8 E Cytochrome c The second mobile electron
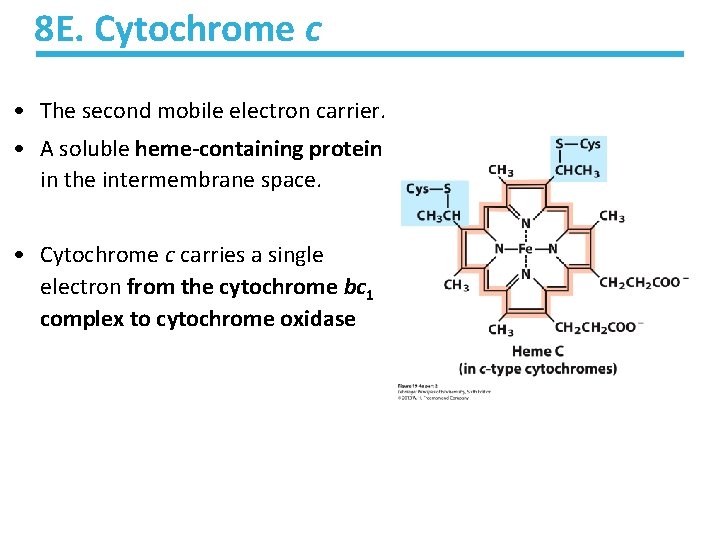
8 E. Cytochrome c • The second mobile electron carrier. • A soluble heme-containing protein in the intermembrane space. • Cytochrome c carries a single electron from the cytochrome bc 1 complex to cytochrome oxidase

8 F. Cytochrome Oxidase, a. k. a. Complex IV • Mammalian cytochrome oxidase is a membrane protein with 13 subunits • Contains two heme groups: a and a 3 • Contains copper ions – Cu. A: two ions that accept electrons from Cyt c – Cu. B: bonded to heme a 3 forming a binuclear center that transfers four electrons to oxygen
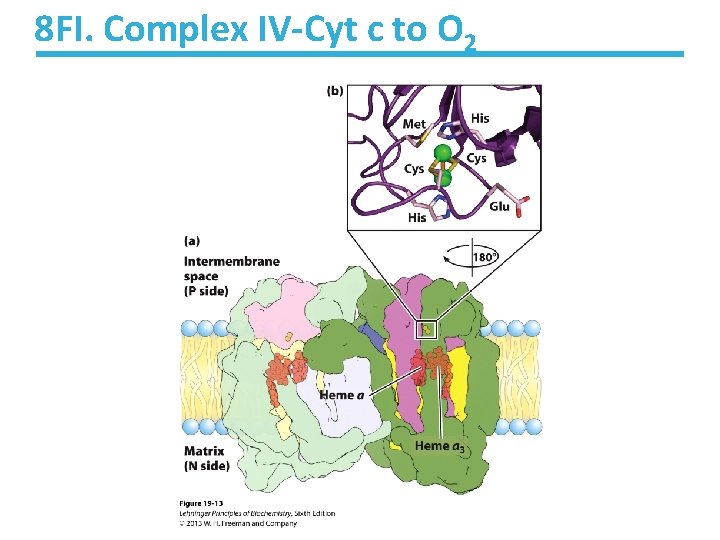
8 FI. Complex IV-Cyt c to O 2

