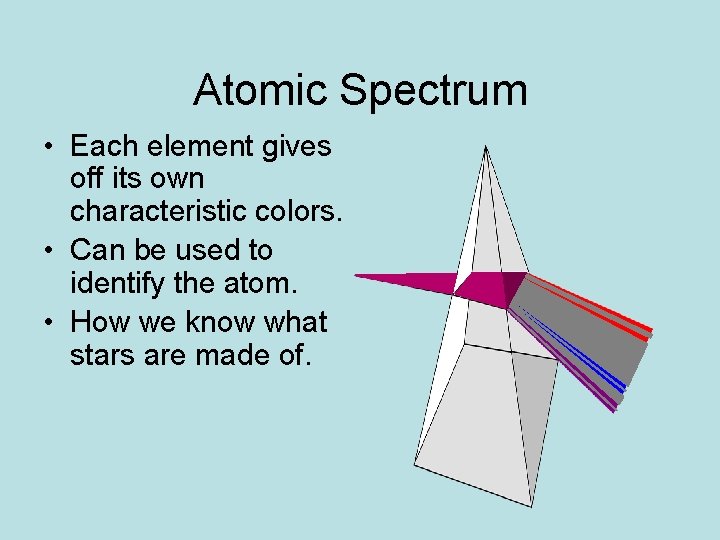
Atomic Spectrum • Each element gives off its own characteristic colors. • Can be used to identify the atom. • How we know what stars are made of.

• These are called the discontinuous spectra (or emission line spectra) and is unique to each element. • Shown here are emission line spectra for 3 different elements. It is what the light being emitted from the elements is really made up of.
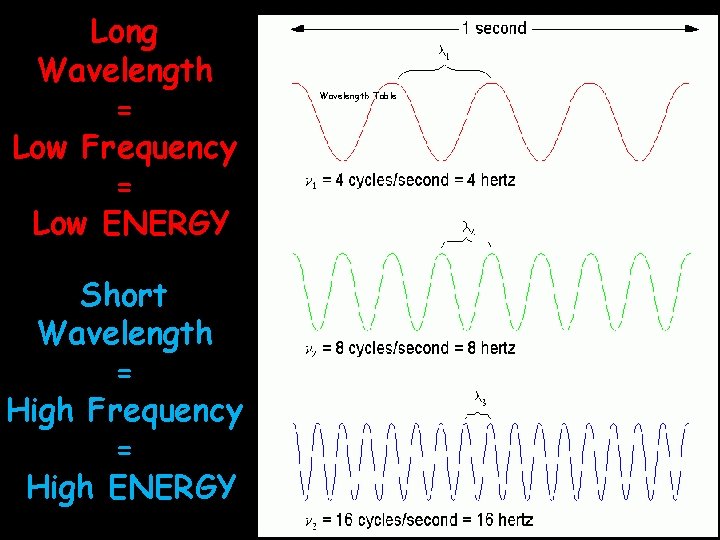
Long Wavelength = Low Frequency = Low ENERGY Short Wavelength = High Frequency = High ENERGY Wavelength Table
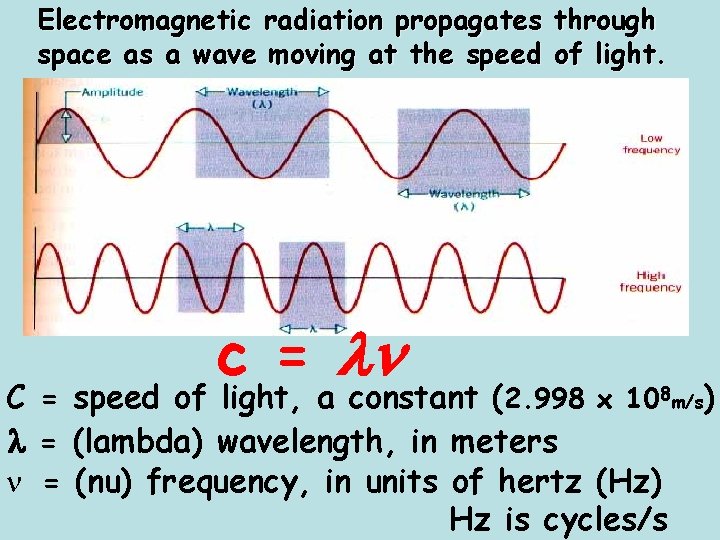
Electromagnetic radiation propagates through space as a wave moving at the speed of light. c = C = speed of light, a constant (2. 998 x 108 m/s) = (lambda) wavelength, in meters n = (nu) frequency, in units of hertz (Hz) Hz is cycles/s
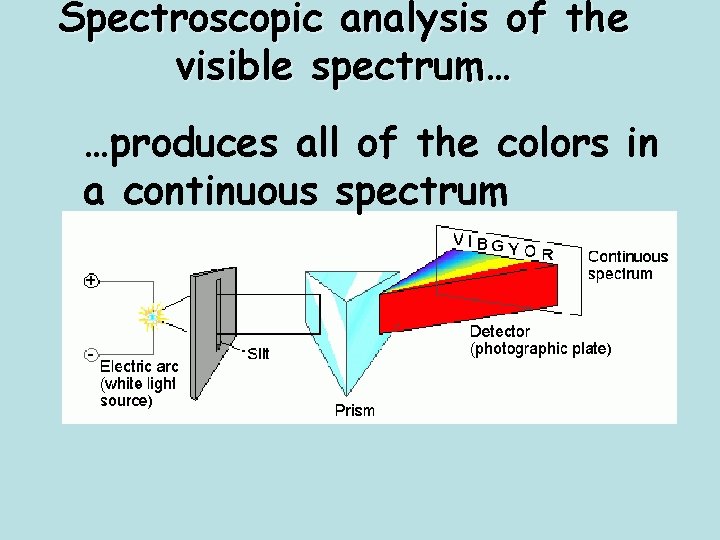
Spectroscopic analysis of the visible spectrum… …produces all of the colors in a continuous spectrum
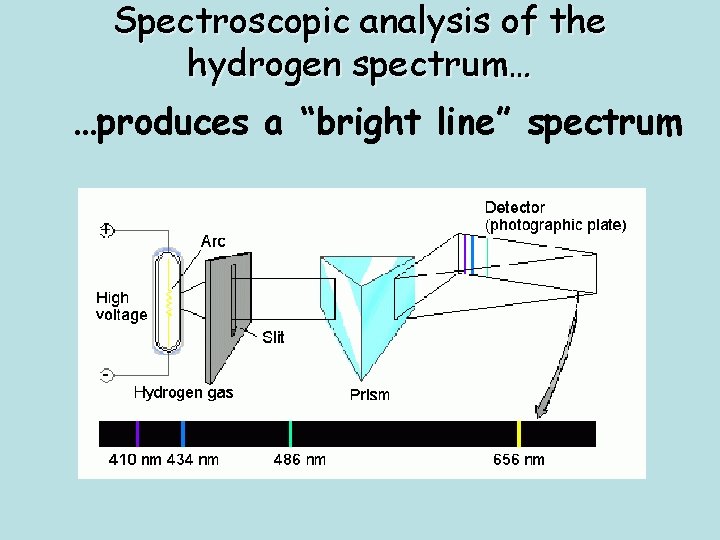
Spectroscopic analysis of the hydrogen spectrum… …produces a “bright line” spectrum
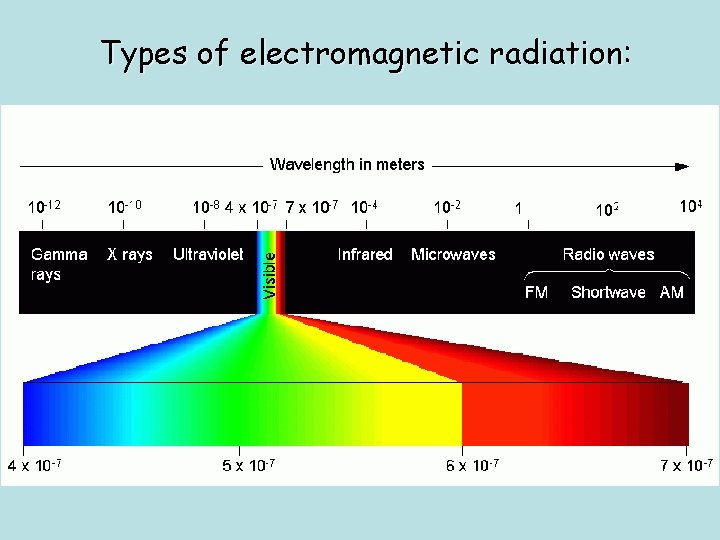
Types of electromagnetic radiation:
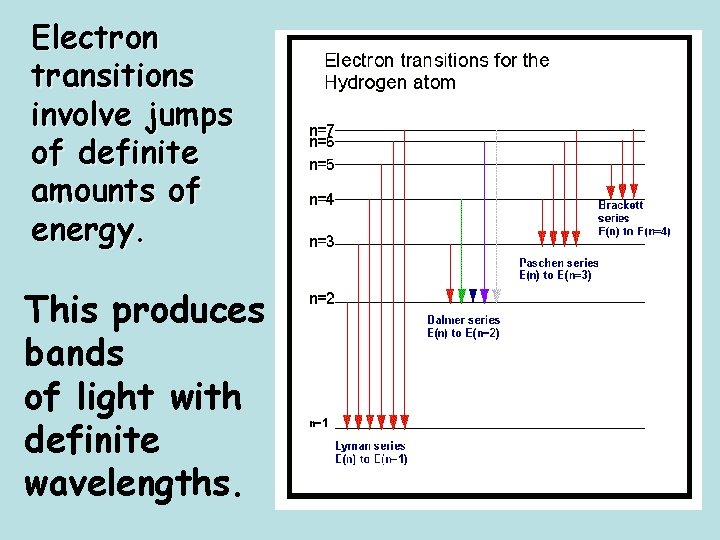
Electron transitions involve jumps of definite amounts of energy. This produces bands of light with definite wavelengths.
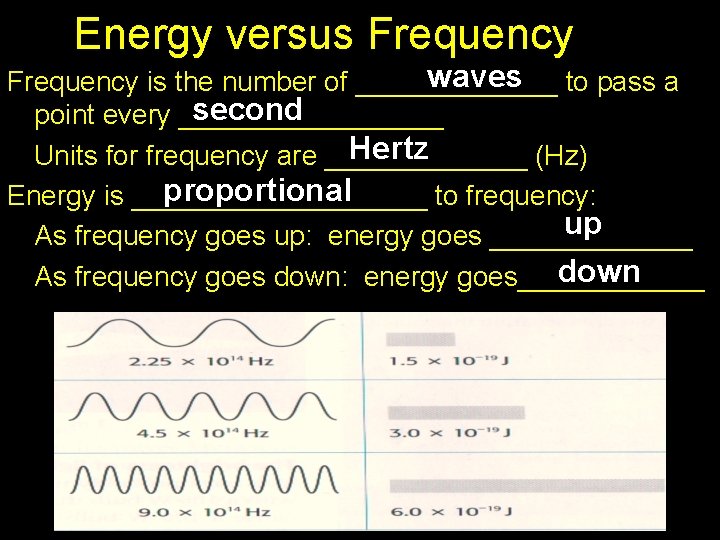
Energy versus Frequency waves to pass a Frequency is the number of _______ second point every _________ Hertz Units for frequency are _______ (Hz) proportional Energy is __________ to frequency: up As frequency goes up: energy goes _______ down As frequency goes down: energy goes______

