9 8 NOTES Alkane Boiling Points A 10

9. 8 – NOTES Alkane Boiling Points
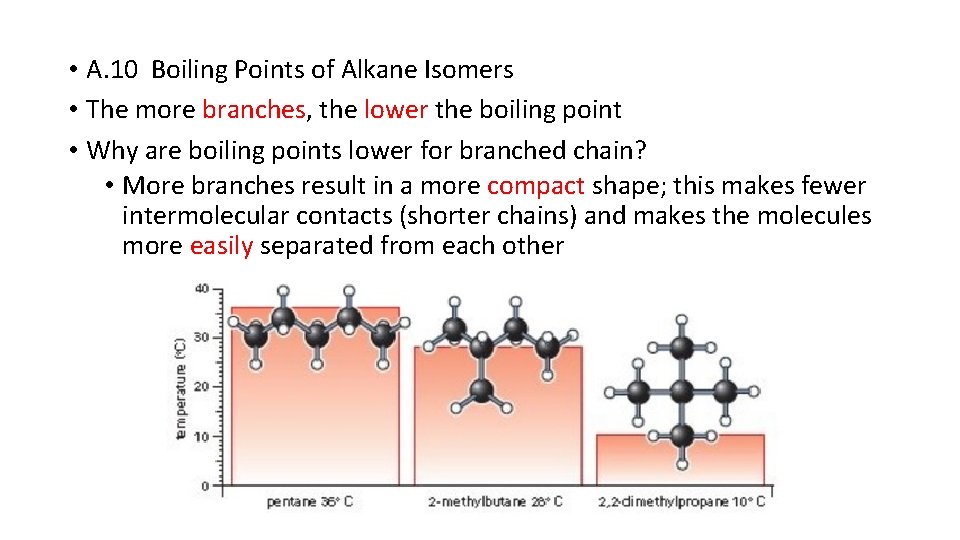
• A. 10 Boiling Points of Alkane Isomers • The more branches, the lower the boiling point • Why are boiling points lower for branched chain? • More branches result in a more compact shape; this makes fewer intermolecular contacts (shorter chains) and makes the molecules more easily separated from each other

• Viscosity • Resistance to flow • The more viscous something is, the less easily it flows • Viscosity and Temperature • Temperature can affect viscosity of a substance • When you increase the temperature of the fluid, the viscosity decreases, or flows more easily. • Heat goes up, viscosity goes down •


• A. 11 Fuels and Climate • Different parts of the country have different climates, resulting in different fuel additives added • Automobile fuel used in Maine during the cold winter months differs from the fuel used in Arizona during hot summer •

• Why do different areas need different types of fuel? • Volatility affects performance • Warmer climates need fuels that don’t easily vaporize (vapor lock) • Colder climates need fuels that don’t solidify or become viscous

• What physical property should be engineered to make the fuel appropriate for each climate? • Volatility • Viscosity • • Molecular structure is used to vary properties to make the different chemical substances for the various materials made from petroleum
- Slides: 7