8 2 Solubility and Concentration Key Concepts How

8. 2 Solubility and Concentration Key Concepts • How are solutions with different amounts of solute described? • What factors determine the solubility of a solute? • What are three ways to measure the concentration of a solution?

Solubility maximum • The ________ amount of solute that dissolves in a given amount of solvent at a constant temperature is called ________ solubility • Solubilities are usually expressed grams of solute per in _______ solvent 100 grams of ______
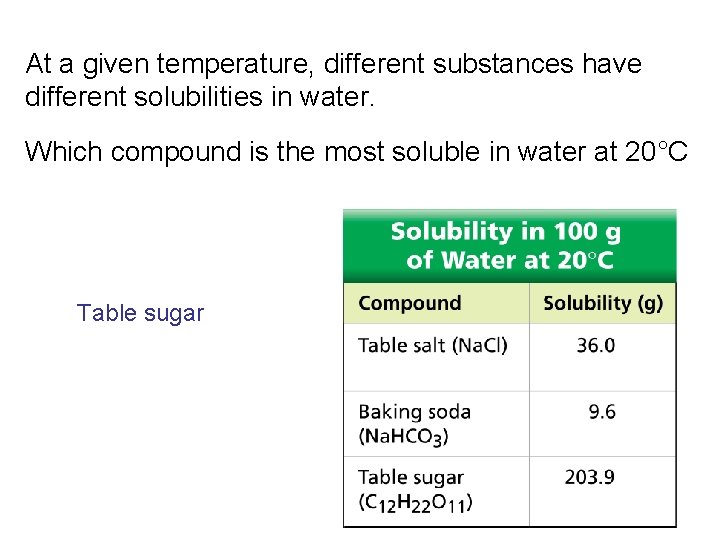
At a given temperature, different substances have different solubilities in water. Which compound is the most soluble in water at 20°C Table sugar

Unsaturated Versus Saturated less • If the amount of solute dissolved is _______ than the maximum that could be dissolved, the unsaturated solution is called an _______ solution less – Ex: Earth’s oceans contain _______ dissolved salt than they can hold

maximum amount of solute • A solution that holds the ______ per amount of solution under the given conditions is saturated solution called a _______ 25% salt – Ex: The Dea Sea has a salinity of ______ 3. 5 compared to only about ______ percent for ocean water more • ________ solute Supersaturated solutions contain ____ than the usual maximum amount and are typically unstable _______ – Ex: While making fudge, you heat a highly concentrated mixture of sugar, chocolate and a water-based solvent to a high _____ enough temperature to make a _____ solution that is supersaturated sugar

Like Dissolves Like • Water can dissolve an enormous variety of NOT dissolve substances, but it does ______ everything • The phrase that scientists often use when “like dissolves like” predicting solubility is ___________ • This phrase means that dissolving occurs anytime a solute and solvent have _______ in polarity similarities

Practice Solubility Questions! • Can water dissolve other polar molecules? Yes…both are polar • Can water dissolve other compounds that are similar to it in structure and hydrogen bonding? Yes…similar structure and bonding • Can water dissolve a nonpolar molecule like oil? No…not similar or “unlike” • Can oil dissolve other nonpolar molecules? Yes…. both are nonpolar

