8 CHAPTER BONDING IN TRANSITION METAL COMPOUNDS AND

8 CHAPTER BONDING IN TRANSITION METAL COMPOUNDS AND COORDINATION COMPLEXES 8. 1 Chemistry of the Transition Metals 8. 2 Introduction to Coordination Chemistry 8. 3 Structures of Coordination Complexes 8. 4 Crystal Field Theory: Optical and Magnetic Properties 8. 5 Optical Properties and the Spectrochemical Series General Chemistry I 8. 6 Bonding in Coordination Complexes

347 Emerald 3 Be. O∙Al 2 O 3∙ 6 Si. O 2 with some Al 3+ replaced by Cr 3+ General Chemistry I
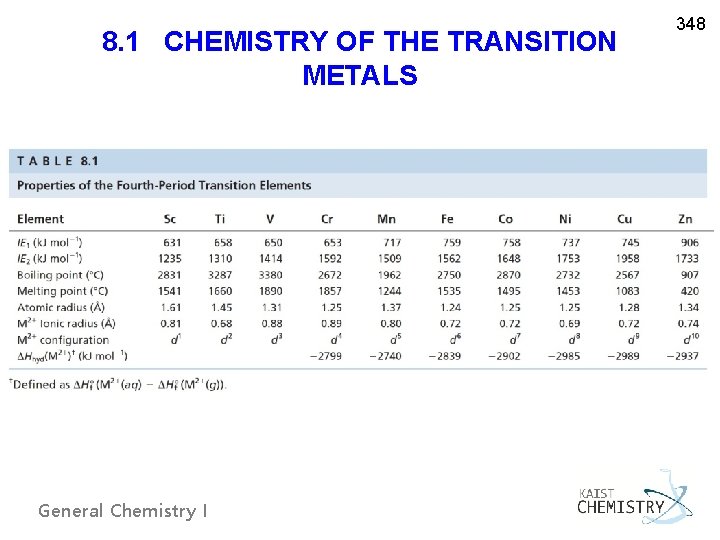
8. 1 CHEMISTRY OF THE TRANSITION METALS General Chemistry I 348
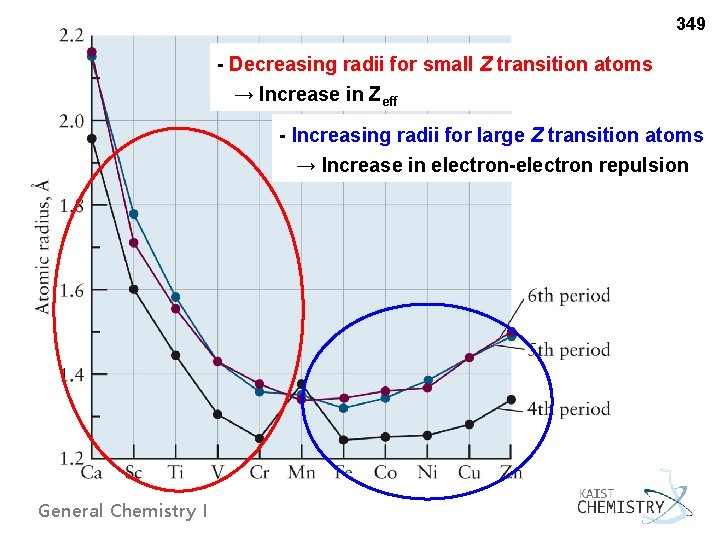
349 - Decreasing radii for small Z transition atoms → Increase in Zeff - Increasing radii for large Z transition atoms → Increase in electron-electron repulsion General Chemistry I
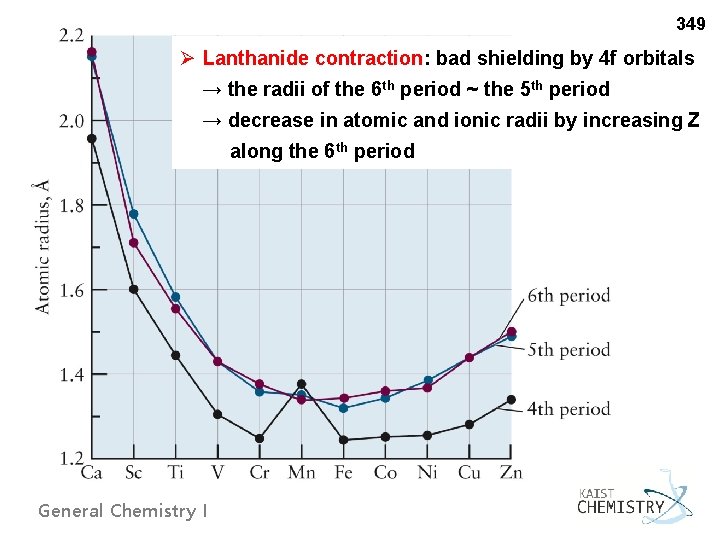
349 Ø Lanthanide contraction: bad shielding by 4 f orbitals → the radii of the 6 th period ~ the 5 th period → decrease in atomic and ionic radii by increasing Z along the 6 th period General Chemistry I

350 Ø melting point: function of the bond strength in solids - roughly correlated with the number of unpaired e- General Chemistry I
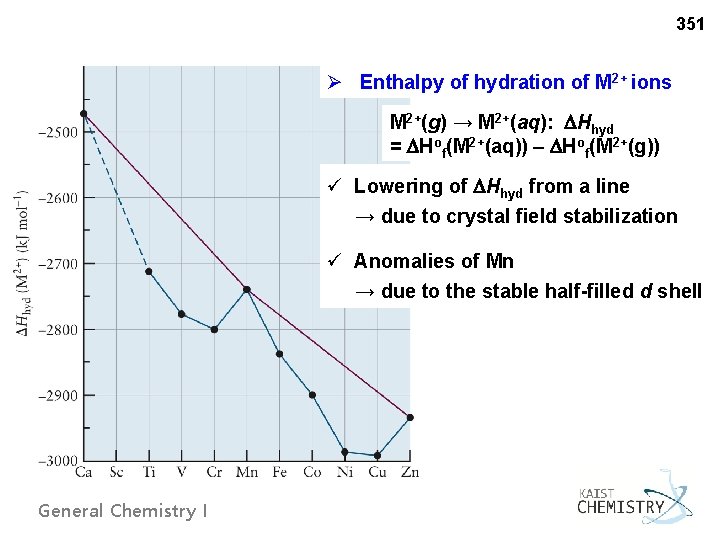
351 Ø Enthalpy of hydration of M 2+ ions M 2+(g) → M 2+(aq): Hhyd = Hof(M 2+(aq)) – Hof(M 2+(g)) ü Lowering of Hhyd from a line → due to crystal field stabilization ü Anomalies of Mn → due to the stable half-filled d shell General Chemistry I