8 FII. Cytochrome oxidase passes electrons to O 2 • Four electrons are used to reduce one oxygen molecule into two water molecules. • Four protons are picked up from the matrix in this process. • Four additional protons are passed from the matrix to the intermembrane space. 4 Cyt c (reduced) + 8 H+NO. S. + O 20→ 4 Cyt c (oxidized) + 4 H+p + 2 H O. S. 2 O -2
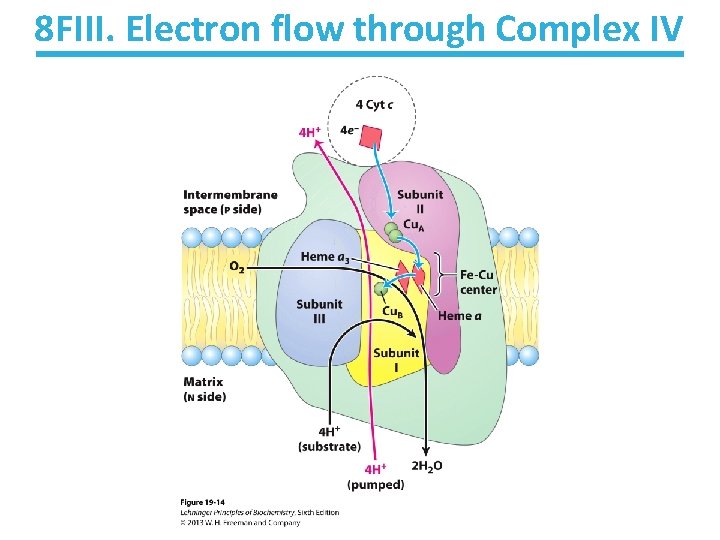
8 FIII. Electron flow through Complex IV
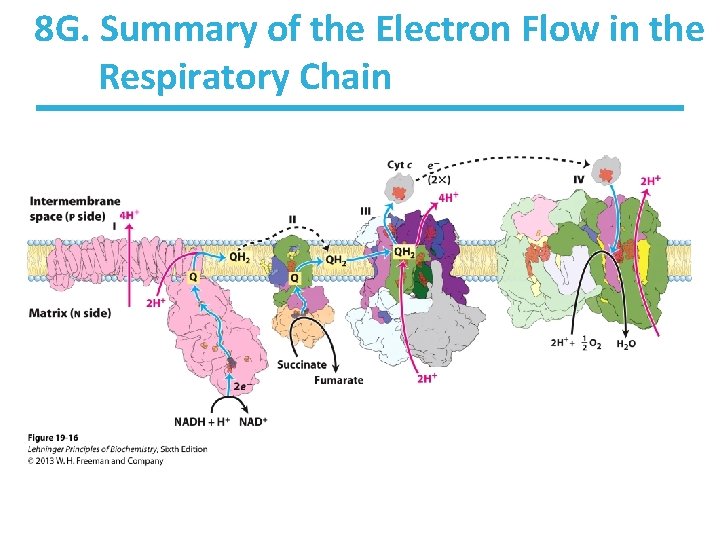
8 G. Summary of the Electron Flow in the Respiratory Chain

8 G. Summary of the Electron Flow in the Respiratory Chain • Complex IV 1 NADH + 11 H+(N) + ½O 2 ——> NAD+ + 10 H+(P) + H 2 O • Complex II Complex IV FADH 2 + 6 H+(N) + ½O 2 ——> FAD + 6 H+(P) + H 2 O Difference in number of protons transported ultimately reflects differences in ATP synthesized.

8 GI. Standard Free Energy for Electron Transfer 1 NADH + 11 H+(N) + ½O 2 ——> NAD+ + 10 H+(P) + H 2 O The transfer of 2 e- from NADH through the respiratory chain to O 2 can be written as: 1 NADH + H+ + ½O 2 → NAD+ + H 2 O Consider the redox pairs: NAD+/NADH, E’°= -0. 320 V O 2 /H 2 O, E’°= 0. 816 V ΔE’°= 1. 14 V

8 GI. Standard Free Energy for Electron Transfer 1 NADH + 11 H+(N) + ½O 2 ——> NAD+ + 10 H+(P) + H 2 O The transfer of 2 e- from NADH through the respiratory chain to O 2 can be written as: 1 NADH + H+ + ½O 2 → NAD+ + H 2 O ΔG’°= -n. FΔE’° = -2(96. 5 k. J/V · mol)(1. 14 V) = -220 k. J/mol The reaction is highly exergonic.

