8 1 Organisms metabolism transforms matter energy subject

8. 1 Organism’s metabolism transforms matter & energy, subject to the laws of thermodynamics I. Metabolism A. Sum of all chemical Rxn. in an organism 1. Reactants 2. Products B. Metabolic pathway 1. A series of defined steps in which a specific molecule is altered to form a specific product a. Each step catalyzed by enzymes b. Must be controlled 1. Prevents excess & depletion 2. What controls? a. Catabolic 1. Breakdown pathway 2. Cellular respiration

3. Released energy available to do work b. Anabolic pathway 1. Biosynthetic pathway 2. Consumes energy to create c. Catabolic fuels the anabolic C. Energy is central to metabolic pathways II. Forms of energy A. Terminology 1. Energy a. Capacity to cause change 2. Kinetic energy a. Energy of motion b. Heat/thermal energy 3. Potential energy a. Energy possessed by matter because of location or structure b. Chemical energy 1. Potential energy stored in molecules due to arrangement of atoms

III. The laws of energy transformation A. 1 st law of thermodynamics 1. Energy can be transformed or transferred, but it cannot be created or destroyed B. 2 nd law of thermodynamics 1. Every energy transfer or transformation increases entropy a. Indicates the relative amount of disorginization b. the mov’t from order disorder, high energy low energy 2. Every energy transfer or transformation results in a loss of usable energy a. Heat increases entropy 3. The natural processes of the universe are spontaneous a. Spontaneous processes increase entropy C. Cells & entropy 1. Processes that occur in cells are energy transformations

2. How do cells bring about order from disorder a. Input of energy 1. Cells rely upon a constant input of energy to offset entropy a. Where does the energy come from? b. Cells are a temporary repository of order purchased at the cost of a constant flow of energy c. Constant balance between catabolic and anabolic pathways 1. What process ultimately “wins” 8. 2 The free energy change of a reaction tells us whether or not the reaction occurs spontaneously I. Intro A. Laws of thermodynamics 1. Apply to universe “system” + “surroundings” B. Free energy 1. Allows us to look at system & remove surroundings

II. Free energy change, ∆G A. Free energy 1. Amount of energy that can be used to do work after a rxn has taken place 2. ∆G a. Allows us to determine whether a rxn is spontaneous or nonspontaneous 1. –∆G= Spontaneous 2. +∆G= nonspontaneous b. ∆G= G final state– G initial state 3. Based on free energy changes, rxn can be classified a. Exergonic rxn & endergonic rxn
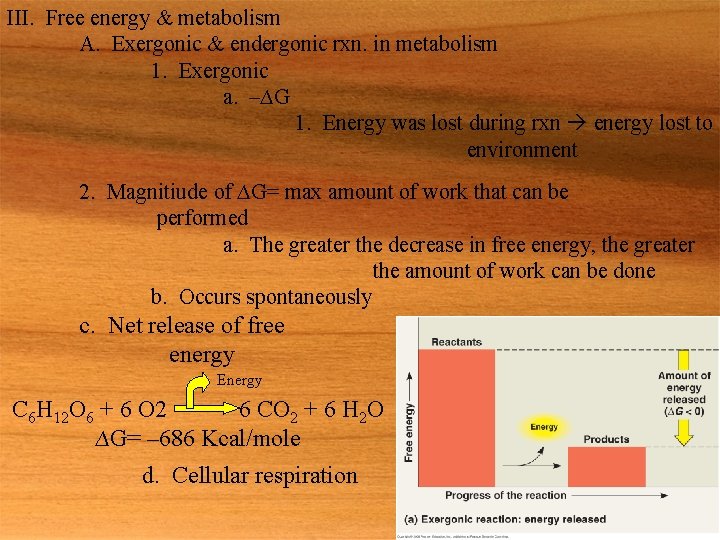
III. Free energy & metabolism A. Exergonic & endergonic rxn. in metabolism 1. Exergonic a. –∆G 1. Energy was lost during rxn energy lost to environment 2. Magnitiude of ∆G= max amount of work that can be performed a. The greater the decrease in free energy, the greater the amount of work can be done b. Occurs spontaneously c. Net release of free energy Energy C 6 H 12 O 6 + 6 O 2 6 CO 2 + 6 H 2 O ∆G= – 686 Kcal/mole d. Cellular respiration
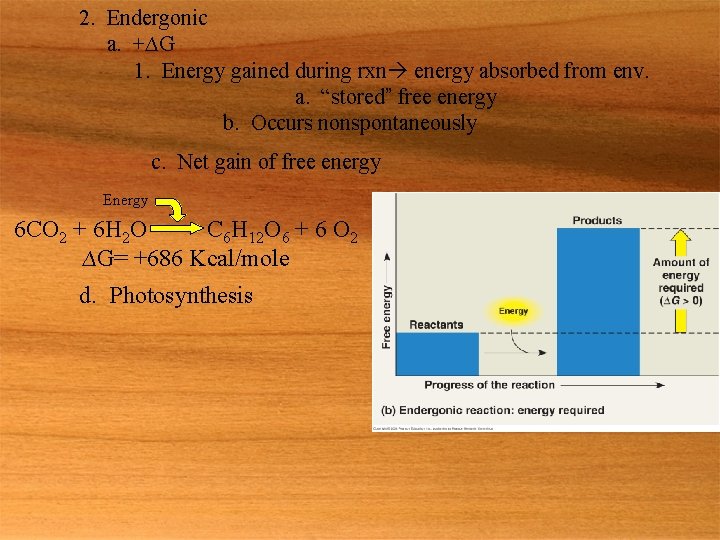
2. Endergonic a. +∆G 1. Energy gained during rxn energy absorbed from env. a. “stored” free energy b. Occurs nonspontaneously c. Net gain of free energy Energy 6 CO 2 + 6 H 2 O C 6 H 12 O 6 + 6 O 2 ∆G= +686 Kcal/mole d. Photosynthesis