351 Ø Oxidation states more common oxidation state v Increasing tendency toward higher oxidation states among heavier transition elements in the same group: Fe (2, 3) → Ru (2, 3, 4, 6, 8), Ni(2, 3) → Pd(2, 4) General Chemistry I

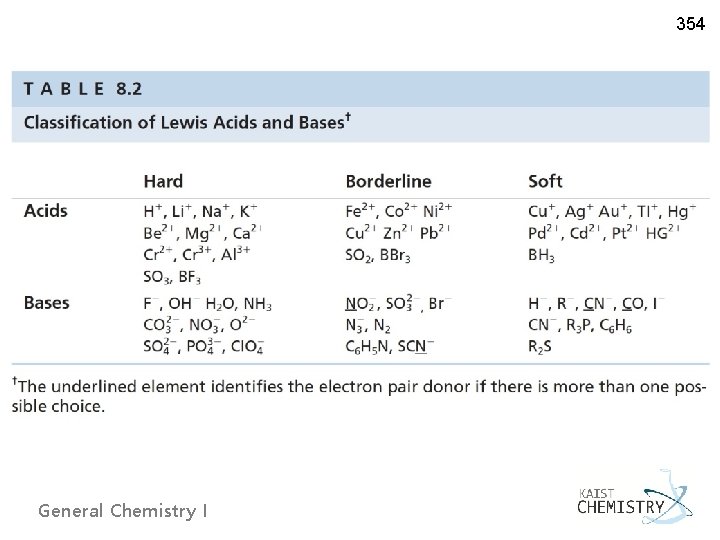
353 Ø Hard and Soft Acids and Bases v Pearson (1963) ~ Extension of Lewis’ definition – electron pair acceptor (acid) and donor (base) – by adding categories ‘hard’ and ‘soft. ’ ~ 'Hard' species: small, high charge states, low electronegativities, weakly polarizable ~ 'Soft' species: large, low charge states, high electronegativities, General Chemistry I Ralph Pearson (US, 1919 - )
354 General Chemistry I

354 Ø Prediction of chemical reactivities of inorganic reactions ~ Preferred direction: hard acid/hard base or soft acid/soft base Hg. F 2(g) + Be. I 2(g) → Be. F 2(g) + Hg. I 2(g) s/h h/s h/h s/s Ag. Br(s) + I–(aq) → Ag. I(s) + Br–(aq) s/b s/s b EXAMPLE 8. 2 Predict whether the following reactions will occur. (a) Ca. F 2(s) + Cd. I 2(s) → Ca. I 2(s) + Cd. F 2(s) (b) Cr(CN)2(s) + Cd(OH)2(s) → Cd(CN)2(s) + Cr(OH)2(s) General Chemistry I NO YES

355 8. 2 INTRODUCTION TO COORDINATION CHEMISTRY Ø Formation of Coordination Complexes v Werner’s investigation: Compound 1: Co. Cl 3 6 NH 3 (orange-yellow) Compound 2: Co. Cl 3 5 NH 3 (purple) Compound 3: Co. Cl 3 4 NH 3 (green) Compound 4: Co. Cl 3 3 NH 3 (green) Alfred Werner (Swiss, 1866 -1919) Nobel prize in chemistry(’ 13) v Treatment with HCl → did not remove NH 3 Ag. NO 3 + Cl- → Ag. Cl(s) in the ratio of 3 : 2 : 1 : 0 General Chemistry I
![356 v Conductivity measurements: Compound 1: [Co(NH 3)6]3+(Cl–)3 → Conductivity of Al(NO 3)3 Compound 356 v Conductivity measurements: Compound 1: [Co(NH 3)6]3+(Cl–)3 → Conductivity of Al(NO 3)3 Compound](http://slidetodoc.com/presentation_image_h/afcb00e7f91369b9d7c01709f99ff232/image-13.jpg)
356 v Conductivity measurements: Compound 1: [Co(NH 3)6]3+(Cl–)3 → Conductivity of Al(NO 3)3 Compound 2: [Co(NH 3)5 Cl]2+(Cl–)2 → Conductivity of Mg(NO 3)2 Compound 3: [Co(NH 3)4 Cl 2]+(Cl–) → Conductivity of Na. NO 3 Compound 4: [Co(NH 3)3 Cl 3] → Nonelectrolyte → Concept of “coordination sphere” around the central metal ion inner and outer coordination sphere → Formation of an octahedral complex In the above complexes, NH 3 and Cl- that are attached to Co are called LIGANDS General Chemistry I
![357 Cu. SO 4∙ 5 H 2 O → [Cu(OH 2)4]SO 4∙H 2 O 357 Cu. SO 4∙ 5 H 2 O → [Cu(OH 2)4]SO 4∙H 2 O](http://slidetodoc.com/presentation_image_h/afcb00e7f91369b9d7c01709f99ff232/image-14.jpg)
357 Cu. SO 4∙ 5 H 2 O → [Cu(OH 2)4]SO 4∙H 2 O General Chemistry I anhydrous Cu. SO 4
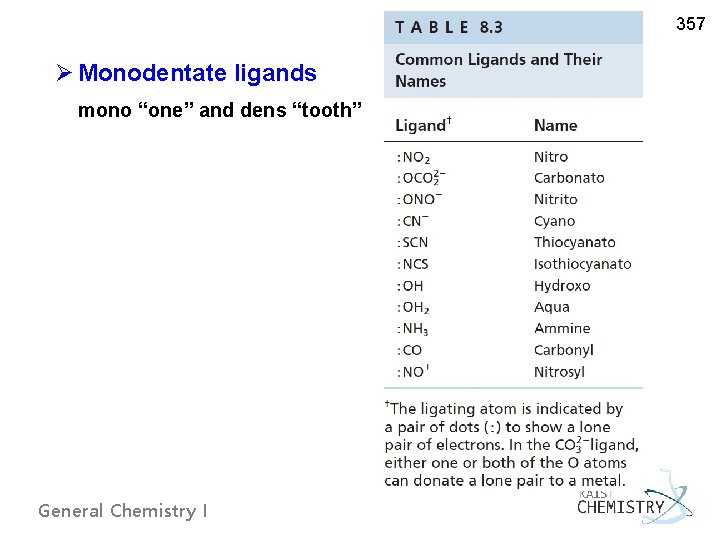
357 Ø Monodentate ligands mono “one” and dens “tooth” General Chemistry I
![358 Ø Bidentate ligands (‘ox’) (‘en’) Ø Chelating ligands: chelate (G. chele, “claw”) [Pt(en)3]4+ 358 Ø Bidentate ligands (‘ox’) (‘en’) Ø Chelating ligands: chelate (G. chele, “claw”) [Pt(en)3]4+](http://slidetodoc.com/presentation_image_h/afcb00e7f91369b9d7c01709f99ff232/image-16.jpg)
358 Ø Bidentate ligands (‘ox’) (‘en’) Ø Chelating ligands: chelate (G. chele, “claw”) [Pt(en)3]4+ ~ 3 bidentates General Chemistry I [Co(EDTA)]– ~ 1 hexadentate
![359 Ø Naming coordination compounds 1) Single word for a coordination complex ~ [prefix-ligand-metal] 359 Ø Naming coordination compounds 1) Single word for a coordination complex ~ [prefix-ligand-metal]](http://slidetodoc.com/presentation_image_h/afcb00e7f91369b9d7c01709f99ff232/image-17.jpg)
359 Ø Naming coordination compounds 1) Single word for a coordination complex ~ [prefix-ligand-metal] 2) Cation first followed by anion ~ K[…] or […]Cl 3) Ending with the suffix “-o” for anionic ligand, chlorido (Cl), no change for neutral ligands except aqua (H 2 O), ammine (NH 3), carbonyl (CO). Note: “chloro” for Cl in a compound ligand 4) Prefixes for the number of ligands ~ di-, tri-, tetra-, penta-, hexa-, … (bis-, tris-, tetrakis-, … for ligands with di- (etc) in their names) 5) Alphabetical ordering for many ligands 6) Roman numeral (oxidation state) in (. . ) after the name of metal ~ […cobalt(III)]Cl or K[…ferrate(III)] anionic complex ions: the ending “-ate” General Chemistry I
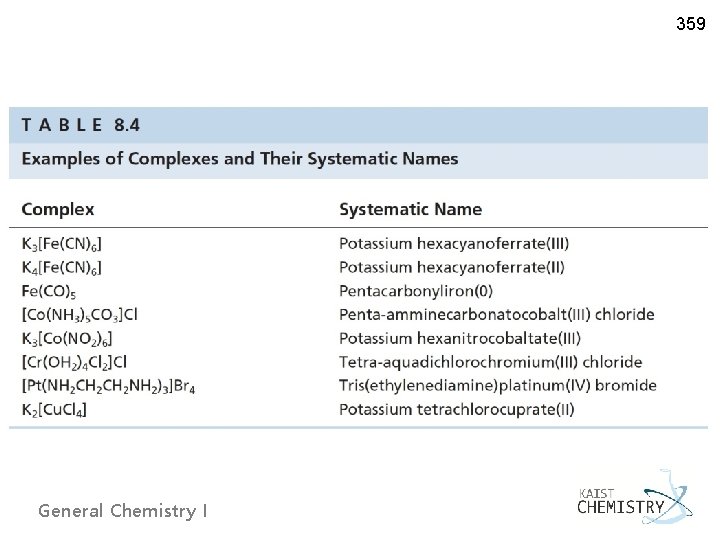
359 General Chemistry I
![Ø Ligand substitution reactions [Ni(OH 2)6]2+(aq) + 6 NH 3(aq) → [Ni(NH 3)6]2+(aq) + Ø Ligand substitution reactions [Ni(OH 2)6]2+(aq) + 6 NH 3(aq) → [Ni(NH 3)6]2+(aq) +](http://slidetodoc.com/presentation_image_h/afcb00e7f91369b9d7c01709f99ff232/image-19.jpg)
Ø Ligand substitution reactions [Ni(OH 2)6]2+(aq) + 6 NH 3(aq) → [Ni(NH 3)6]2+(aq) + 6 H 2 O General Chemistry I 360