Special Topic: Soap • Soap has unique properties because one end of the molecule is ______ and the other polar end is _______ nonpolar • The polar end attracts ______ and the water oil nonpolar end attracts ______ dissolves • Basically, the nonpolar end ______ the oil and the polar end interacts with water to rinse ______ the oil away
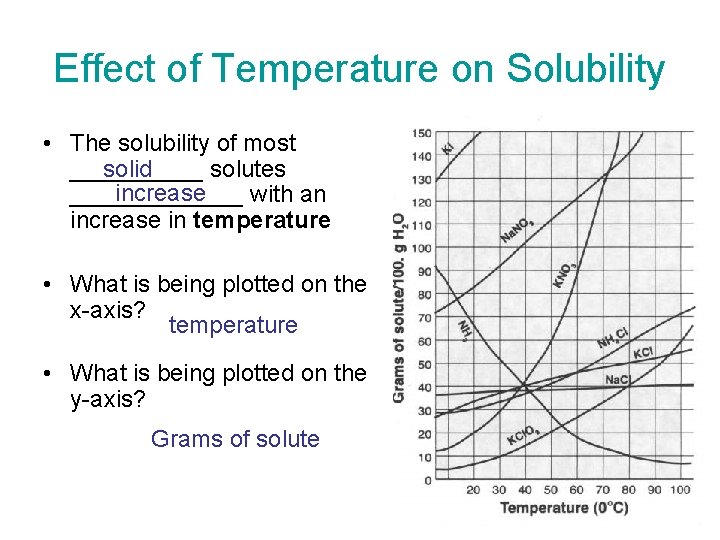
Effect of Temperature on Solubility • The solubility of most _____ solutes solid increase _______ with an increase in temperature • What is being plotted on the x-axis? temperature • What is being plotted on the y-axis? Grams of solute
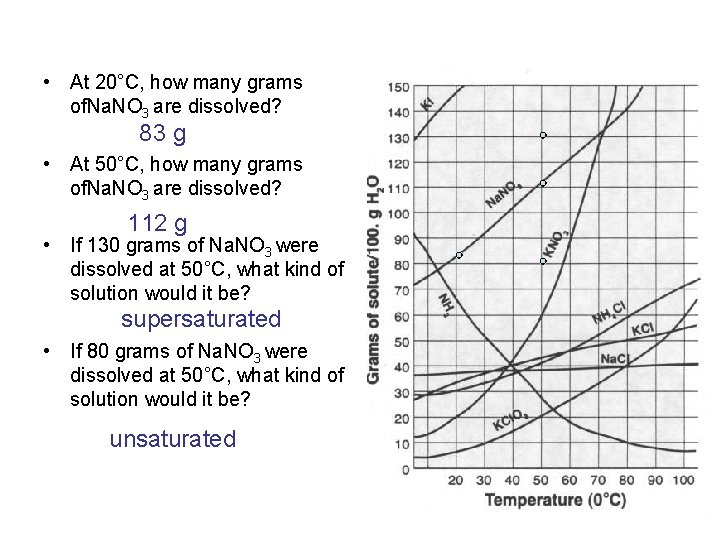
• At 20°C, how many grams of. Na. NO 3 are dissolved? 83 g • At 50°C, how many grams of. Na. NO 3 are dissolved? 112 g • If 130 grams of Na. NO 3 were dissolved at 50°C, what kind of solution would it be? supersaturated • If 80 grams of Na. NO 3 were dissolved at 50°C, what kind of solution would it be? unsaturated

Solubility of Gases • The solubility of a _____ in a liquid actually gas decreases with increases in temperature _______ • Due to this many fish must live in _______ cold water so that the concentration of dissolved oxygen ______ remains high • Think of soda in a bottle…does it taste more carbonated (bubbly) when it is cold or hot? Cold…contains more CO 2

• Pressure also affects the solubility of a gas… more • Increasing pressure allows ____ carbon dioxide to dissolve in a liquid • Think of soda in a bottle again…is it more carbonated with or without the cap on? With the cap…more pressure

Molarity • Suppose you are hospital or pharmacy worker, and you need to make a salt solution that matches the salt concentration of a _______ blood patient’s dilute • Blood is a ______ solution, but this term gives only _________ information qualitative • You will need to measure things accurately using numbers or quantitative _________ information

• Concentration units can ____ greatly. vary • For chemistry applications, the concentration molarity term ______ is generally the most useful M • Molarity, symbolized with an ______, is defined moles as the number of _____ of solute per _____ of solution liter Molarity = _moles of solute_ liter of solution volume • Notice that the _______ in liters is the solution total volume for the _______

• Concentration can also be expressed as volume percent by _______ – Example: Fruit juice labels – What percentage of cranberrry juice is found in the product shown? 27%

Concentration of Solutions Percent by Mass Concentration expressed as a percent by ______ is more useful when the solute is a mass solid.

Review Question 1 • Explain the difference between an unsaturated, and supersaturated solution Contains less than the max. amount of dissolved solute Contains more than the max. amount of dissolved solute

Review Question 2 • How does temperature affect solubility for most solid solutes? As temp. increases, so does solubility • And for gases dissolved in a liquid? As temp. increases, solubility decreases

Review Question 3 • What is the equation for molarity? M = _moles of solute_ liter of solution
- Slides: 19