B. Equalibrium & metabolism 1. Equalibrium = 0 ∆G = death 2. Cells never reach equalibrium a. Why? 1. 0∆G = No energy = No work = No manufacturing 2. Constant flow of material in & out a. Products of one rxn become reactants of another 1. Product does not accumulate

b. How? 1. Catabolic (exergonic) offset by anabolic (endergonic) free energy c. A constant supply of free energy is necessary 8. 3 ATP powers cellular work by coupling exergonic & endergonic reactions I. Intro A. Types of work by cell 1. Chemical 2. Transport 3. Mechanical B. Energy coupling 1. ATP is the coupler
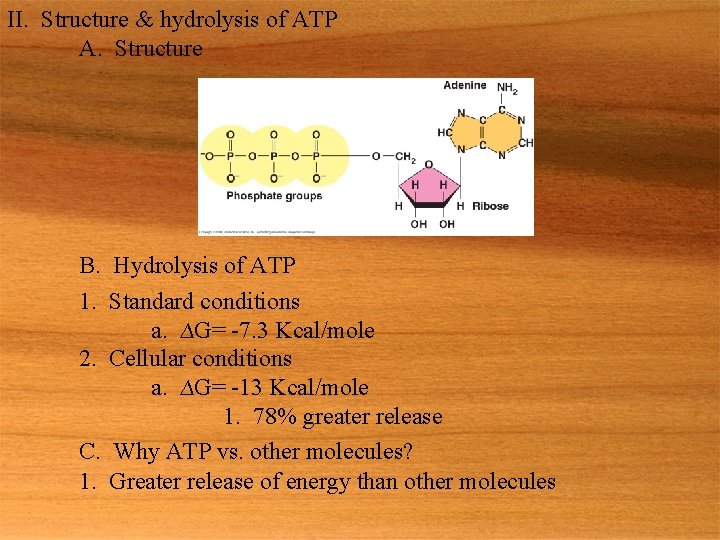
II. Structure & hydrolysis of ATP A. Structure B. Hydrolysis of ATP 1. Standard conditions a. ∆G= -7. 3 Kcal/mole 2. Cellular conditions a. ∆G= -13 Kcal/mole 1. 78% greater release C. Why ATP vs. other molecules? 1. Greater release of energy than other molecules