Difference between ‘inert’ and ‘labile’ v ‘Inert’ coordination complex: thermodynamically unstable, kinetically stable (inert) takes a week v ‘Labile’ coordination complex: thermodynamically unstable, kinetically unstable (labile) takes a matter of seconds General Chemistry I 361
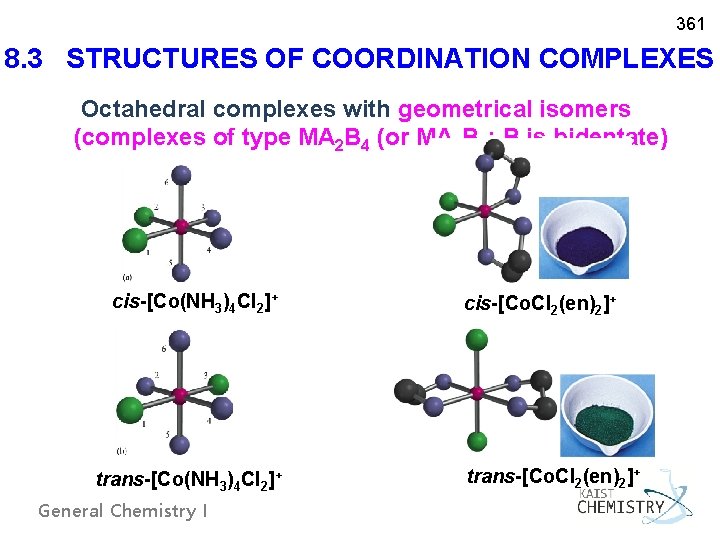
361 8. 3 STRUCTURES OF COORDINATION COMPLEXES Octahedral complexes with geometrical isomers (complexes of type MA 2 B 4 (or MA 2 B 2; B is bidentate) cis-[Co(NH 3)4 Cl 2]+ trans-[Co(NH 3)4 Cl 2]+ General Chemistry I cis-[Co. Cl 2(en)2]+ trans-[Co. Cl 2(en)2]+
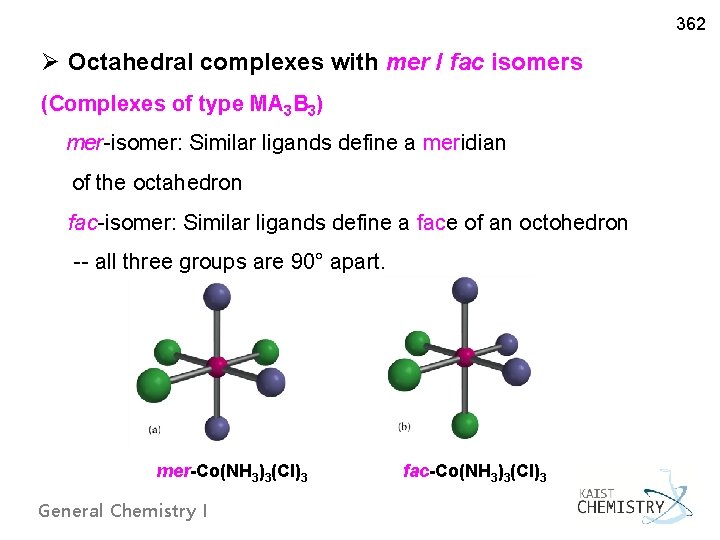
362 Ø Octahedral complexes with mer / fac isomers (Complexes of type MA 3 B 3) mer-isomer: Similar ligands define a meridian of the octahedron fac-isomer: Similar ligands define a face of an octohedron -- all three groups are 90° apart. mer-Co(NH 3)3(Cl)3 fac-Co(NH 3)3(Cl)3 General Chemistry I
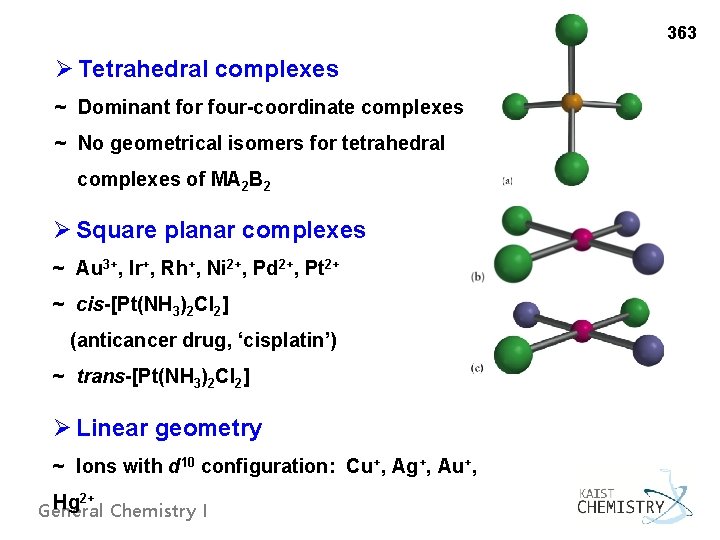
363 Ø Tetrahedral complexes ~ Dominant for four-coordinate complexes ~ No geometrical isomers for tetrahedral complexes of MA 2 B 2 Ø Square planar complexes ~ Au 3+, Ir+, Rh+, Ni 2+, Pd 2+, Pt 2+ ~ cis-[Pt(NH 3)2 Cl 2] (anticancer drug, ‘cisplatin’) ~ trans-[Pt(NH 3)2 Cl 2] Ø Linear geometry ~ Ions with d 10 configuration: Cu+, Ag+, Au+, 2+ Hg General Chemistry I