Is light a wave? Wave – light has a measurable frequency wavelength ________ and ________, just like all other waves energy - Light waves can have _________ - Light is ______ when it hits a mirror, or reflected _________ when it hits a lens Refracted (bent)

Is light made of particles? Particle - certain colors (blue) will cause electrons to fly off of a piece of metal while others (red) don’t Particles of blue light have the right Why? _________________ energy/frequency to knock them free! Photons Particles of light are called _______, mass they have no _______, but are tiny packets of ________ energy The Final Answer: Light BOTH is ______ a wave and a particle!!!!!

Speed of Light 2. 998 x 108 8 6. 70 x 10 All light travels at _______m/s or _____mph medium Speed varies depending on the _______ vacuum Light travels fastest in a ______, which No particles contains ________ Question: What can travel faster than the speed NOTHING!!! of light? _________ intensity Another word for “brightness” is _______, it measures the number of ______ that photons pass a given area

Electromagnetic Spectrum (EMS) Light is also called electromagnetic radiation (EMR) Visible light is only a small part of the EM spectrum The EM Spectrum (EMS) contains radiation of all frequencies possible ______ wavelengths and _______
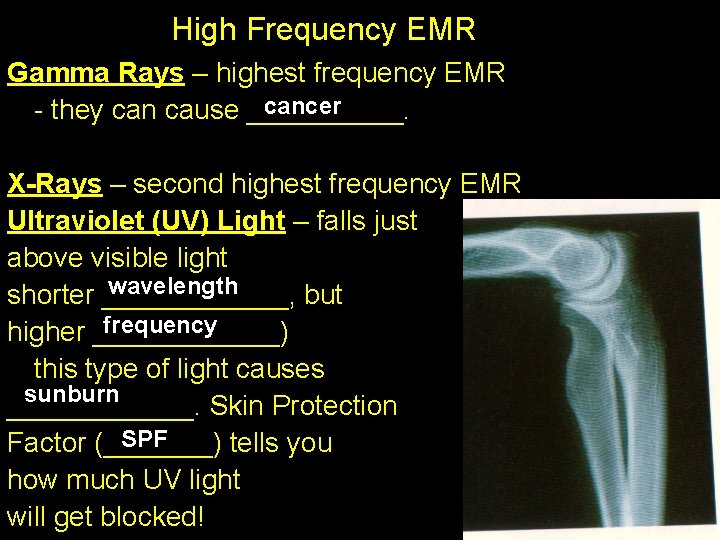
High Frequency EMR Gamma Rays – highest frequency EMR cancer - they can cause _____. X-Rays – second highest frequency EMR Ultraviolet (UV) Light – falls just above visible light wavelength shorter ______, but frequency higher ______) this type of light causes sunburn ______. Skin Protection SPF Factor (_______) tells you how much UV light will get blocked!

