9 Sci Chemistry Chemical Bonding Chemical Bonding Where

9 Sci - Chemistry Chemical Bonding
Chemical Bonding Where is water?

Chemical Bonding A chemical bond is an attraction between atoms. Atoms are held together by the interaction of their outer shells (valence electrons) Atoms can join together to form larger substances.

Types of Bonding Chemical bonds can be either ‘strong’ or ‘weak’ Strong Bonds » Metallic Bonding – Metal with metal » Ionic Bonding – Metal with non-metal » Covalent Bonding – Non-metal with non-metal Weak Bonds » Dipole-dipole » Hydrogen bonding » Dispersion forces

Ionic Bonding An ionic bond results from the complete transfer of one or more electrons from a metal to a nonmetal. Atoms of different elements can join together to form new substances. A substance which is made up of two or more different types of atoms is known as a compound. One way this can occur is for atoms to form ions.
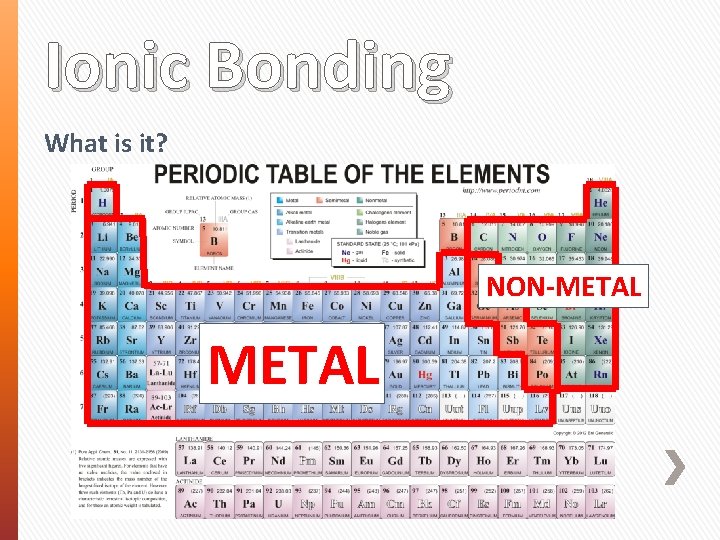
Ionic Bonding What is it? NON-METAL

Ionic Bonding Metals usually want to give electrons (electron doners) » Becomes positively charged ion (cation) Non-Metals usually want to take electrons (electron receivers) » Becomes negatively charged ion (anion) » Changes name suffix to –ide (e. g. Chlorine Chloride) The metal will donate the electron to the non-metal The cation and anion are opposite in charge and become attracted to each other (electrostatic forces)
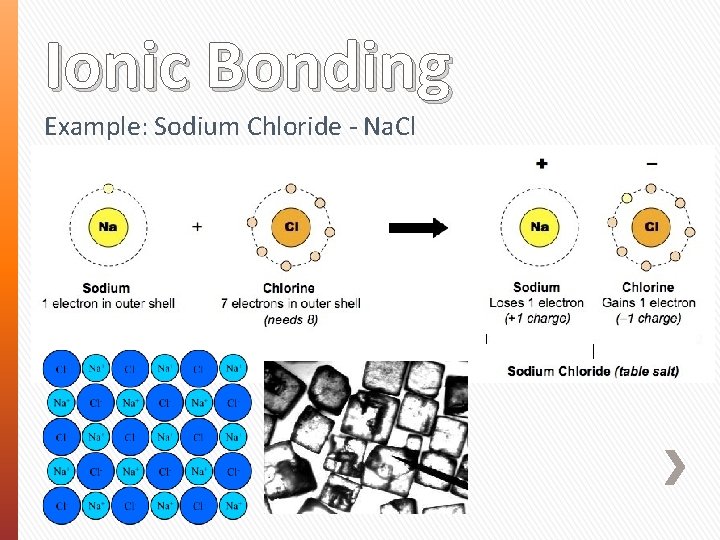
Ionic Bonding Example: Sodium Chloride - Na. Cl
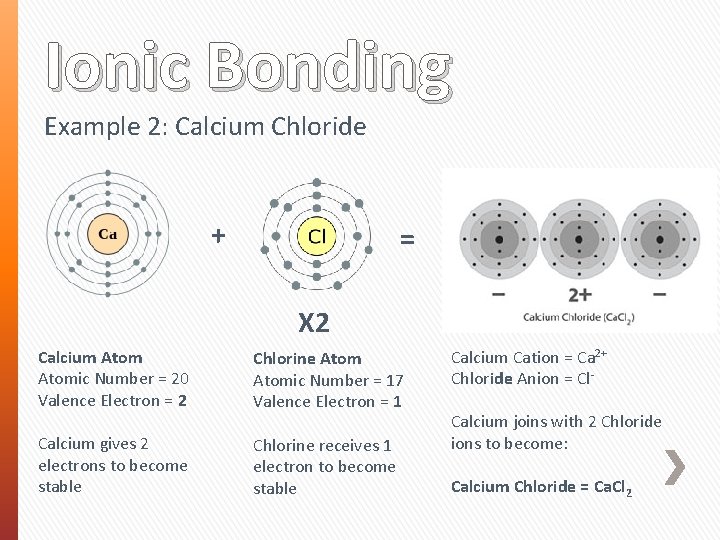
Ionic Bonding Example 2: Calcium Chloride + = X 2 Calcium Atomic Number = 20 Valence Electron = 2 Chlorine Atomic Number = 17 Valence Electron = 1 Calcium gives 2 electrons to become stable Chlorine receives 1 electron to become stable Calcium Cation = Ca 2+ Chloride Anion = Cl. Calcium joins with 2 Chloride ions to become: Calcium Chloride = Ca. Cl 2
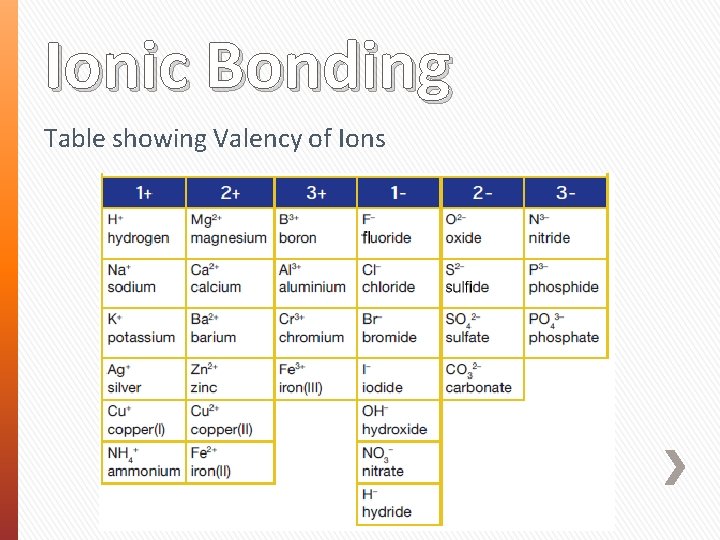
Ionic Bonding Table showing Valency of Ions
- Slides: 10