366 u Chiral Structures Ø Optical isomers are molecules that rotate plane polarized light Ø Enantiomers (Gk. e ά τιος, “opposite”, and μέρος, “part or portion”) are optical isomers whose structures are nonsuperimposable mirror images (they lack reflection-rotation symmetry) Ø Chiral center (chirality [G. χειρ (kheir), "hand"] ~ handedness) is a central atom around which enantiomers are formed Ø A racemic mixture has equal amount of enantiomers (net rotation of plane polarized light = 0) General Chemistry I
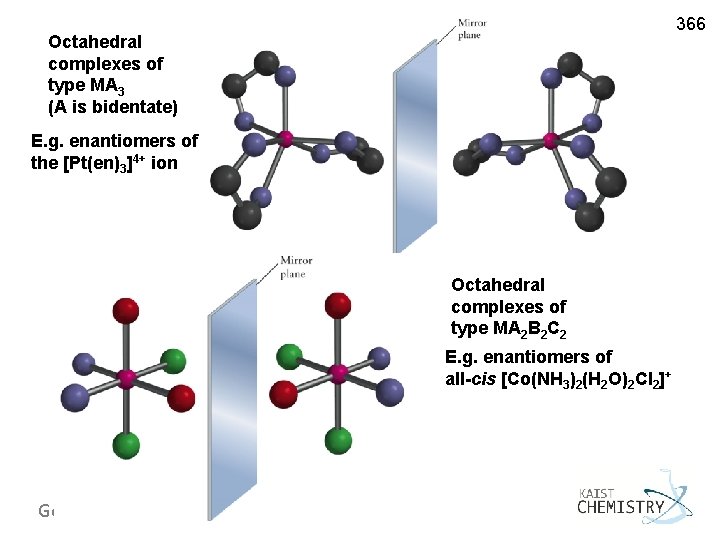
366 Octahedral complexes of type MA 3 (A is bidentate) E. g. enantiomers of the [Pt(en)3]4+ ion Octahedral complexes of type MA 2 B 2 C 2 E. g. enantiomers of + all-cis [Co(NH 3)2(H 2 O)2 Cl 2] General Chemistry I
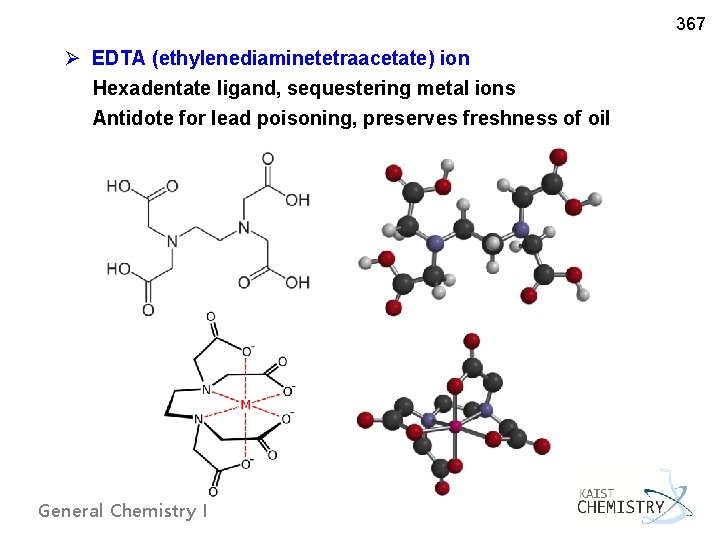
367 Ø EDTA (ethylenediaminetetraacetate) ion Hexadentate ligand, sequestering metal ions Antidote for lead poisoning, preserves freshness of oil General Chemistry I