Low Frequency EMR Infrared Radiation – has slightly less energy than visible light. Special devices can be used to detect this form of radiation. Microwaves – used to cook food. Waves cause food vibrate molecules to _______, moving molecules kinetic = _______ energy longest Radio Waves - ________ wavelengths and frequency energy smallest _______ = very low _____ RADAR _______ can be used to track airplanes. It bounces waves off the planes and measures the time _______ it takes for them to come back.

Schrodinger Wave Equation Erwin Schrodinger This is an equation for the probability of a single electron being found along a single axis (this one shows x axis)

Quantum Mechanical Model of the Atom Mathematical laws can identify the regions outside of the nucleus where electrons are most likely to be found. These laws are beyond the scope of this class…
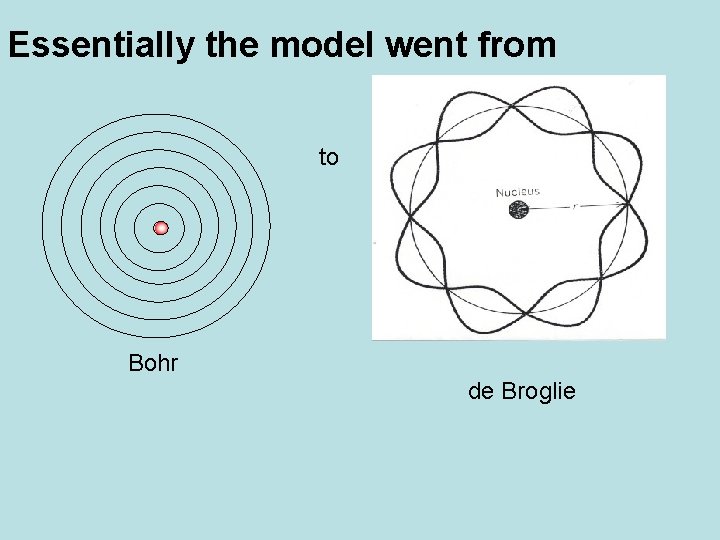
Essentially the model went from to Bohr de Broglie
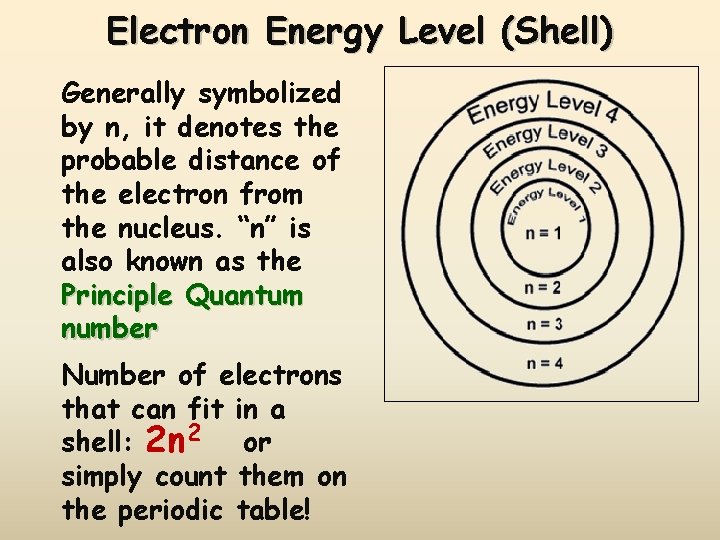
Electron Energy Level (Shell) Generally symbolized by n, it denotes the probable distance of the electron from the nucleus. “n” is also known as the Principle Quantum number Number of electrons that can fit in a shell: 2 n 2 or simply count them on the periodic table!

Energy Sublevels (subshells) A sublevel is a region within an energy level where there is a probability of finding an electron. Orbital shapes are defined as the surface that contains 90% of the total electron probability. Make map now.

Write the electron configurations (no shortcuts!) • • • Lithium (Li) - 3 electrons 2 1 1 s 2 s Oxygen (O) - 8 electrons 2 2 4 1 s 2 s 2 p Neon (Ne) - 10 electrons 2 2 6 1 s 2 s 2 p

Write the electron configurations (no shortcuts!) • • • Magnesium (Mg) – 12 electrons 2 2 6 2 1 s 2 s 2 p 3 s Chlorine (Cl) – 17 electrons 2 2 6 2 5 1 s 2 s 2 p 3 s 3 p Scandium (Sc) – 21 electrons 2 2 6 2 1 1 s 2 s 2 p 3 s 3 p 4 s 3 d

Write the electron configurations (no shortcuts!) • Titanium(Ti) - 22 electrons • Vanadium(V) - 23 electrons