8 GI. Standard Free Energy for Electron Transfer •
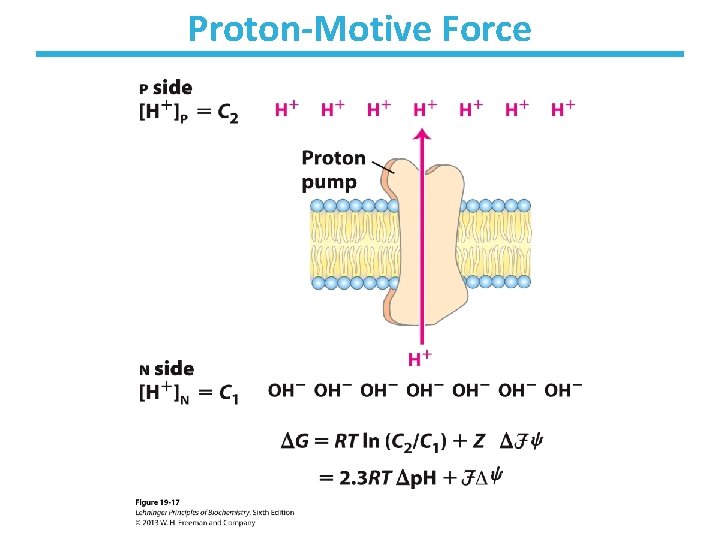
Proton-Motive Force

8 GI. Standard Free Energy for Electron Transfer •

8 GI. Standard Free Energy for Electron Transfer •
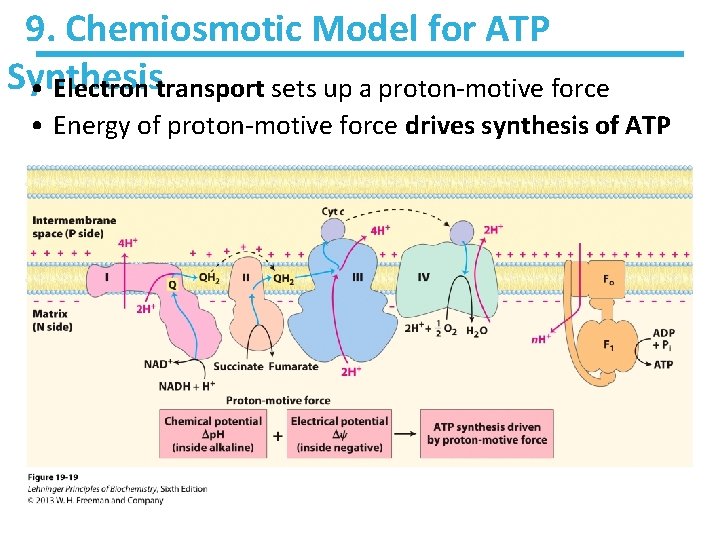
9. Chemiosmotic Model for ATP Synthesis • Electron transport sets up a proton-motive force • Energy of proton-motive force drives synthesis of ATP
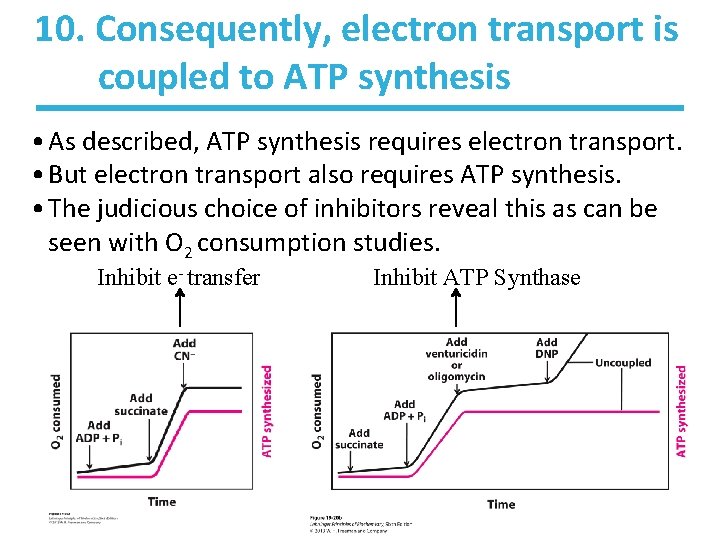
10. Consequently, electron transport is coupled to ATP synthesis • As described, ATP synthesis requires electron transport. • But electron transport also requires ATP synthesis. • The judicious choice of inhibitors reveal this as can be seen with O 2 consumption studies. Inhibit e- transfer Inhibit ATP Synthase