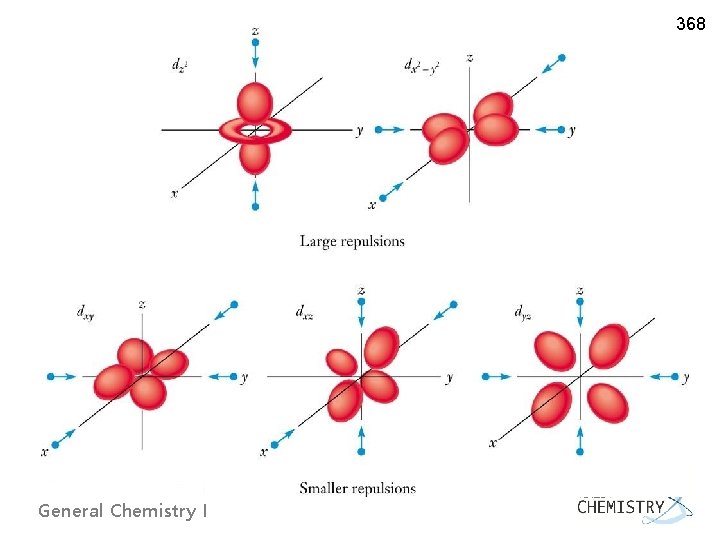
367 8. 4 CRYSTAL FIELD THEORY: OPTICAL AND MAGNETIC PROPERTIES u Crystal Field Theory ~ Ionic description of metal-ligand bonds ~ Ligands are treated as point charges approaching the central metal ion Octahedral coordination complexes ØDegeneracy of d-orbitals lifted into two groups : General Chemistry I
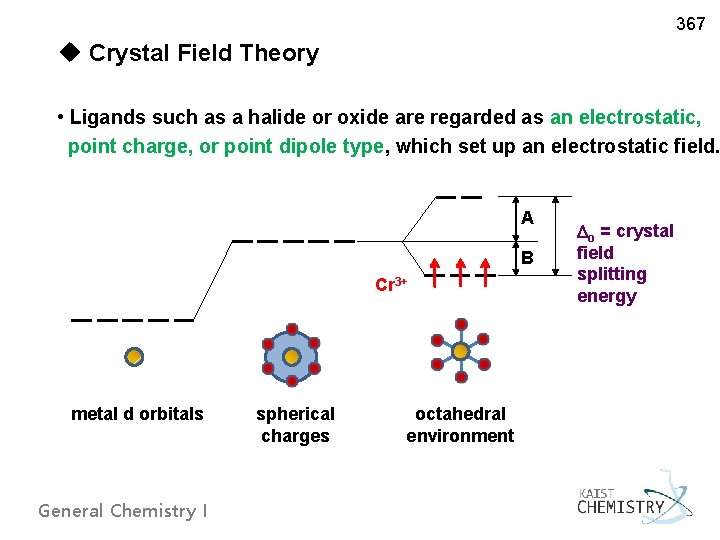
367 u Crystal Field Theory • Ligands such as a halide or oxide are regarded as an electrostatic, point charge, or point dipole type, which set up an electrostatic field. A B Cr 3+ metal d orbitals General Chemistry I spherical charges octahedral environment o = crystal field splitting energy
368 General Chemistry I
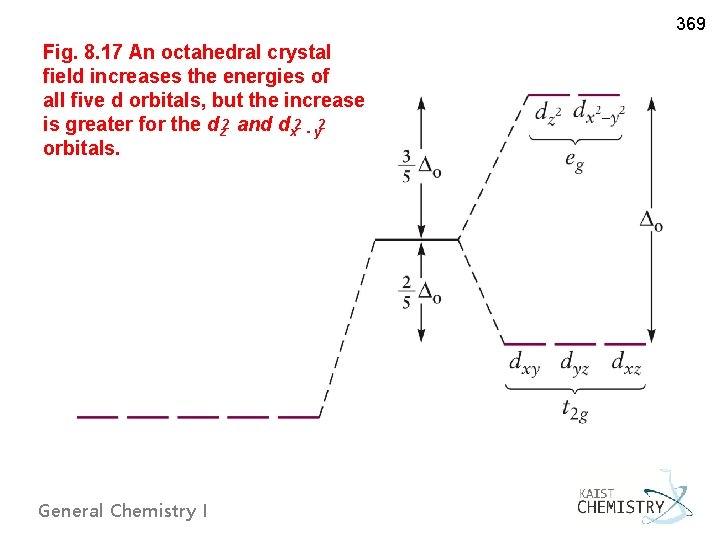
369 Fig. 8. 17 An octahedral crystal field increases the energies of all five d orbitals, but the increase is greater for the dz 2 and dx 2 - y 2 orbitals. General Chemistry I
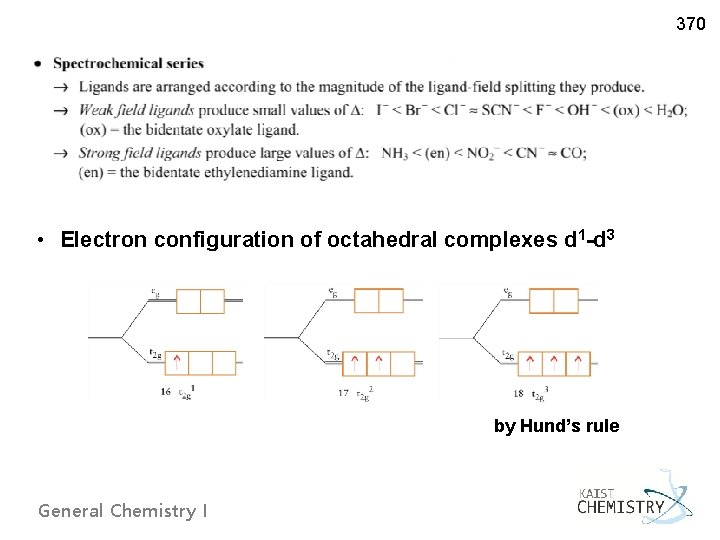
370 • Electron configuration of octahedral complexes d 1 -d 3 by Hund’s rule General Chemistry I
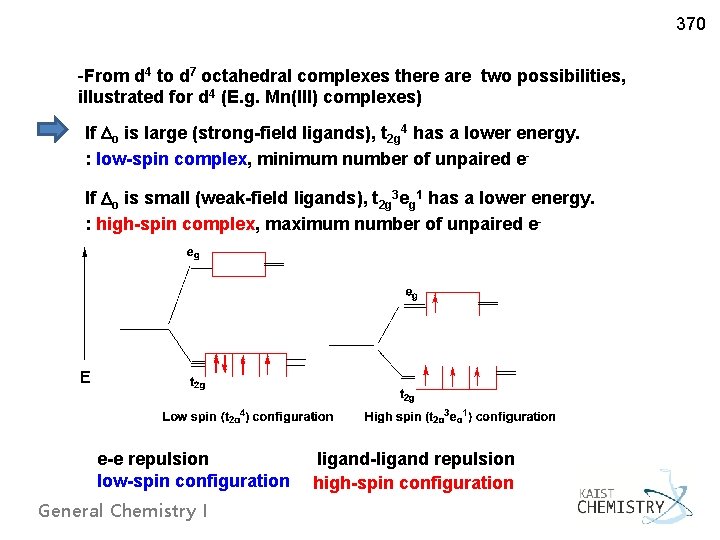