Write the electron configurations (no shortcuts!) • Titanium(Ti) - 22 electrons 2 2 6 2 2 • 1 s 2 s 2 p 3 s 3 p 4 s 3 d • Vanadium(V) - 23 electrons • 1 s 22 p 63 s 23 p 64 s 23 d 3
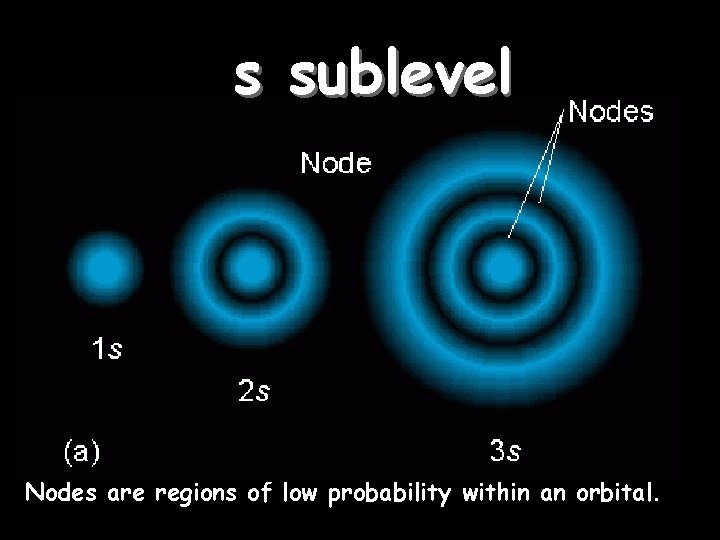
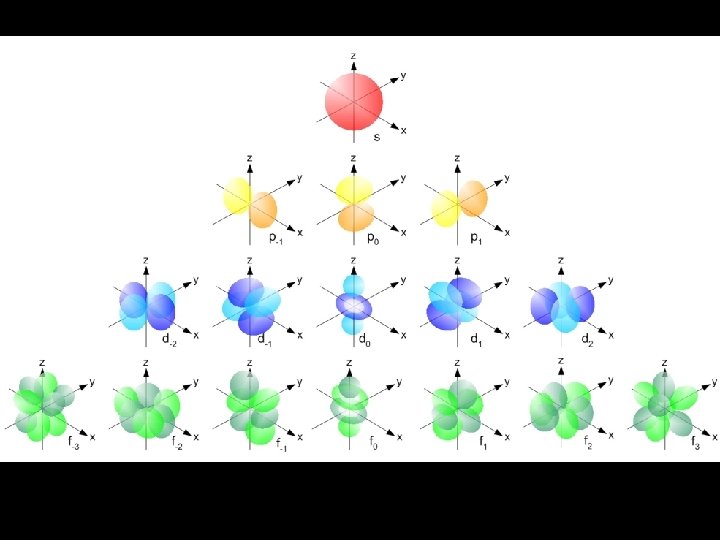
s sublevel Orbitals of the same shape (s, for instance) grow larger as n increases… Nodes are regions of low probability within an orbital.
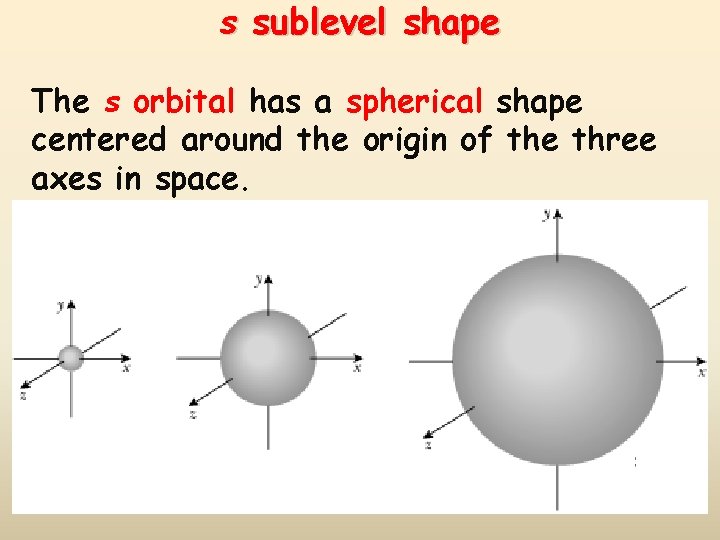
s sublevel shape The s orbital has a spherical shape centered around the origin of the three axes in space.
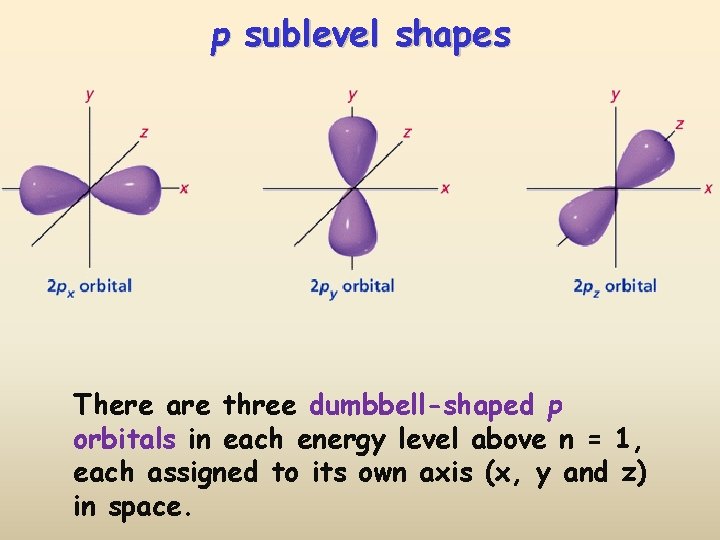
p sublevel shapes There are three dumbbell-shaped p orbitals in each energy level above n = 1, each assigned to its own axis (x, y and z) in space.

d sublevel shapes Things get a bit more complicated with the five d orbitals that are found in the d sublevels beginning with n = 3. To remember the shapes, think of “double dumbbells” …and a “dumbbell with a donut”!
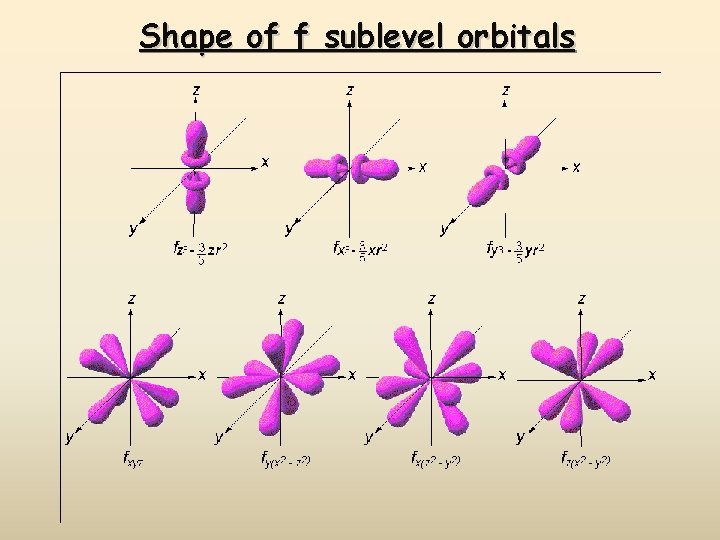
Shape of f sublevel orbitals

DO NOW In your notebook, write the electron configuration for Iron 2 2 6 2 6 1 s 2 s 2 p 3 s 3 p 4 s 3 d
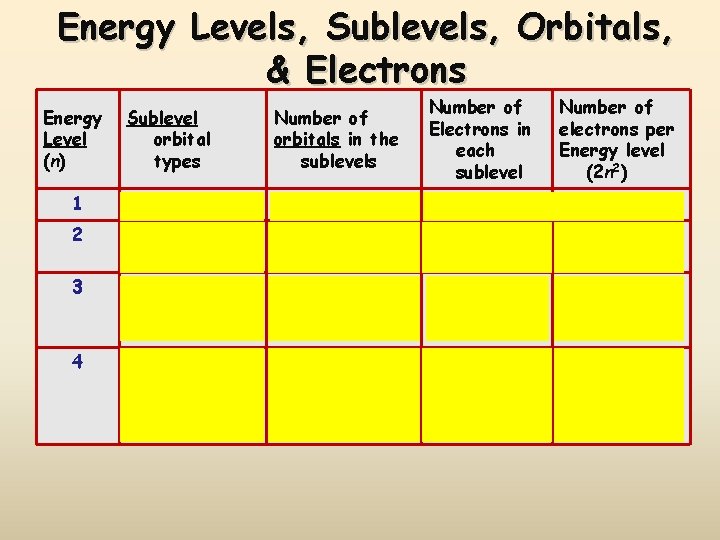
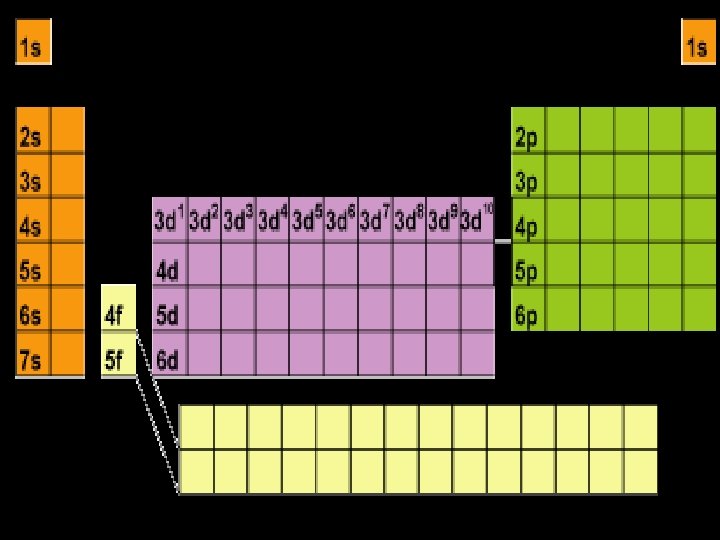
Energy Levels, Sublevels, Orbitals, & Electrons Energy Level (n) Sublevel orbital types Number of orbitals in the sublevels Number of Electrons in each sublevel Number of electrons per Energy level (2 n 2) 1 s 1 2 2 2 s p 1 3 2 6 8 3 s p d 1 3 5 2 6 10 18 4 s p d f 1 3 5 7 2 6 10 14 32

3 Rules for electron configurations: 1) Aufbau Principle – electrons fill lowest energy level orbitals first (lower energy = more stable!) • The word aufbau is German for “building up” • *Note: Atoms always do things to become the most stable!

