9 6 Multiple Bonds In covalent bonds as
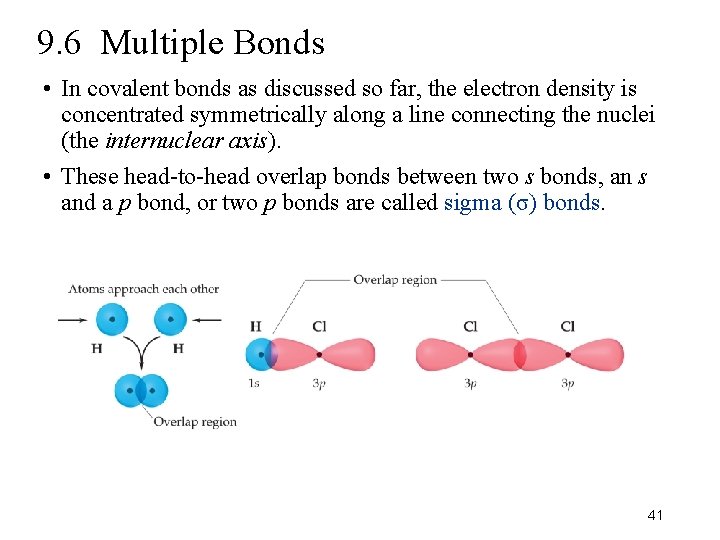
9. 6 Multiple Bonds • In covalent bonds as discussed so far, the electron density is concentrated symmetrically along a line connecting the nuclei (the internuclear axis). • These head-to-head overlap bonds between two s bonds, an s and a p bond, or two p bonds are called sigma (σ) bonds. 41
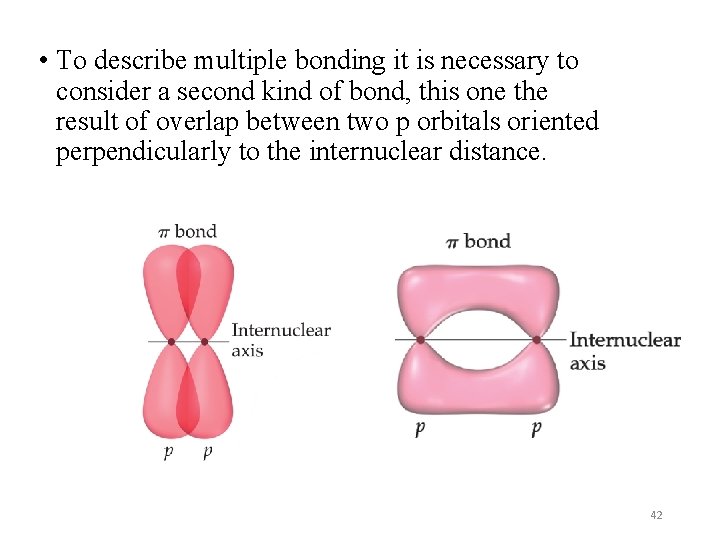
• To describe multiple bonding it is necessary to consider a second kind of bond, this one the result of overlap between two p orbitals oriented perpendicularly to the internuclear distance. 42
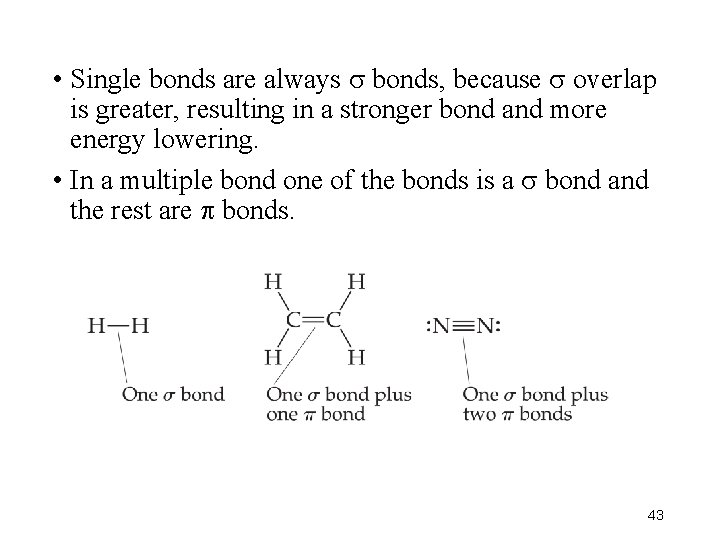
• Single bonds are always bonds, because overlap is greater, resulting in a stronger bond and more energy lowering. • In a multiple bond one of the bonds is a bond and the rest are bonds. 43

