9 Sci Chemistry Periodic Table Elements A group

9 Sci - Chemistry Periodic Table
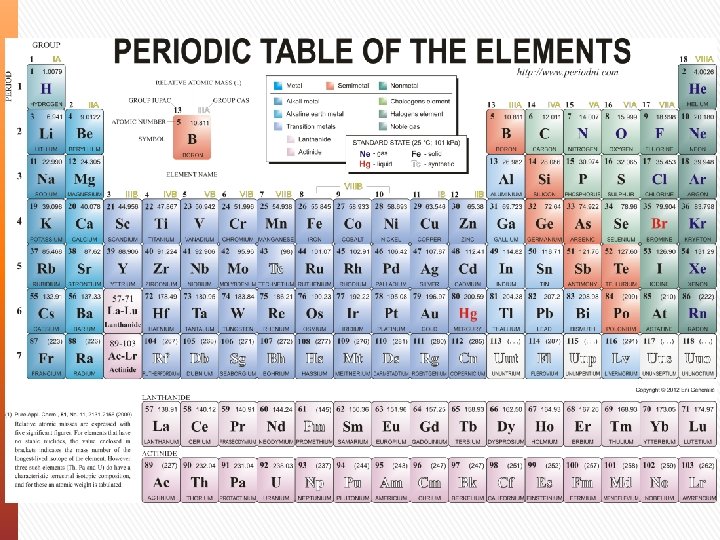

Elements A group of the same atoms are called elements

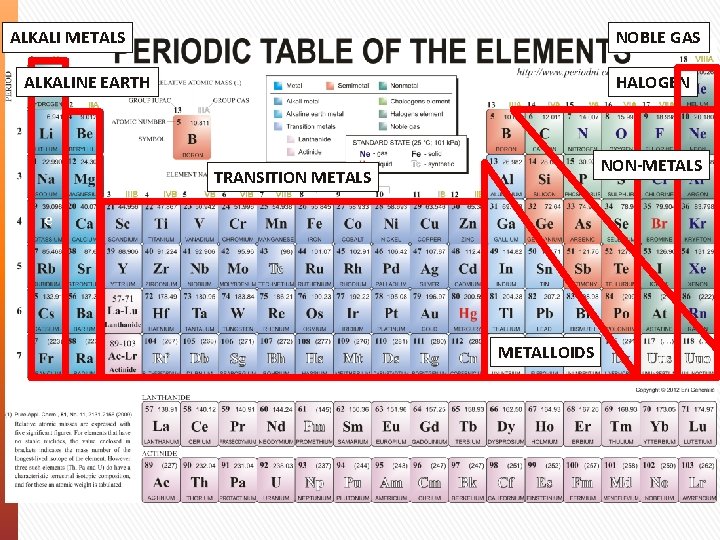
Periodic Table Periodic Law - The properties of elements aren’t random. They follow a distinct pattern and have similar chemical and physical properties at certain intervals. In your periodic table: » Elements are shown by order of size (atomic weight) » Rows of elements are known as Periods » Columns of elements are known as Groups (I to VIII)
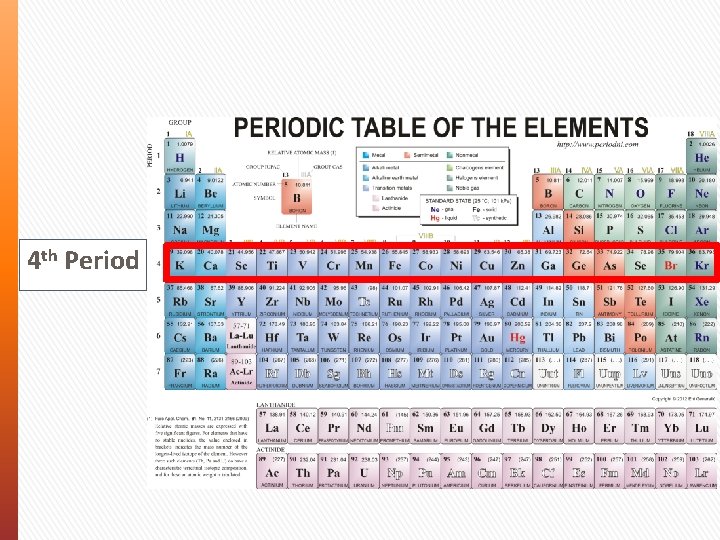
4 th Period
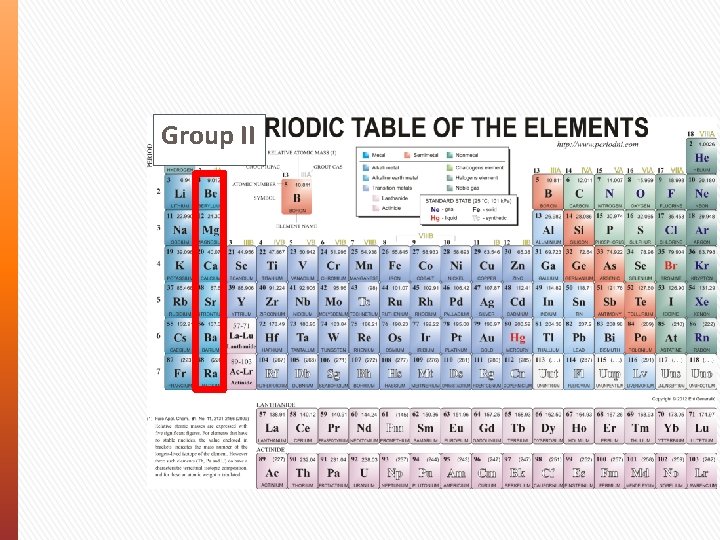
Group II

Elements Chemical symbol/formula – The letter(s) that represent the element. Most elements are just the first letter of their name, but this isn’t the case for all elements. Atomic Number – Number of Protons/Electrons in the atom in its neutral state. *In any neutral atom: No. Protons = No. Electrons Relative Atomic Mass The total mass of the atom (relative to Carbon-12) Mass Number Total number of protons and neutrons (not shown)

Elements » Chemical Formula of Cobalt? » Atomic number of Aluminium? » Period and Group of Gold? » What element is in Group IV, Period 2? » How many protons does Sodium have? » How many electrons does Titanium have?