2) Hund’s Rule – • Electrons will space out evenly. • Want highest number of unpaired electrons with the same spin. • Electrons want the most space between electrons with same energy! • Why? ? ?

Pauli Exclusion Principle Each orbital can hold a MAXIMUM of two electrons! Wolfgang Pauli

Pauli Exclusion Principle Two electrons occupying the same orbital must have opposite spins Wolfgang Pauli

Electron Spin Electron spin describes the behavior (direction of spin) of an electron within a magnetic field. Possibilities for electron spin:

Summarize electron the 3 rules for configuration
![Element Lithium Configuration notation Orbital notation (spin diagram) 1 s 22 s 1 [He]2 Element Lithium Configuration notation Orbital notation (spin diagram) 1 s 22 s 1 [He]2](http://slidetodoc.com/presentation_image_h2/81b21120030e7fcb6cbf33ee832c16d9/image-58.jpg)
Element Lithium Configuration notation Orbital notation (spin diagram) 1 s 22 s 1 [He]2 s 1 ____ 1 s Beryllium ____ 2 p ____ 2 s ____ 2 p ____ [He]2 s 22 p 2 ____ 2 s ____ 2 p ____ 1 s 22 p 3 [He]2 s 22 p 3 ____ 2 s ____ 2 p ____ 1 s 22 p 4 [He]2 s 22 p 4 ____ 2 s ____ 2 p ____ 1 s 22 p 5 [He]2 s 22 p 5 ____ 1 s Neon ____ 2 s 1 s 22 p 2 ____ 1 s Fluorine ____ [He]2 s 22 p 1 ____ 1 s Oxygen ____ 2 p 1 s 22 p 1 ____ 1 s Nitrogen ____ [He]2 s 2 ____ 1 s Carbon ____ 2 s 1 s 22 s 2 ____ 1 s Boron Noble gas notation ____ 2 s ____ 2 p ____ 1 s 22 p 6 [He]2 s 22 p 6 ____ 1 s ____ 2 s ____ 2 p ____

Nuclear Chemistry Bravo – 15, 000 kilotons

Nuclear Chemistry • Title a new section in your notebook called Nuclear Chemistry • Fold, and paste the guided notes into your notebook. • Also, get a periodic table in front of you.

Write these 3 symbols (big!) on the outside of the guided notes packet (space them out a bit) Alpha Beta Gamma

Radioactive Decay • Radioactive decay is the spontaneous breakdown of an unstable atomic nucleus. It releases alpha, beta, and/or gamma radiation. • Very large atoms (atomic # 84 and up) are radioactive. Find these now on your periodic table. • Smaller atoms with an unstable number of protons and neutrons can be radioactive too. • Atoms will be unstable if they do not have the “right” ratio of protons : neutrons.

Draw a nucleus of 8 He
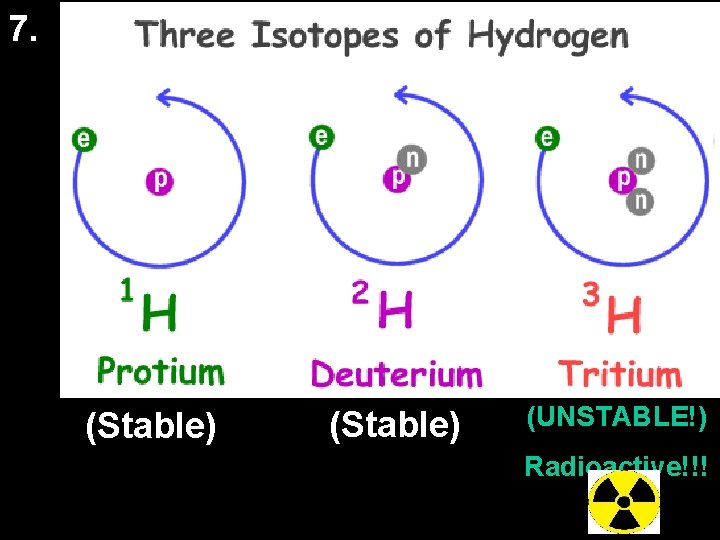
7. (Stable) (UNSTABLE!) Radioactive!!!

• Strong Nuclear Force is an attractive force that holds heavy particles together in the nucleus, overcoming the electrostatic repulsion from positively charged protons. • This strong force keeps the nucleus from falling apart. • Only works at very close distances!!

Marie Curie • Isolated the radioactive elements Po, Ra and U • Coined the term “radioactivity” • Both Marie and her daughter Irene died of leukemia as a result of prolonged exposure to