11. Mitochondrial ATP Synthase Complex • Contains two functional units: – F 1 • • Soluble complex in the matrix Individually catalyzes the hydrolysis of ATP – F 0 Integral membrane complex Transports protons from IMS to matrix, dissipating the proton gradient • Energy transferred to F 1 to catalyze phosphorylation of ADP • •
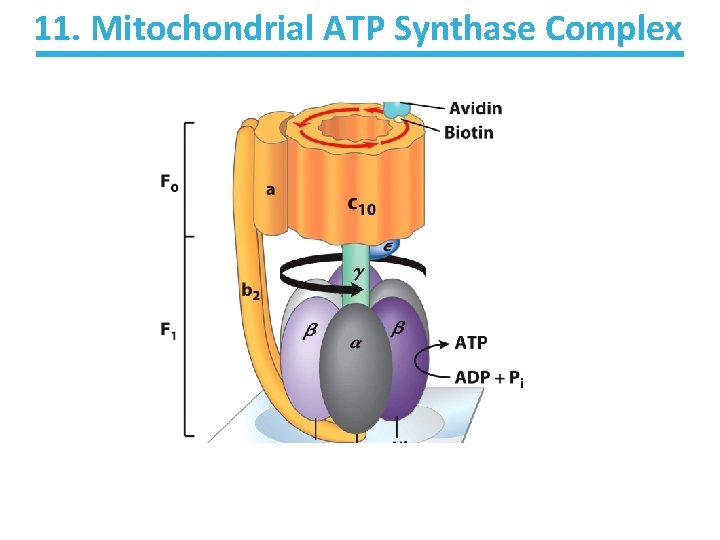
11. Mitochondrial ATP Synthase Complex
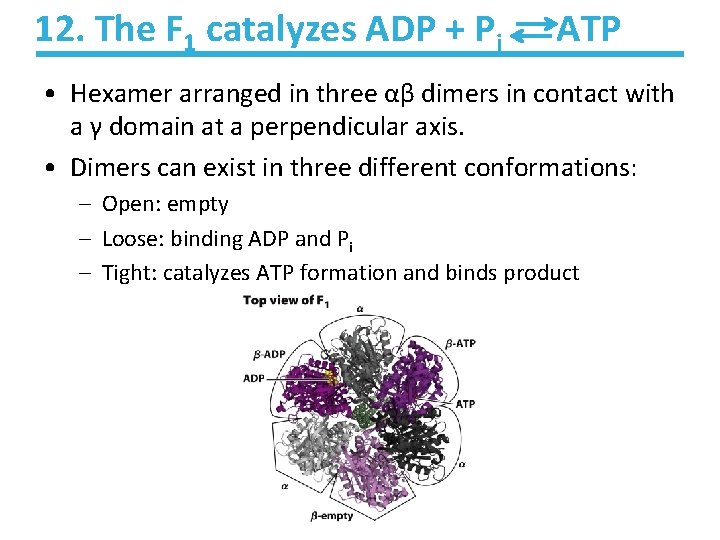
12. The F 1 catalyzes ADP + Pi ATP • Hexamer arranged in three αβ dimers in contact with a γ domain at a perpendicular axis. • Dimers can exist in three different conformations: – Open: empty – Loose: binding ADP and Pi – Tight: catalyzes ATP formation and binds product
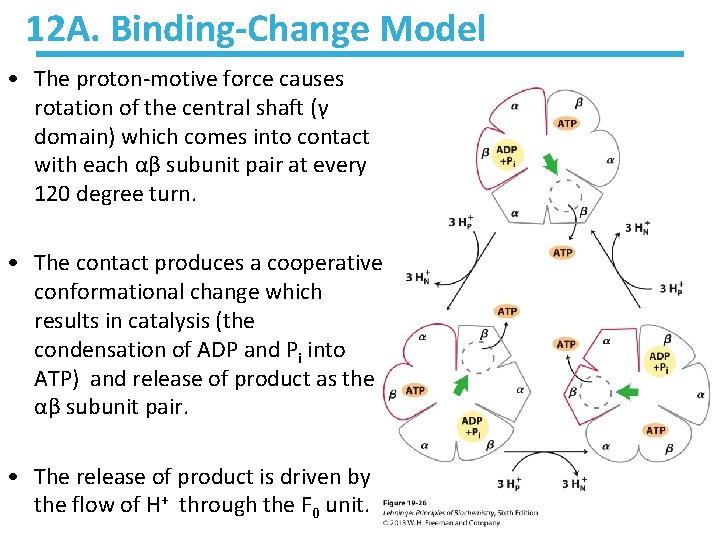
12 A. Binding-Change Model • The proton-motive force causes rotation of the central shaft (γ domain) which comes into contact with each αβ subunit pair at every 120 degree turn. • The contact produces a cooperative conformational change which results in catalysis (the condensation of ADP and Pi into ATP) and release of product as the αβ subunit pair. • The release of product is driven by the flow of H+ through the F 0 unit.
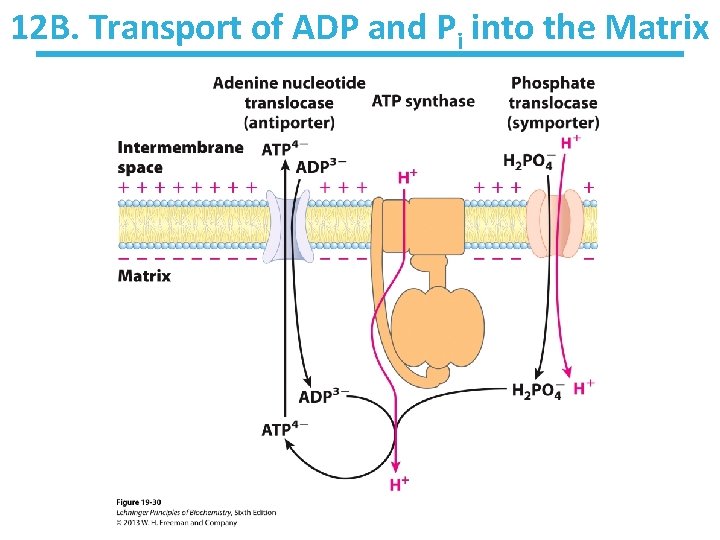
12 B. Transport of ADP and Pi into the Matrix
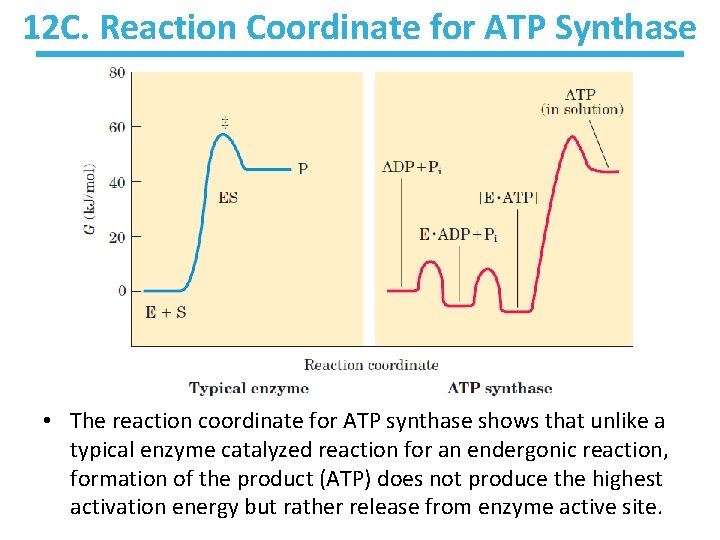
12 C. Reaction Coordinate for ATP Synthase • The reaction coordinate for ATP synthase shows that unlike a typical enzyme catalyzed reaction for an endergonic reaction, formation of the product (ATP) does not produce the highest activation energy but rather release from enzyme active site.

