8 4 Chemical Equilibrium Objectives S 4 ad

8. 4 Chemical Equilibrium Objectives S 4: a-d; 5 Section 1 - chemistry

Essential Questions Ø What is a reversible reaction? Ø What is a chemical equilibrium? Ø How does a reaction(s) come to equilibrium? Ø What does Le Chatelier’s Principle say? Ø What are ways to affect chemical equilibrium?

Reversible Reactions Ø What is a reversible reaction? l A reaction that can go backwards so that products are reforming as reactants. Example p 552: Forward reaction Reverse Reaction 2 NO 2 (g) → N 2 O 4 (g) → 2 NO 2 (g) Ø Write reversible RXN using a double headed arrow.

Reversible Reactions (cont)

18. 2 Reversible Reactions (don’t need to write SO 2 and O 2 react to give SO 3 decomposes to SO 2 and O 2 At equilibrium, all three types of molecules are present.

Chemical Equilibrium Ø Any system involving a reversible reaction will reach equilibrium if left alone Ø Chemical equilibrium is when the rate of forward reaction equals the rate of reverse reaction Ø Concentration of reactants and products stay constant because they are being formed as soon as they are being consumed
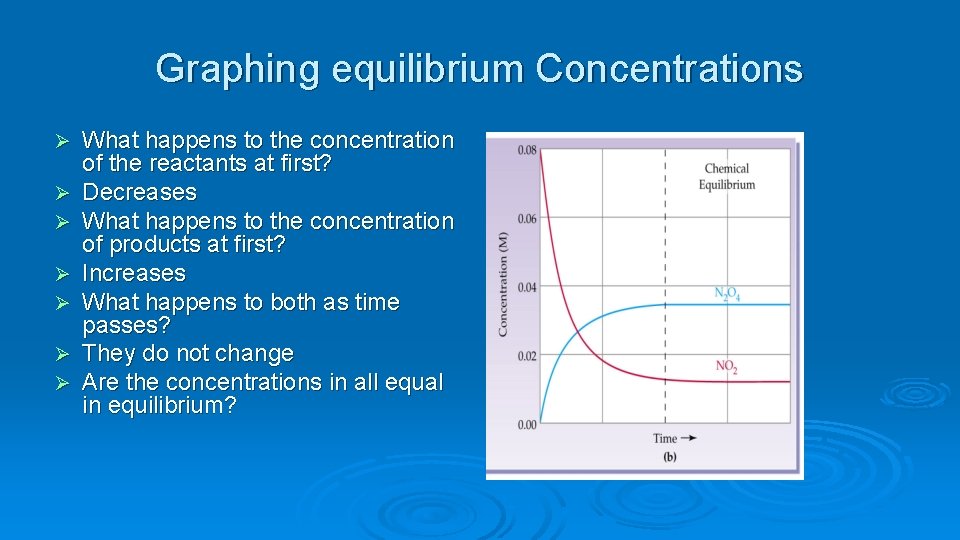
Graphing equilibrium Concentrations Ø Ø Ø Ø What happens to the concentration of the reactants at first? Decreases What happens to the concentration of products at first? Increases What happens to both as time passes? They do not change Are the concentrations in all equal in equilibrium?
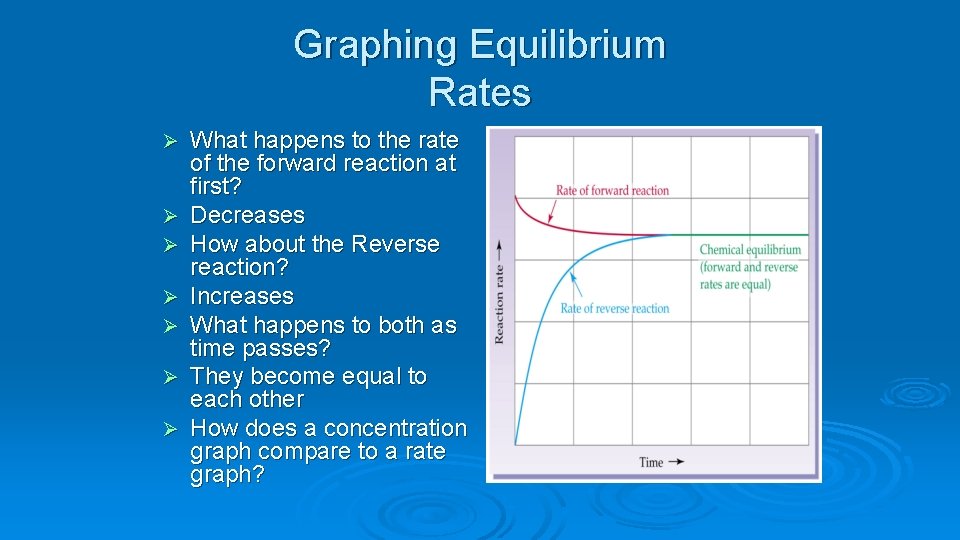
Graphing Equilibrium Rates Ø Ø Ø Ø What happens to the rate of the forward reaction at first? Decreases How about the Reverse reaction? Increases What happens to both as time passes? They become equal to each other How does a concentration graph compare to a rate graph?

Le Chatelier’s Principle (don’t need to write) Ø Ø Ø How does a see-saw work? Weight being placed on one end causes the see-saw to move as it tries to get balanced How do you think that principle applies to a chemical equilibrium? Ø When there is a change in conditions, the chemical equilibrium will shift like a see-saw to maintain balance

Le Chatelier’s Principle (cont) Ø Le Chatelier’s Principle states that when conditions of a chemical equilibrium are changed the equilibrium will shift towards the direction that will reduce that change Ø The 3 conditions that affect equilibrium are concentration of reactants/products, pressure, or temperature Ø Adding a catalyst affects each reaction equally and no overall impact

Changes in Concentration Ø When you add concentration the reaction shifts to the opposite side Ø When you remove the reaction shifts to the same side Ø What will happen if NO 2 is added? Ø What will happen if N 2 O 4 is taken away?

Changes in Pressure For gasses if you increase pressure then volume decreases causing the equilibrium to shift to side with fewest number of gas molecules so they will take up less space N 2 (g) + 3 H 2 (g) ↔ 2 NH 3 (g) Ø If I wanted more ammonia should I increase or decrease the pressure? Ø

Changes in Temperature Ø Depends on if reaction is Endothermic or Exothermic Ø Treat heat term as a reactant (left side) for endothermic and as product (right side) for exothermic

Changes in Temperature (cont) Ø For endothermic reaction which direction will the equilibrium shift if heat is added? l To the right (products) Ø How would removing heat affect an l endo? To the left (reactants) Ø How about an exothermic when heated? l To the left (reactants) Ø How would removing heat affect an exo? l To the right (products)

Changes in Temperature Ø Example: H 2 (g) + I 2 (g) ↔ 2 HI (g) + heat Ø What will happen if you increase the temperature? Ø If I wanted more HI, what would you change?

Essential Questions Ø What is a reversible reaction? Ø What is a chemical equilibrium? Ø How does a reaction(s) come to equilibrium? Ø What does Le Chatelier’s Principle say? Ø What are ways to affect chemical equilibrium?

8. 4 Tracked Assignment Ø worksheet
- Slides: 17