• Radiation is the penetrating rays and particles emitted by a radioactive source. • Radioactivity is the process by which materials give off such rays.
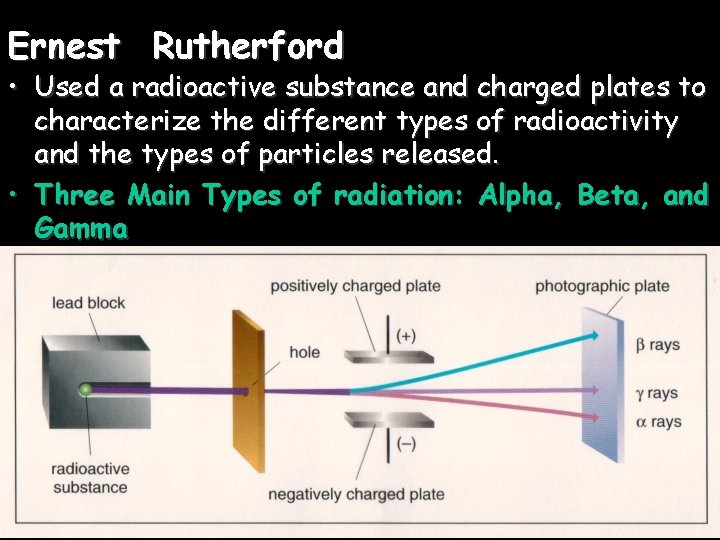
Ernest Rutherford • Used a radioactive substance and charged plates to characterize the different types of radioactivity and the types of particles released. • Three Main Types of radiation: Alpha, Beta, and Gamma
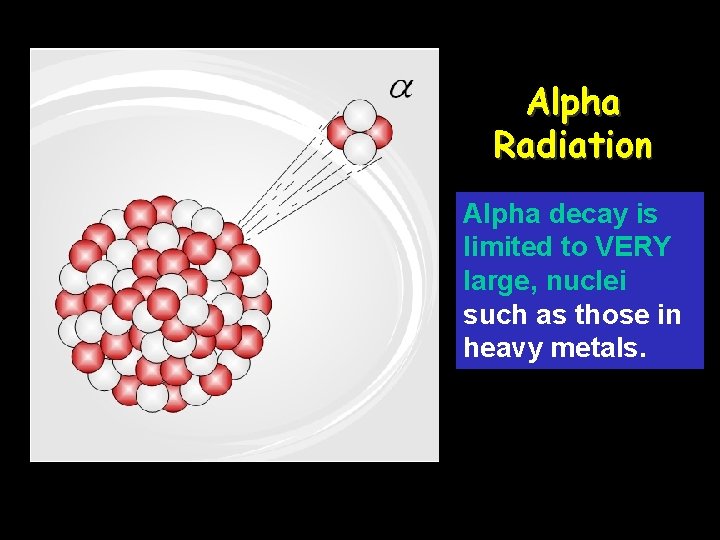
Alpha Radiation Alpha decay is limited to VERY large, nuclei such as those in heavy metals.
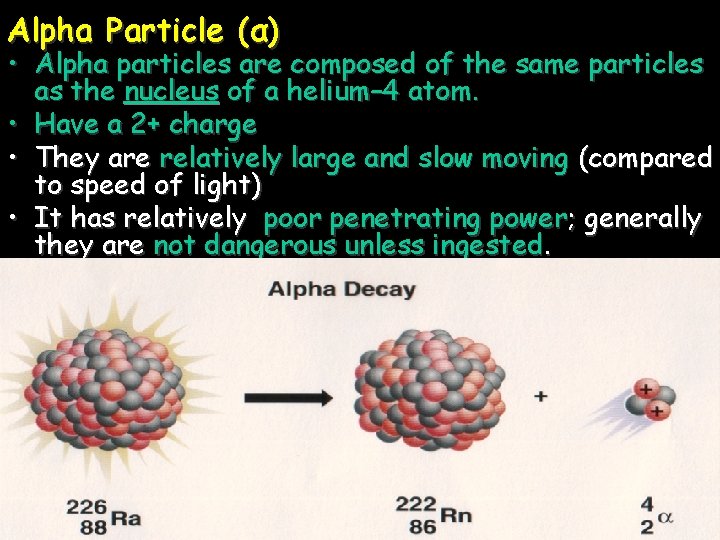
Alpha Particle (α) • Alpha particles are composed of the same particles as the nucleus of a helium– 4 atom. • Have a 2+ charge • They are relatively large and slow moving (compared to speed of light) • It has relatively poor penetrating power; generally they are not dangerous unless ingested.
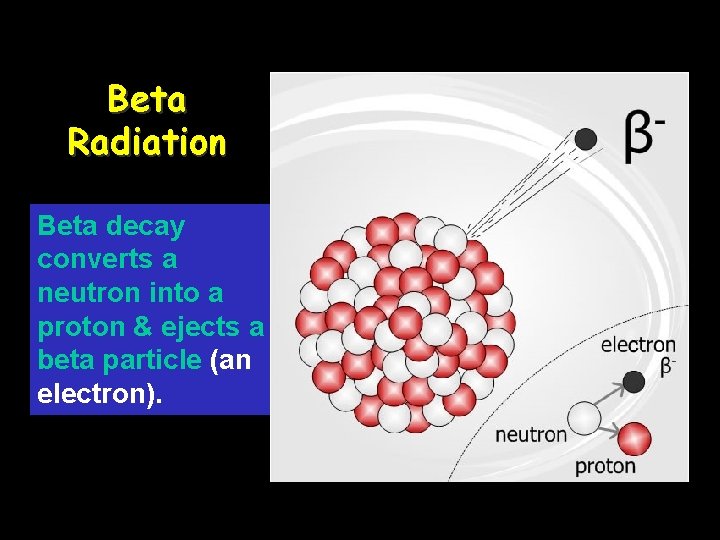
Beta Radiation Beta decay converts a neutron into a proton & ejects a beta particle (an electron).

Beta particle (β): Beta particles are essentially the same as an electron, they have very little mass and a charge of -1. Beta particles are created when a neutron in the nucleus ejects an electron and becomes a proton. Because of their low mass and moderate speed, they have a moderate level of penetration and damage.

• • Gamma Radiation (γ): On the electromagnetic spectrum Has no charge No mass, but travels at the speed of light Very high energy, high frequency radiation, like a super-powered ray of light or X-ray (just with much more energy) Excellent penetration (lead/concrete suit!) Very dangerous and damaging (has the most energy) Gamma radiation is a byproduct of both beta and alpha decay
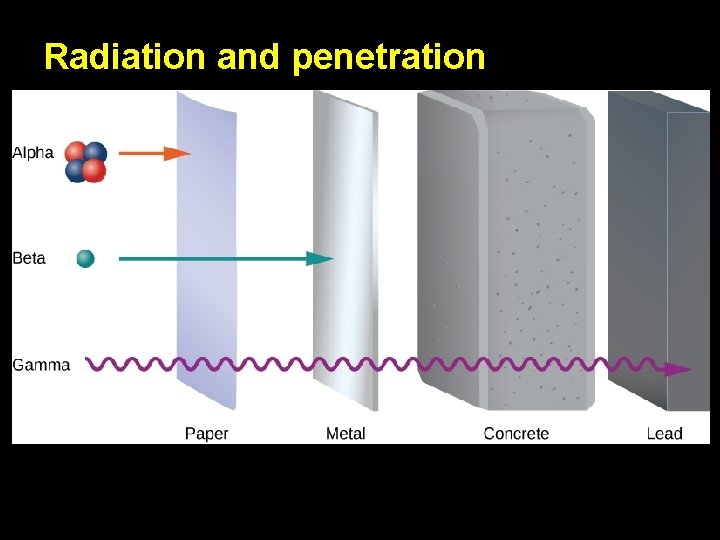
Radiation and penetration
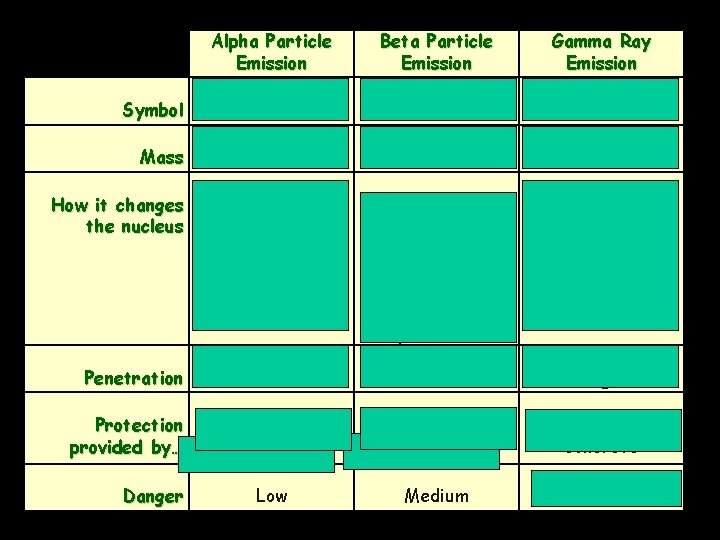
Symbol Mass How it changes the nucleus Penetration Protection provided by… Danger Alpha Particle Emission Beta Particle Emission or or Heavy Light Decreases the mass number by 4 Decreases the atomic number by 2 Converts a neutron into a proton (Mass # doesn’t change) Increases atomic number by 1 Gamma Ray Emission No Mass No change to the nucleus Low Medium High Skin; paper Clothing; wood; thin sheets of metal Lead; super thick concrete Low Medium High

Nuclear reactions are far greater than ordinary chemical reactions • http: //www. youtube. com/watch? v=h 7 vy. KDc. STa. E&fe ature=related • http: //www. youtube. com/watch? v=v. Bdtw 5 Ha. PTQ&fe ature=related

Radioisotopes and Half Life - A radioisotope is an isotope which will undergo radioactive decay over time due to an unstable number of protons and neutrons in its nucleus. (It has too many or too few neutrons. ) -Most elements have at least one naturally occurring radioisotope. There is radioactive decay happening inside of you right now. -Most naturally occurring decay is at such low levels that it is harmless. -Some very unstable nuclei can decay very quickly, others will decay much more slowly.
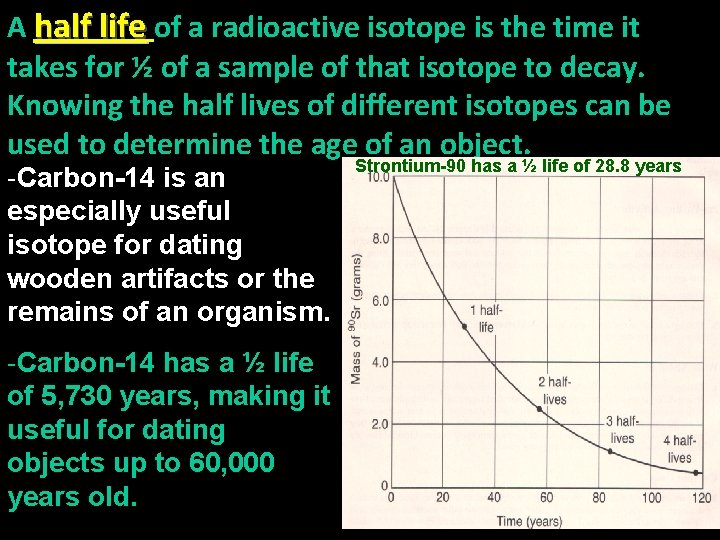
A half life of a radioactive isotope is the time it takes for ½ of a sample of that isotope to decay. Knowing the half lives of different isotopes can be used to determine the age of an object. -Carbon-14 is an especially useful isotope for dating wooden artifacts or the remains of an organism. -Carbon-14 has a ½ life of 5, 730 years, making it useful for dating objects up to 60, 000 years old. Strontium-90 has a ½ life of 28. 8 years

Half-life Practice Sample Question 1: The ½ life of mercury-195 is 31 hours. If you begin with a 5. 00 g sample of 195 Hg, how much of it will be present after 93 hours?
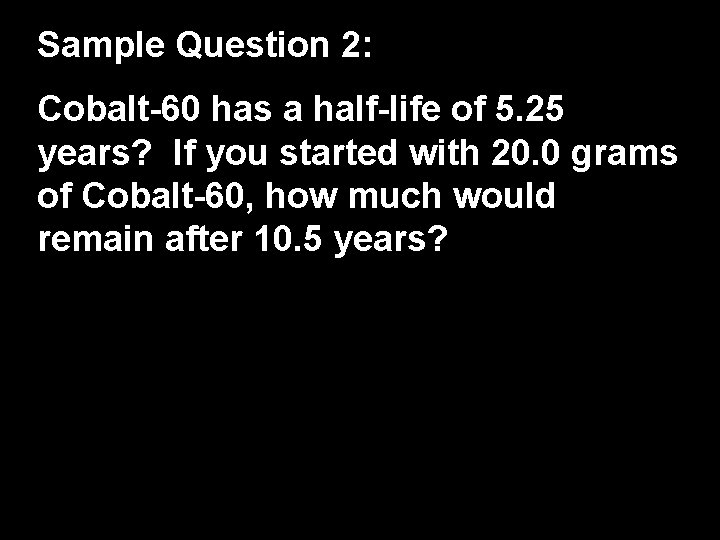
Sample Question 2: Cobalt-60 has a half-life of 5. 25 years? If you started with 20. 0 grams of Cobalt-60, how much would remain after 10. 5 years?

Sample Question 3: A 64. 0 g sample of germanium-66 is left undisturbed for 25 hours. At this time, only 2. 0 g of germanium-66 remained. How many half lives did germanium-66 undergo?

Sample Question 3: A 64. 0 g sample of germanium-66 is left undisturbed for 25 hours. At this time, only 2. 0 g of germanium-66 remained. How many half lives did germanium-66 undergo?

• Where are the transuranic elements on the periodic table? • “trans” prefix means “after” • Uranic looks like what element? • = “after” Uranium • So transactinide elements would be where?

In a nuclear fission reaction, a large nucleus is split into two smaller nuclei of approximately equal mass. -During nuclear fission a very small amount of mass is “lost” as it is converted into an enormous amount of energy!
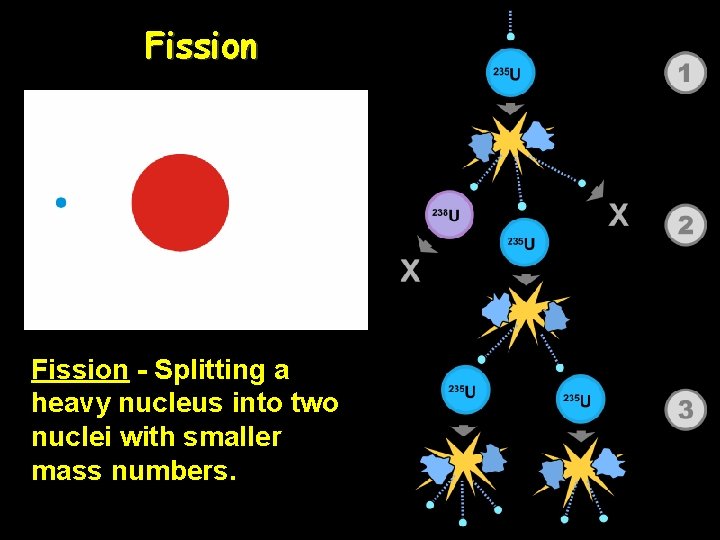
Fission - Splitting a heavy nucleus into two nuclei with smaller mass numbers.

In a chain reaction the neutrons produced at the end of one nuclear fission reaction are used to initiate a successive reaction. This can lead to a geometric expansion of fission reactions. -Atomic bombs are designed to create a “runaway” chain reaction which will use up all of the uranium present and create an incredibly powerful explosion. -Nuclear reactor plants do not contain enough 235 U to sustain a chain reaction. The major “meltdown” concern is the uncontrolled release of the radioactive waste produced during these reactions!