Electron shells Electrons are found within specific electron shells. » The first shell holds max 2 electrons » The second shell holds max 8 electrons » The third shell holds max 18 electrons » The fourth shell holds max 32 electrons Valence Electrons – The electrons in the outer shell. This determines the chemical properties of elements Octet Rule – Atoms are most stable and want to have 8 electrons in their outer shell (Valence electrons)

Electron shells Period number will tell you how many electron shells the atom has. Group number (I to VIII) will tell you how many electrons are in the outer shell (Valence electrons)

Periodic Trends » The size of the atoms increase down the group There’s an increase in the number of shells going down. » The size of the atoms decrease from left to right of a period. This is because the more protons and electrons, the more compact the atom becomes.
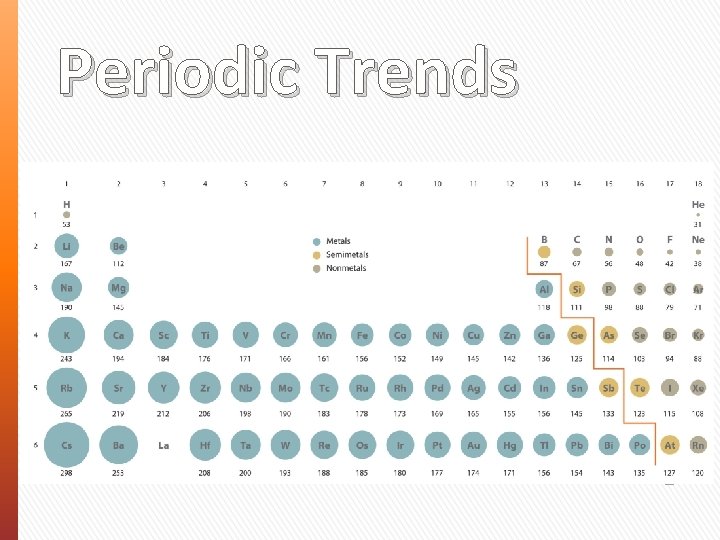
Periodic Trends

Periodic Trends » Group I (1): Alkali Metals – They are very reactive and naturally react with water to form basic alkaline solutions. » Group II (2): Alkaline Earth Metals – Abundant in the earth and also react to form basic alkaline solutions. » Group 3 -12: Transition metals – Most of the strong metals » Group III-VIII (13 -18): Non-metals » Group VII (17): Halogens – Reactive non-metals. » Group VIII (18): Noble gas – Stable & non-reactive
ALKALI METALS NOBLE GAS ALKALINE EARTH HALOGEN NON-METALS TRANSITION METALS e METALLOIDS

Ions The number of electrons an atom contains is not fixed. Atoms are most stable when they have 8 electrons in their outer shell (except for the first shell). Atoms can gain or lose electrons by interacting with other atoms to become stable. When an atom gains or loses electrons they become charged (+ or -). This is called an Ion Cation = Positive Ion Anion = Negative Ion

Ions Example: Sodium SODIUM ATOM • Atomic No. = 11 • Electronic Configuration = 2, 8, 1 • Valence electrons = 1 To become stable it wants to lose 1 electron. SODIUM ION Sodium Ion: Lost 1 electron to become stable • Valence electrons = 8 • No. protons = 11 • No. electrons = 10 Na 1+ or Na+ Sodium Cation
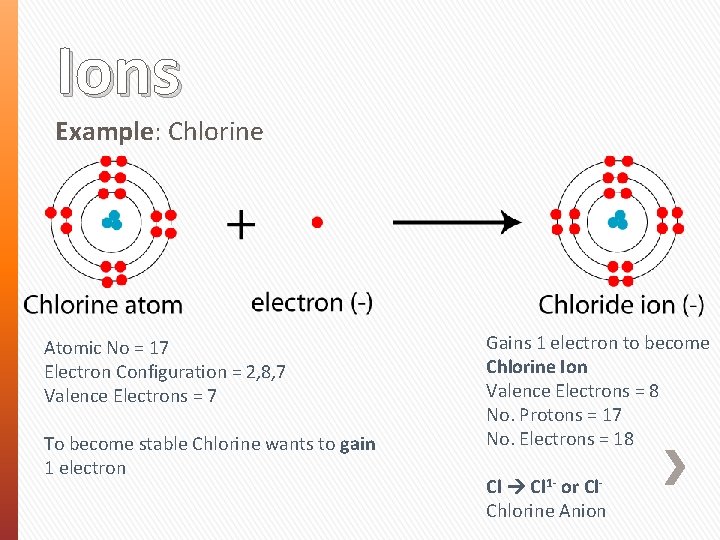
Ions Example: Chlorine Atomic No = 17 Electron Configuration = 2, 8, 7 Valence Electrons = 7 To become stable Chlorine wants to gain 1 electron Gains 1 electron to become Chlorine Ion Valence Electrons = 8 No. Protons = 17 No. Electrons = 18 Cl 1 - or Cl. Chlorine Anion
- Slides: 17