13. ATP Output per Source of Electron Carrier • For every NADH that enters the respiratory chain, 2. 5 ATP are produced. • For every succinate (or FADH 2) that enters, 1. 5 ATP are produced.
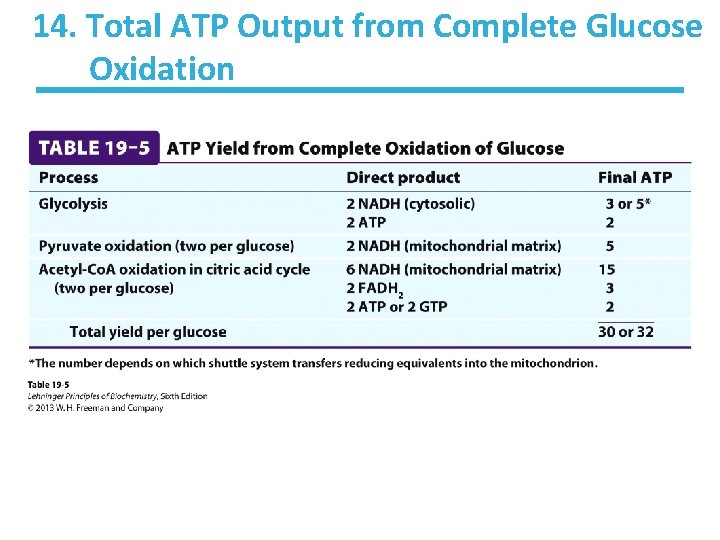
14. Total ATP Output from Complete Glucose Oxidation