III. How ATP performs work A. Chemical work 1. Exergonic coupled with endergonic a. Only if ∆G of endergonic is < energy release by ATP hydrolysis b. Overall coupled rxn is negative
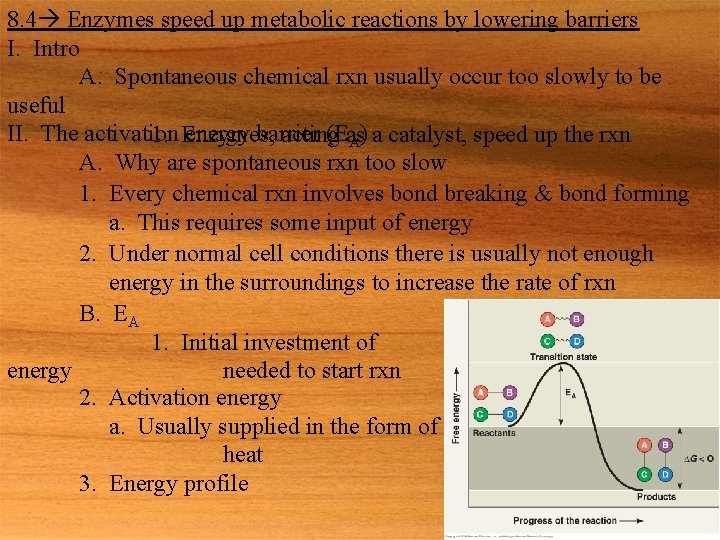
8. 4 Enzymes speed up metabolic reactions by lowering barriers I. Intro A. Spontaneous chemical rxn usually occur too slowly to be useful II. The activation energy barrier (Eas 1. Enzymes, acting A) a catalyst, speed up the rxn A. Why are spontaneous rxn too slow 1. Every chemical rxn involves bond breaking & bond forming a. This requires some input of energy 2. Under normal cell conditions there is usually not enough energy in the surroundings to increase the rate of rxn B. EA 1. Initial investment of energy needed to start rxn 2. Activation energy a. Usually supplied in the form of heat 3. Energy profile
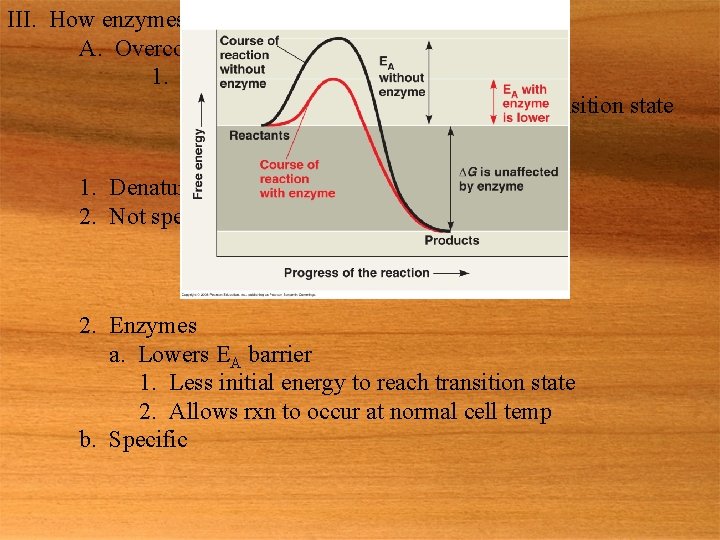
III. How enzymes lower the EA barrier A. Overcoming EA 1. Heat a. Will allow reactants to reach transition state 1. Increase rate of rxn b. Problem 1. Denatures proteins 2. Not specific 2. Enzymes a. Lowers EA barrier 1. Less initial energy to reach transition state 2. Allows rxn to occur at normal cell temp b. Specific
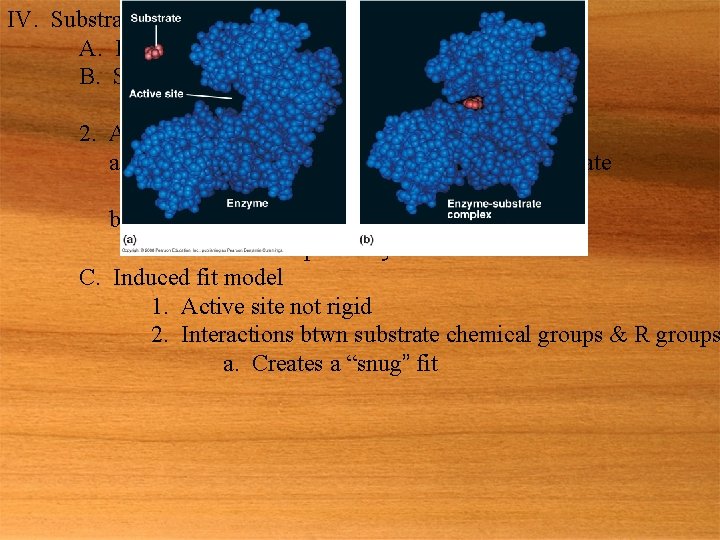
IV. Substrate specificity of enzymes A. E+S ES E+P B. Specificity 1. Arises from enzyme shape 2. Active site a. