9 2 Advanced Materials Lab 1 9 3

제 9장 2원 합금 상태도 작성법 Advanced Materials Lab 1







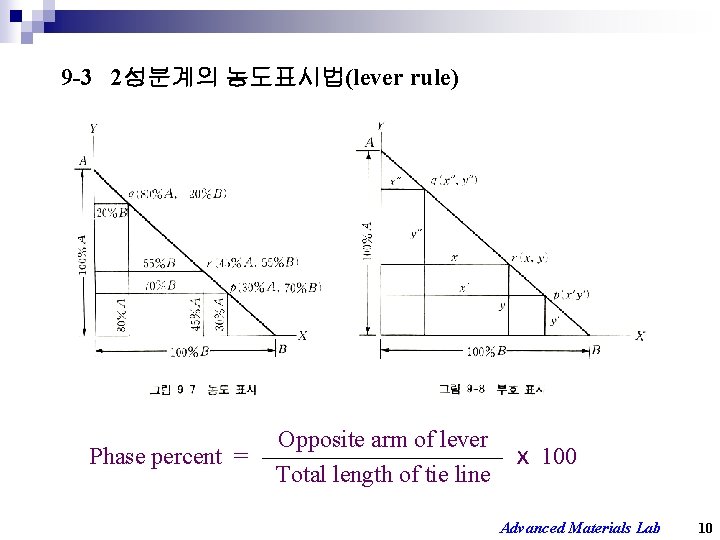
9 -3 2성분계의 농도표시법(lever rule) Opposite arm of lever Phase percent = ⅹ 100 Total length of tie line Advanced Materials Lab 10
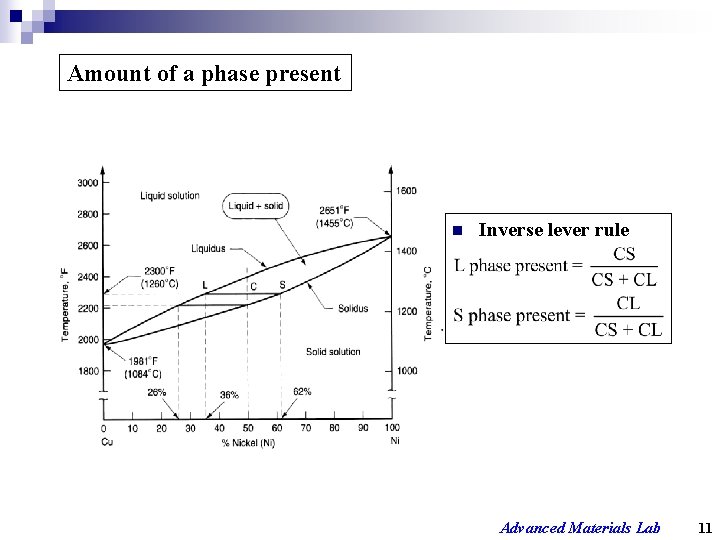
Amount of a phase present n Inverse lever rule Advanced Materials Lab 11
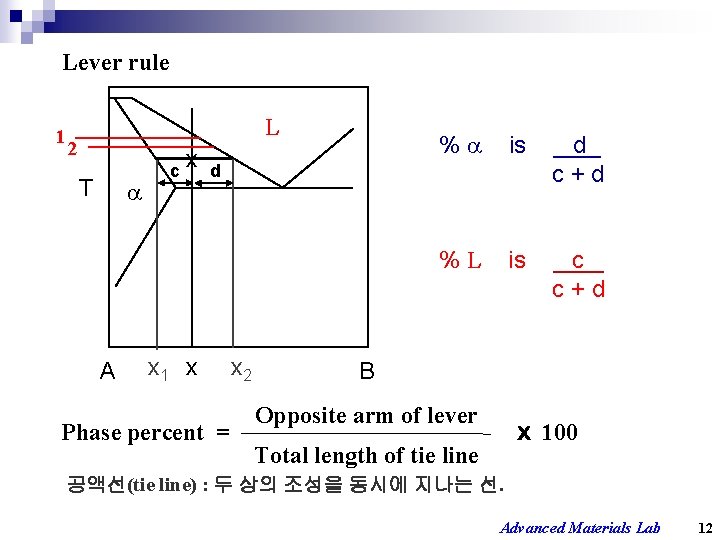
Lever rule 1 L 2 T A x c x 1 x % is d c+d %L is c c+d d x 2 B Opposite arm of lever Phase percent = ⅹ 100 Total length of tie line 공액선(tie line) : 두 상의 조성을 동시에 지나는 선. Advanced Materials Lab 12
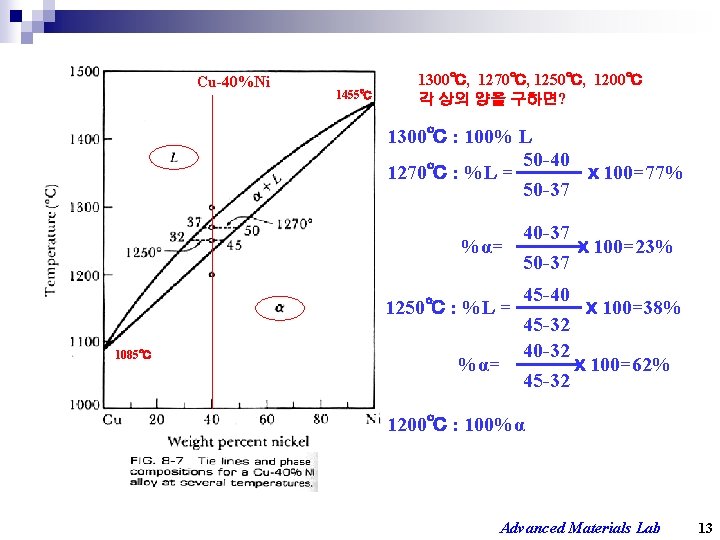
Cu-40%Ni 1455℃ 1300℃, 1270℃, 1250℃, 1200℃ 각 상의 양을 구하면? 1300℃ : 100% L 50 -40 1270℃ : %L = ⅹ 100=77% 50 -37 40 -37 %α= ⅹ 100=23% 50 -37 1085℃ 45 -40 1250℃ : %L = ⅹ 100=38% 45 -32 40 -32 %α= ⅹ 100=62% 45 -32 1200℃ : 100%α Advanced Materials Lab 13