15. Regulation of Oxidative Phosphorylation • Primarily regulated by substrate availability – NADH and ADP/Pi – Due to coupling both substrates required for electron transport and ATP synthesis • Inhibitor of F 1 (IF 1) – Prevents hydrolysis of ATP during low oxygen – Only active at lower p. H, encountered when electron transport it stalled (i. e. , low oxygen) • Inhibition of Ox. Phos leads to accumulation of NADH – Causes feedback inhibition cascade up to PFK-1 in glycoysis
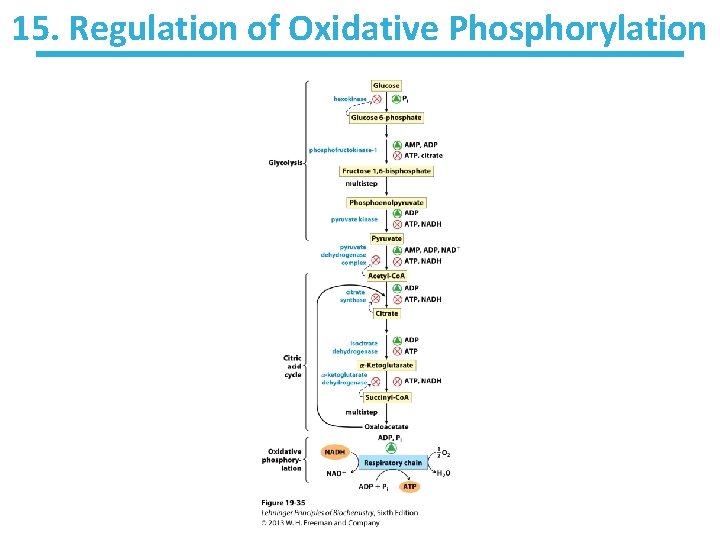
15. Regulation of Oxidative Phosphorylation

Final Extra Credit Assignment 26

In our class discussions, we have focused on aerobic respiration from the context of glucose metabolism. Now I would like for you to look up the metabolic pathways involved in anaerobic respiration starting from the context of glucose metabolism. 1. Draw out a general schematic for the pathways involved. 2. MAKE SURE to references your source(s)!!!!! 3. Due in my office on April 30 th with HW#5.

General Paper Comments 28

Things to Note 1. Science writing is very formal but should NOT be without personality. § With that said, try to avoid the use of the words I and We, unless absolutely necessary. 2. NEVER write an article as a stream of quotes. § The objective is to process the information that you are reading and not regurgitate without your understanding. § Construct your own conclusions from the data presented in primary literature. 3. Number and label all figures and tables properly. § Place legends below figures but above tables and refer to it properly in your writing. 4. Make sure to be consistent about referencing. § Formatting can be different but it is crucial to be consistent and follow a template.

Final Words 30

Be Critical In vitro vs in vivo studies: § In vitro studies in biochemistry are commonly referred to as “test tube” experiments but they generally are experiments performed using components extracted from an organism. - They provide a rich amount of information about different biomolecules and metabolic pathways. - However, they do not provide a realistic context of how biomolecules interact. • Compartmentalization • Concentration • Hydrophobic/hydrophilic environments § In vivo studies are those involving work with living organisms. - A shift towards these types of experiments. - But are costly, complicated, and ethically challenging.

Be Critical Often what we are taught/learn has been obtained from well-constructed in vitro experiments but as techniques become more sophisticated, less sample consuming, and less expensive in vivo studies will be easier to perform and analyze. They may very well change many of our current understandings about biochemistry but in the mean time it is important to be open-minded but be critical about accepting everything as absolute.
- Slides: 32