Examples of Multiple Bond Hybridization 1. Ethylene • Ethylene (C 2 H 4) contains a C=C double bond. • After hybridization, one electron remains in an unhybridized 2 p orbital. 44
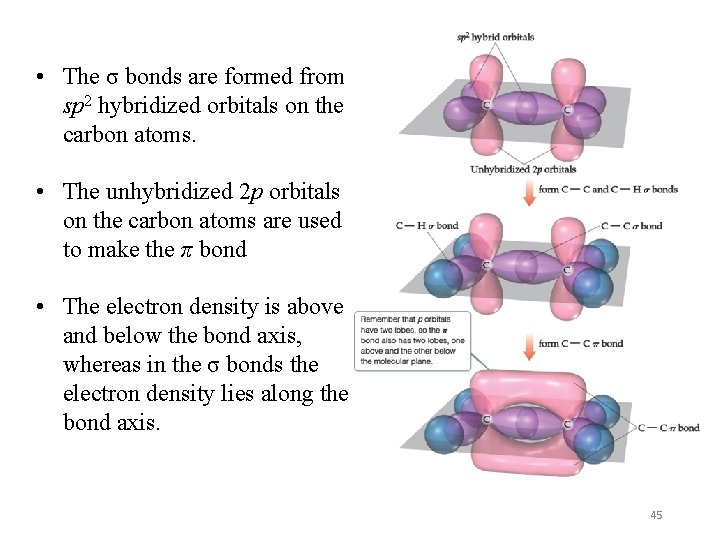
• The σ bonds are formed from sp 2 hybridized orbitals on the carbon atoms. • The unhybridized 2 p orbitals on the carbon atoms are used to make the π bond • The electron density is above and below the bond axis, whereas in the σ bonds the electron density lies along the bond axis. 45

2. Formaldehyde • In a molecule like formaldehyde (shown at left) an sp 2 orbital on carbon overlaps in fashion with the corresponding orbital on the oxygen. • The unhybridized p orbitals overlap in fashion. 46
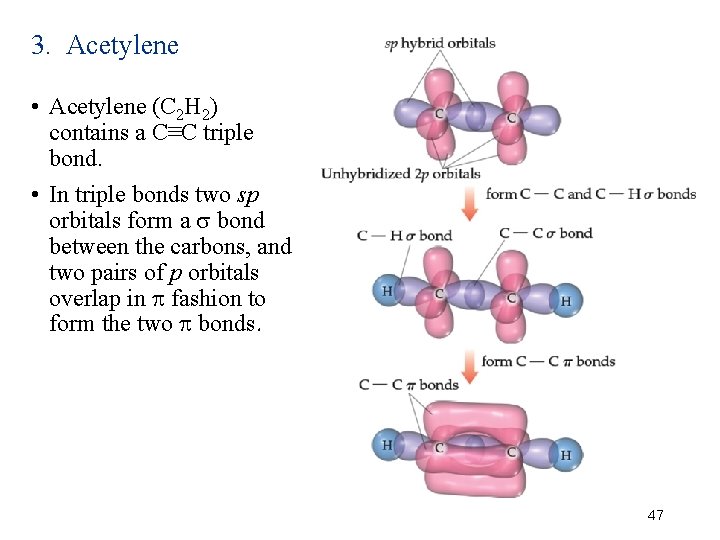
3. Acetylene • Acetylene (C 2 H 2) contains a C≡C triple bond. • In triple bonds two sp orbitals form a bond between the carbons, and two pairs of p orbitals overlap in fashion to form the two bonds. 47