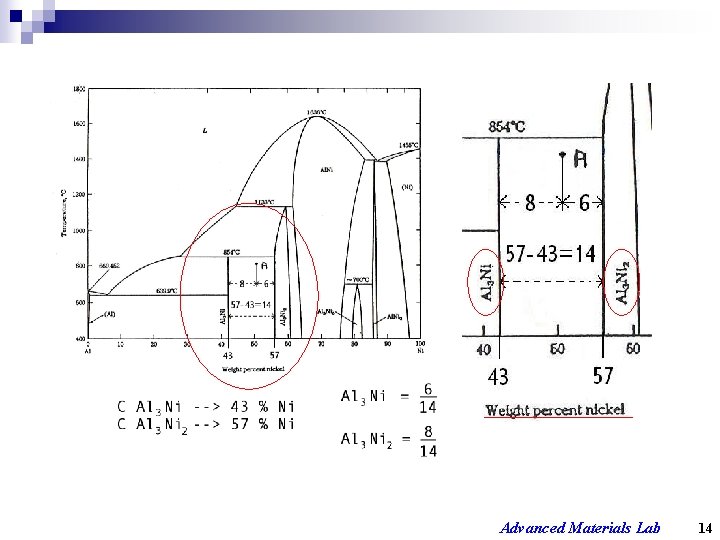
Advanced Materials Lab 14

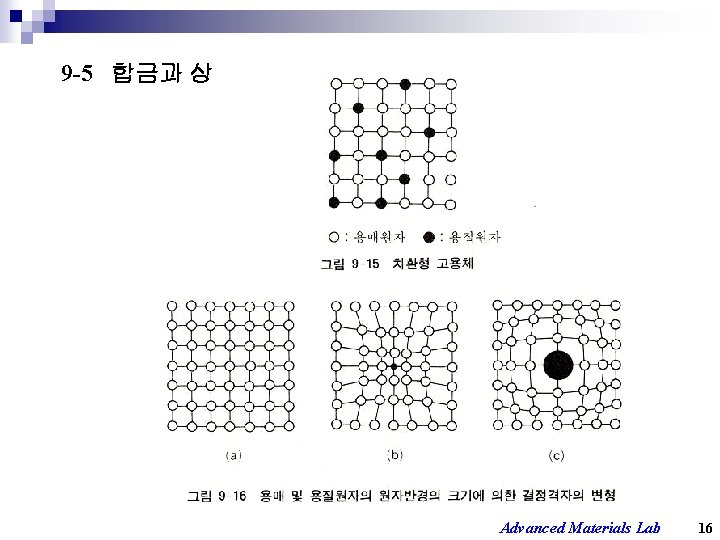
9 -5 합금과 상 Advanced Materials Lab 16

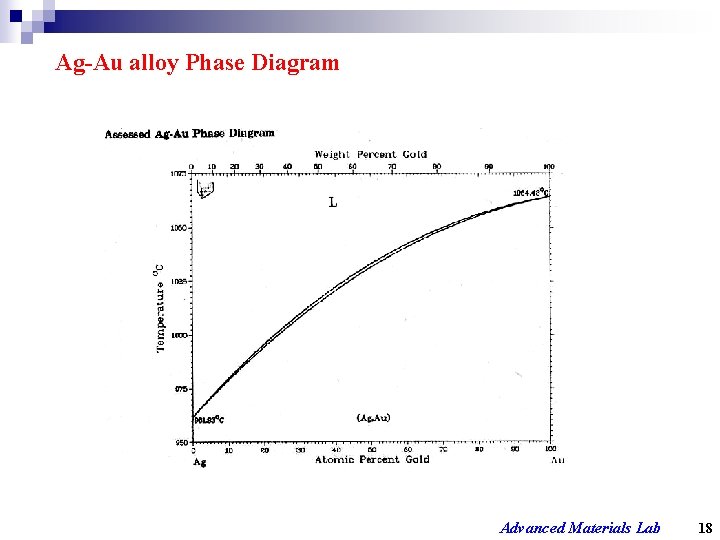
Ag-Au alloy Phase Diagram Advanced Materials Lab 18
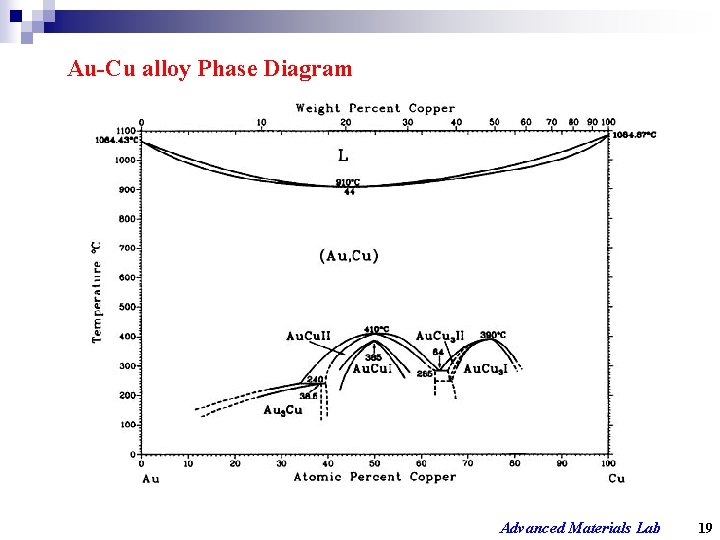
Au-Cu alloy Phase Diagram Advanced Materials Lab 19
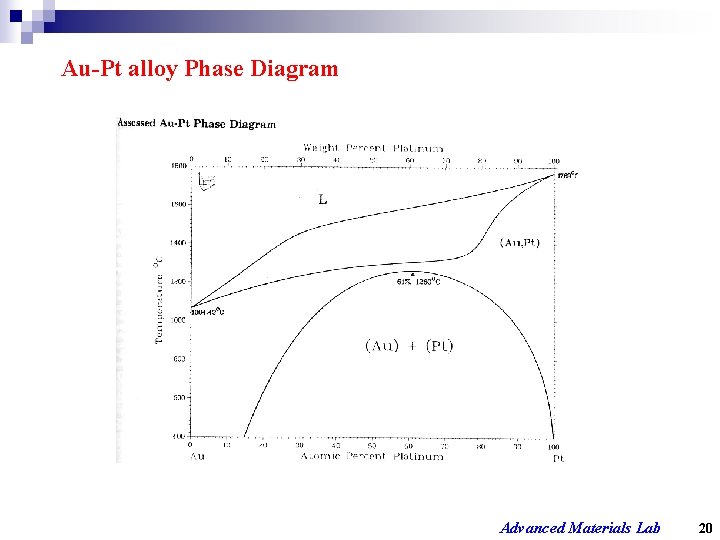
Au-Pt alloy Phase Diagram Advanced Materials Lab 20
Advanced Materials Lab 21
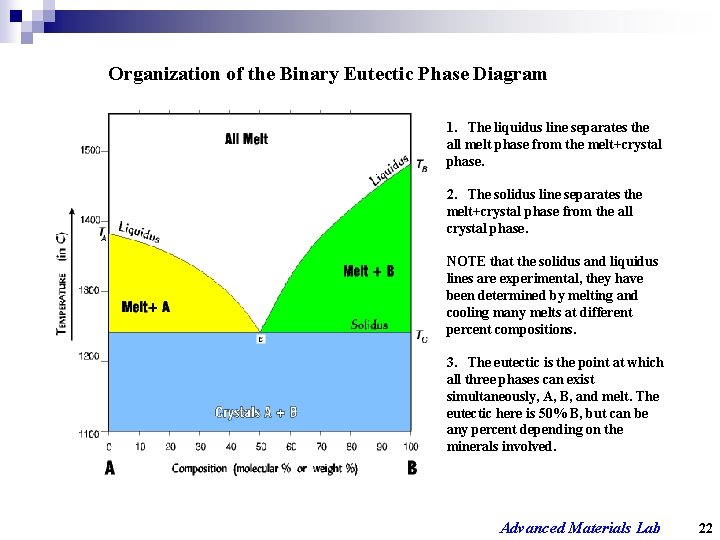
Organization of the Binary Eutectic Phase Diagram 1. The liquidus line separates the all melt phase from the melt+crystal phase. 2. The solidus line separates the melt+crystal phase from the all crystal phase. NOTE that the solidus and liquidus lines are experimental, they have been determined by melting and cooling many melts at different percent compositions. 3. The eutectic is the point at which all three phases can exist simultaneously, A, B, and melt. The eutectic here is 50% B, but can be any percent depending on the minerals involved. Advanced Materials Lab 22
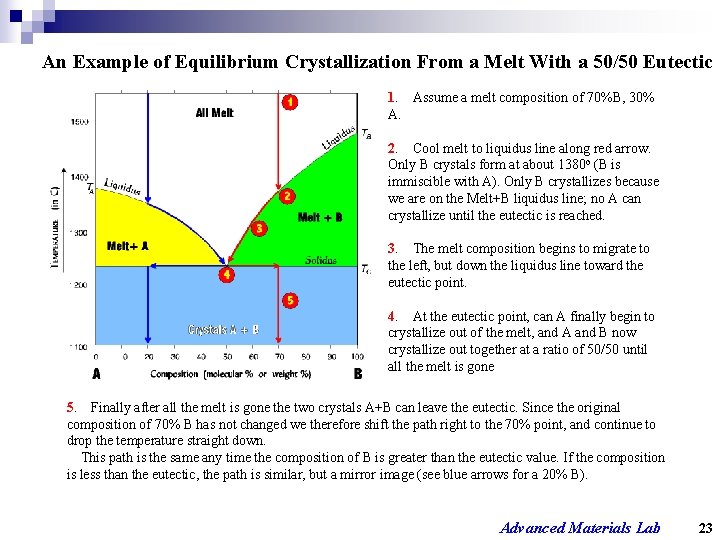
An Example of Equilibrium Crystallization From a Melt With a 50/50 Eutectic 1. Assume a melt composition of 70%B, 30% A. 2. Cool melt to liquidus line along red arrow. Only B crystals form at about 1380 o (B is immiscible with A). Only B crystallizes because we are on the Melt+B liquidus line; no A can crystallize until the eutectic is reached. 3. The melt composition begins to migrate to the left, but down the liquidus line toward the eutectic point. 4. At the eutectic point, can A finally begin to crystallize out of the melt, and A and B now crystallize out together at a ratio of 50/50 until all the melt is gone 5. Finally after all the melt is gone the two crystals A+B can leave the eutectic. Since the original composition of 70% B has not changed we therefore shift the path right to the 70% point, and continue to drop the temperature straight down. This path is the same any time the composition of B is greater than the eutectic value. If the composition is less than the eutectic, the path is similar, but a mirror image (see blue arrows for a 20% B). Advanced Materials Lab 23
- Slides: 22