In a nuclear fusion reaction, two lighter nuclei are fused together to form a heavier nucleus. -This reaction also causes a small amount of mass to be “lost” as it is converted into a tremendous amount of energy. -Fusion is difficult to achieve due to the electrostatic repulsion of likecharged electron clouds and then by the repulsion of the like-charged protons in the nucleus. -Stars use fusion reactions as fuel. They overcome the repulsion problem with a combination of tremendous heat and gravitational force to force the nuclei together! -Cold Fusion research is aimed at finding a viable method for using fusion as a pollution free fuel source. It has not yet been successful.
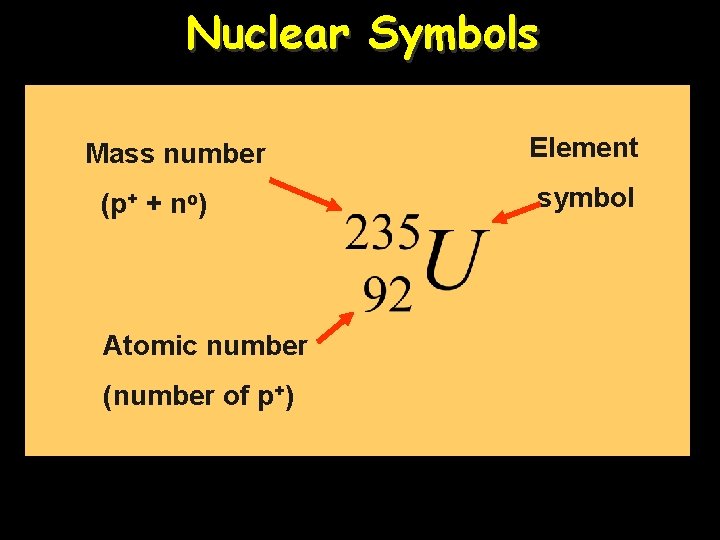
Nuclear Symbols Mass number (p+ + no) Atomic number (number of p+) Element symbol

Types of Radioactive Decay v alpha production (a, He): helium nucleus v beta production (b, e): v gamma ray production (g):

Calculate # of neutrons for elements 1 -84 • Use data table you picked up: • Element | Mass # | #p+ • | #n 0 • Mass : (round to nearest whole number) • Then graph data for elements 1 -84
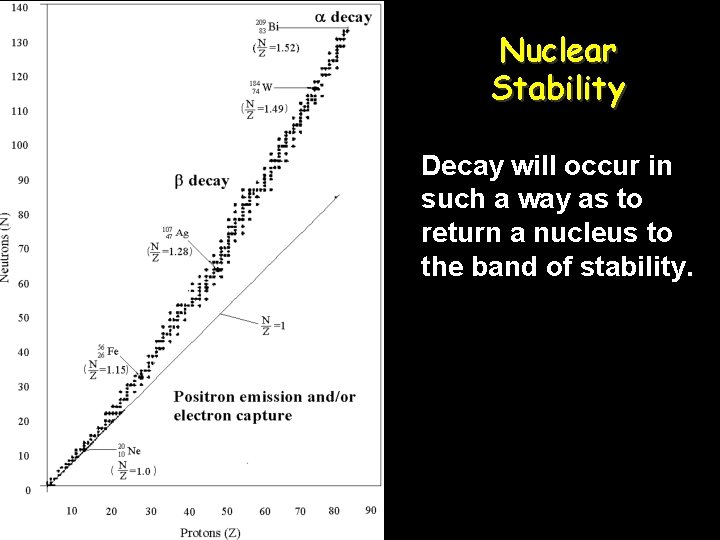
Nuclear Stability Decay will occur in such a way as to return a nucleus to the band of stability.
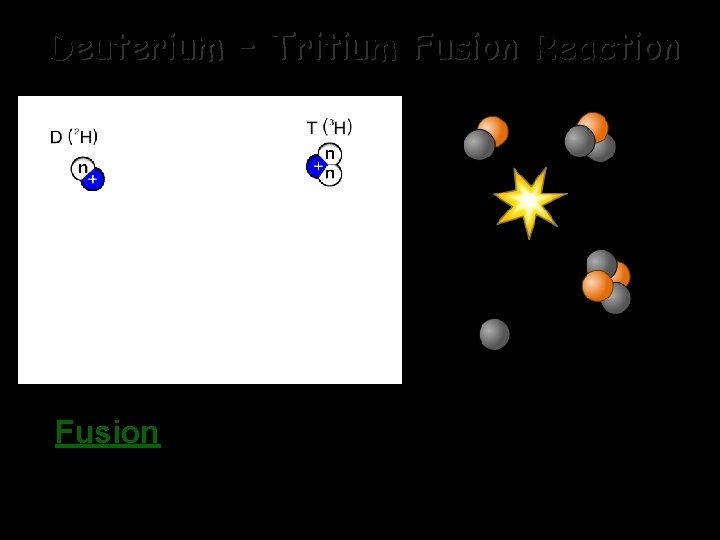
Deuterium – Tritium Fusion Reaction Fusion - Combining two light nuclei to form a heavier, more stable nucleus.

Albert Einstein used the formula: E=mc 2 to describe the possibility for the interchange between energy and mass. This formula shows that an absolutely enormous amount of energy can be derived from a small amount of matter!! E = mc 2 E = energy m = mass defect (mass lost during fission or fusion) c = speed of light (3. 0 x 108 m/s)
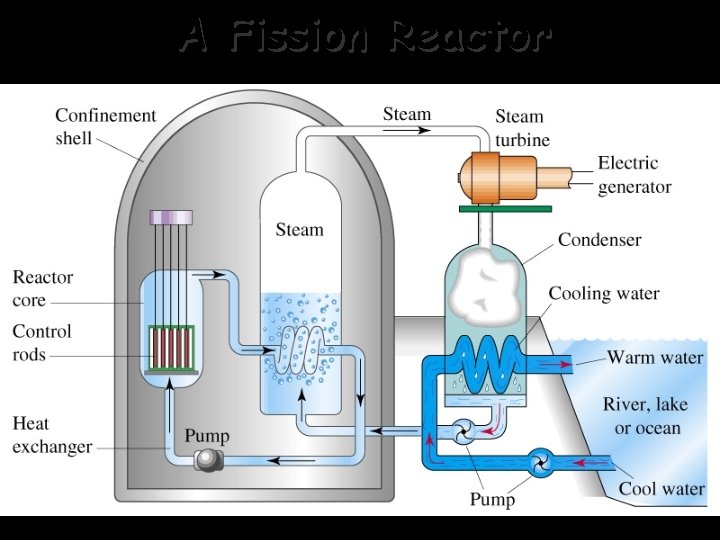
A Fission Reactor

HW: Read Chapter 25. 1 and 25. 2: Do Questions #1 -4 on page 809 and #10 -12 on page 814. Write the questions AND answer in your notebook.

CA Standards Students know protons and neutrons in the nucleus are held together by nuclear forces that overcome the electromagnetic repulsion between the protons. Students know the energy release per gram of material is much larger in nuclear fusion or fission reactions than in chemical reactions. The change in mass (calculated by E = mc 2) is small but significant in nuclear reactions.

CA Standards Students know the three most common forms of radioactive decay (alpha, beta, and gamma) and know how the nucleus changes in each type of decay. Students know alpha, beta, and gamma radiation produce different amounts and kinds of damage in matter and have different penetrations. Students know some naturally occurring isotopes of elements are radioactive, as are isotopes formed in nuclear reactions.
- Slides: 98