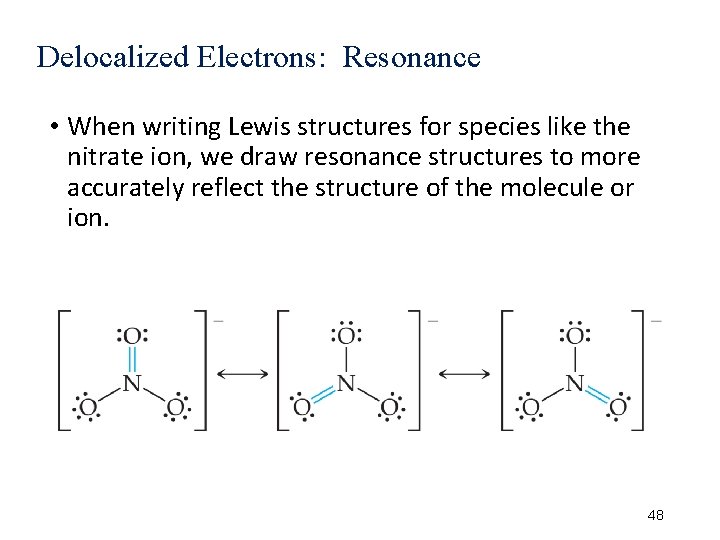
Delocalized Electrons: Resonance • When writing Lewis structures for species like the nitrate ion, we draw resonance structures to more accurately reflect the structure of the molecule or ion. 48
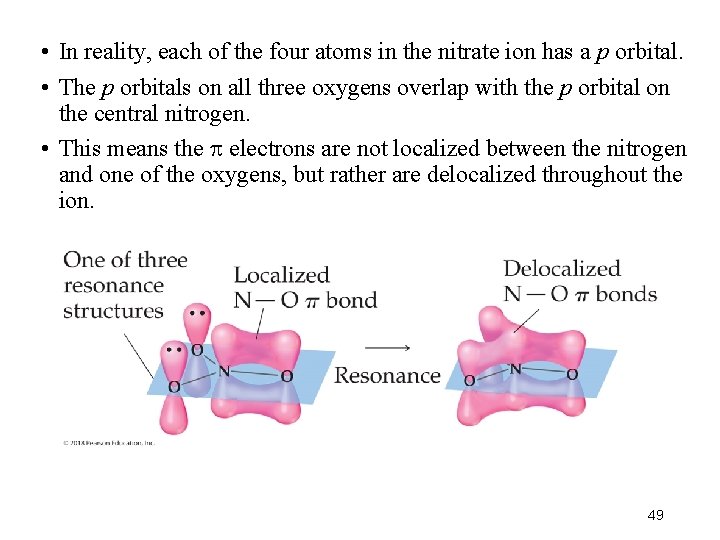
• In reality, each of the four atoms in the nitrate ion has a p orbital. • The p orbitals on all three oxygens overlap with the p orbital on the central nitrogen. • This means the electrons are not localized between the nitrogen and one of the oxygens, but rather are delocalized throughout the ion. 49
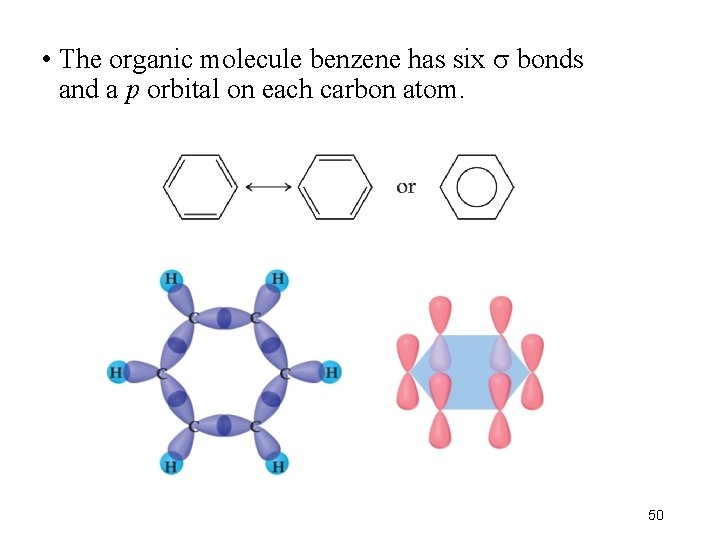
• The organic molecule benzene has six bonds and a p orbital on each carbon atom. 50
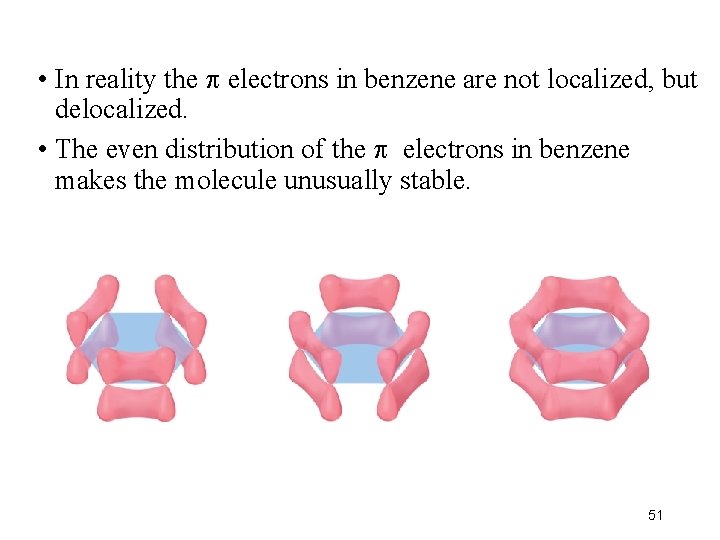
• In reality the electrons in benzene are not localized, but delocalized. • The even distribution of the electrons in benzene makes the molecule unusually stable. 51
- Slides: 11