370 -From d 4 to d 7 octahedral complexes there are two possibilities, illustrated for d 4 (E. g. Mn(III) complexes) If o is large (strong-field ligands), t 2 g 4 has a lower energy. : low-spin complex, minimum number of unpaired e. If o is small (weak-field ligands), t 2 g 3 eg 1 has a lower energy. : high-spin complex, maximum number of unpaired e- e-e repulsion low-spin configuration General Chemistry I ligand-ligand repulsion high-spin configuration
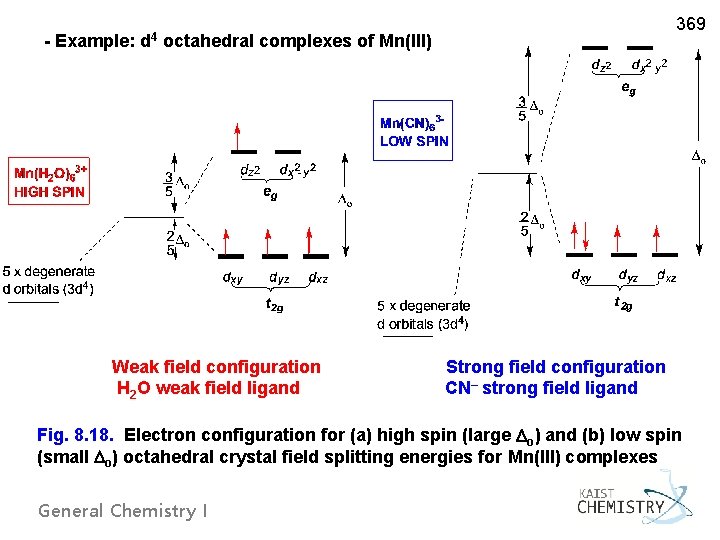
- Example: d 4 octahedral complexes of Mn(III) 369 Weak field configuration Strong field configuration H 2 O weak field ligand CN– strong field ligand Fig. 8. 18. Electron configuration for (a) high spin (large o) and (b) low spin (small o) octahedral crystal field splitting energies for Mn(III) complexes General Chemistry I
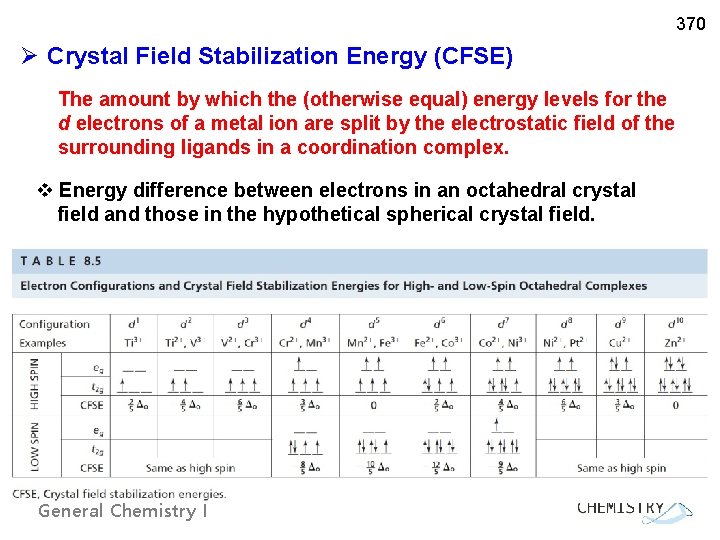
370 Ø Crystal Field Stabilization Energy (CFSE) The amount by which the (otherwise equal) energy levels for the d electrons of a metal ion are split by the electrostatic field of the surrounding ligands in a coordination complex. v Energy difference between electrons in an octahedral crystal field and those in the hypothetical spherical crystal field. General Chemistry I
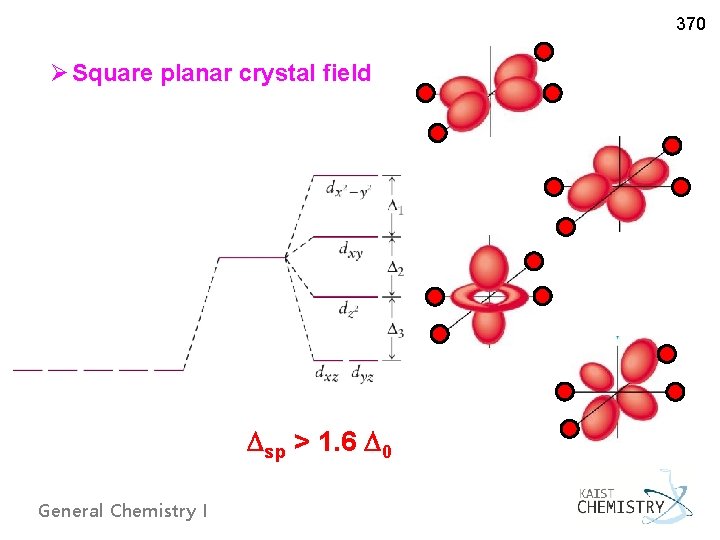
370 Ø Square planar crystal field sp > 1. 6 0 General Chemistry I
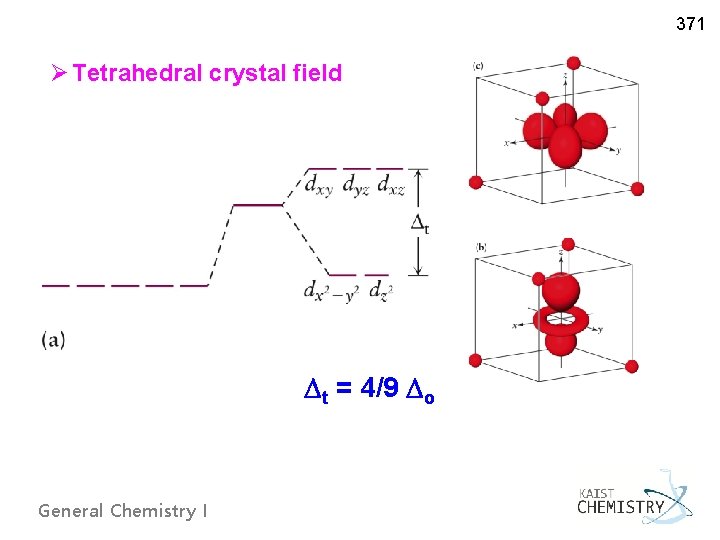
371 Ø Tetrahedral crystal field t = 4/9 o General Chemistry I

372 Fig. 8. 20. Correlation diagram showing the relationships among d-orbital energy levels in crystal fields of different symmetries. General Chemistry I

373 u Magnetic properties Ø Magnetic susceptibility ~ Strength of a sample’s interaction with a magnetic field Ø Paramagnetic compounds ~ One or more unpaired electrons ~ Large, positive magnetic susceptibility ~ Attracted by the magnetic field → “weigh” more ~ Prevalent among transition-metal complexes Ø Diamagnetic compounds ~ All of the electrons are paired ~ Small, negative susceptibility ~ Repelled by the magnetic field General Chemistry I

374 8. 5 OPTICAL PROPERTIES AND THE SPECTROCHEMICAL SERIES Ø Transition-metal complexes ~ absorb visible light → colorful E. g. [Co(NH 3)5 Cl]2+ ion absorbs greenish yellow light (~530 nm) Only red and blue light transmitted → purple (complementary color) Ø Wavelength of the strongest absorption, max d 10 complex ~ colorless (no absorption, all d-levels are filled) High-spin d 5 complex ~ weak absorption (spin flip required) General Chemistry I
![375 Cr(CO)6 [Co(NH 3)5(OH 2)]Cl 3 K 3[Fe(C 2 O 4)3] K 3[Fe(CN)6] [Co(en)3]I 375 Cr(CO)6 [Co(NH 3)5(OH 2)]Cl 3 K 3[Fe(C 2 O 4)3] K 3[Fe(CN)6] [Co(en)3]I](http://slidetodoc.com/presentation_image_h/afcb00e7f91369b9d7c01709f99ff232/image-40.jpg)
375 Cr(CO)6 [Co(NH 3)5(OH 2)]Cl 3 K 3[Fe(C 2 O 4)3] K 3[Fe(CN)6] [Co(en)3]I 3 Colors of the hexaaqua complexes of metal ions prepared from their nitrate salts. E. g. [Co(H 2 O)6]2+ General Chemistry I

376 u Spectrochemical series ~ An ordering of ligands according to their ability to cause crystal field splittings. Ø Spectrochemical series for ligands Ø Spectrochemical series for metal ions Mn 2+ < Ni 2+ < Co 2+ < Fe 3+ < Co 3+ < Mn 4+ < Pd 4+ < Ir 3+ < Pt 4+ v Crystal field theory cannot explain the spectrochemical series! General Chemistry I

377 8. 6 BONDING IN COORDINATION COMPLEXES u Valence bond theory Ø dsp 3 hybrid orbitals ~ linear combination of one s, three p atomic orbitals and the dz 2 atomic orbital ~ five equivalent new hybrid orbitals ~ trigonal bipyramid, PF 5, Cu. Cl 5– General Chemistry I
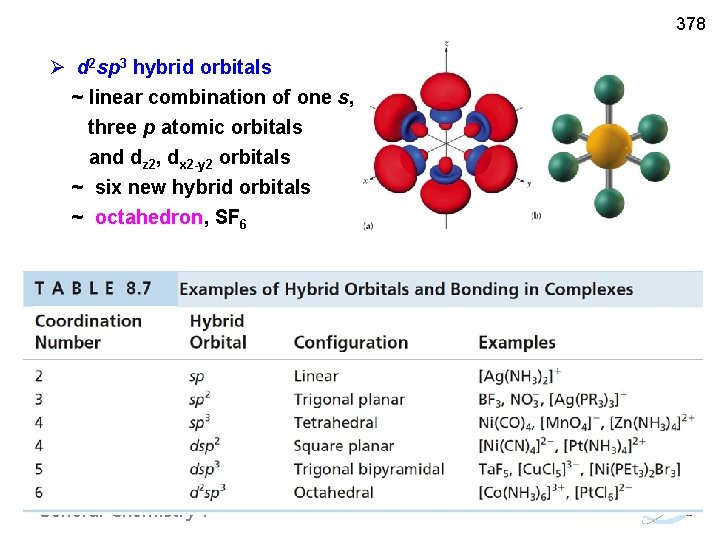
378 Ø d 2 sp 3 hybrid orbitals ~ linear combination of one s, three p atomic orbitals and dz 2, dx 2 -y 2 orbitals ~ six new hybrid orbitals ~ octahedron, SF 6 General Chemistry I

u Molecular orbital theory 378 Ø Ligand field theory ~ Failure of CFT and VB theories to explain the spectrochemical series ~ MO description for ligands Ø Construction of MOs for octahedral complexes (of 1 st row D-block metals) ~ Interaction between the metal 4 s orbital with six ligand orbitals → s and s* orbitals ~ Interaction between three metal p orbitals with three ligand orbitals → triply degenerate p and p* orbitals ~ Interaction of the dz 2 and dx 2 -y 2 orbitals with ligand orbitals → a pair of d and d* orbitals General Chemistry I
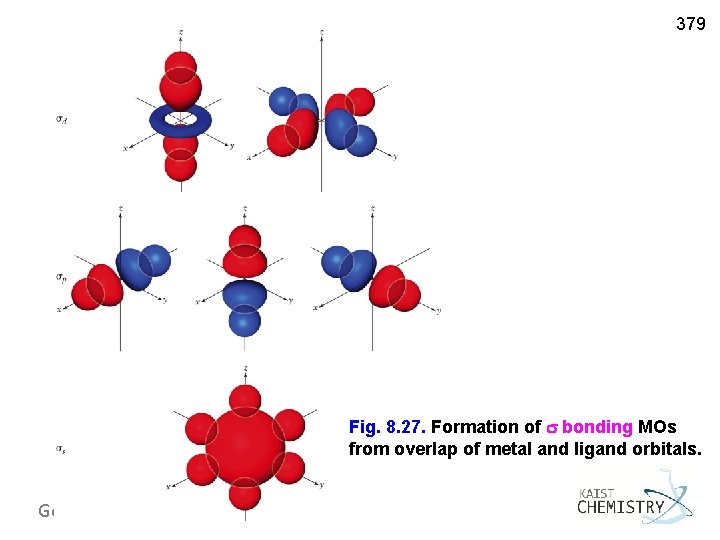
379 Fig. 8. 27. Formation of bonding MOs from overlap of metal and ligand orbitals. General Chemistry I
![380 Antibonding MOs MO correlation diagram for octahedral Cr(III) complex ([Cr. Cl 6]3 -): 380 Antibonding MOs MO correlation diagram for octahedral Cr(III) complex ([Cr. Cl 6]3 -):](http://slidetodoc.com/presentation_image_h/afcb00e7f91369b9d7c01709f99ff232/image-46.jpg)
380 Antibonding MOs MO correlation diagram for octahedral Cr(III) complex ([Cr. Cl 6]3 -): bonding only Nonbonding MOs Bonding MOs General Chemistry I
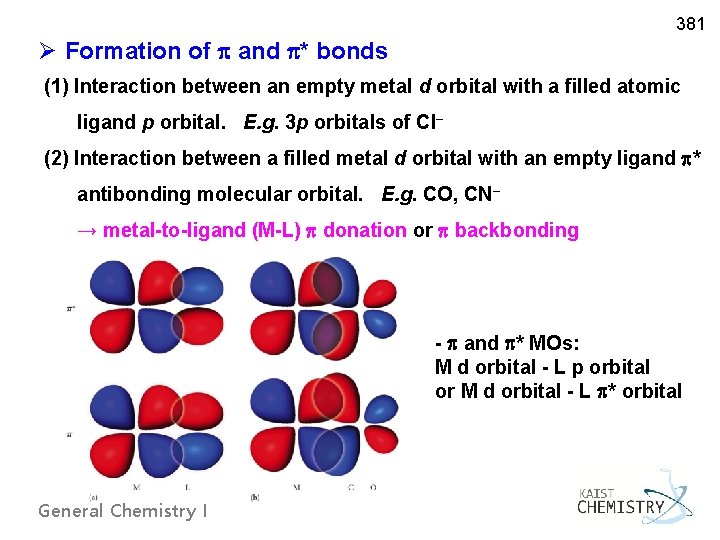
381 Ø Formation of and * bonds (1) Interaction between an empty metal d orbital with a filled atomic ligand p orbital. E. g. 3 p orbitals of Cl– (2) Interaction between a filled metal d orbital with an empty ligand * antibonding molecular orbital. E. g. CO, CN– → metal-to-ligand (M-L) donation or backbonding - and * MOs: M d orbital - L p orbital or M d orbital - L * orbital General Chemistry I
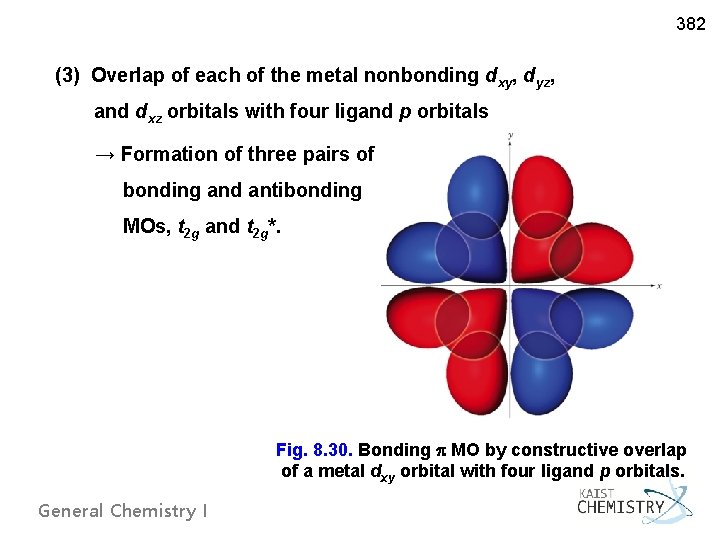
382 (3) Overlap of each of the metal nonbonding dxy, dyz, and dxz orbitals with four ligand p orbitals → Formation of three pairs of bonding and antibonding MOs, t 2 g and t 2 g*. Fig. 8. 30. Bonding MO by constructive overlap of a metal dxy orbital with four ligand p orbitals. General Chemistry I

383 Ø Order of bonding strengths for different ligands v Weak-field ligands (small o) → Overlap between occupied p( ) bonding orbitals of ligands (Br–, Cl–, CO) with t 2 g orbitals of metal → Increase in energy of t 2 g and decrease in o v Strong-field ligands (large o) → Overlap between unoccupied * antibonding orbitals of ligands (CO, CN–) with t 2 g orbitals of metal → Lowering of energy of t 2 g orbitals by back-bonding (M→L) v Intermediate-field ligands ~ H 2 O, NH 3 General Chemistry I
![383 (b) (a) Fig. 8. 31. (a) (M L) [or (b) (M L)] donation 383 (b) (a) Fig. 8. 31. (a) (M L) [or (b) (M L)] donation](http://slidetodoc.com/presentation_image_h/afcb00e7f91369b9d7c01709f99ff232/image-50.jpg)
383 (b) (a) Fig. 8. 31. (a) (M L) [or (b) (M L)] donation showing a reduction (or increase) in Δo compared with that from bonding alone. (a) Slight increase in energy of t 2 g electrons (in t 2 g* MOs) (b) Significant lowering in energy of t 2 g electrons due to back-bonding → Electrons of t 2 g MOs are delocalised into unoccupied *(L) General Chemistry I
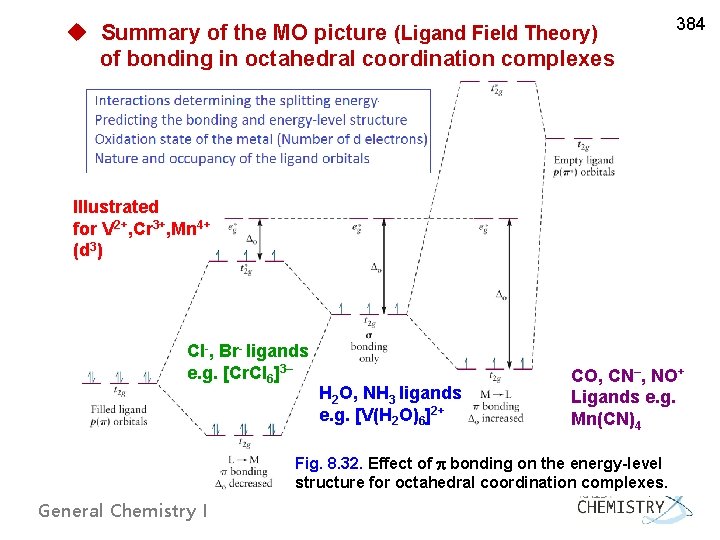
u Summary of the MO picture (Ligand Field Theory) of bonding in octahedral coordination complexes 384 IIlustrated for V 2+, Cr 3+, Mn 4+ (d 3) Cl-, Br- ligands e. g. [Cr. Cl 6]3– H 2 O, NH 3 ligands e. g. [V(H 2 O)6]2+ CO, CN–, NO+ Ligands e. g. Mn(CN)4 Fig. 8. 32. Effect of bonding on the energy-level structure for octahedral coordination complexes. General Chemistry I

10 Problem Sets For Chapter 8, 2, 8, 18, 26, 32, 44, 46, 58, 64, 66 General Chemistry I
- Slides: 52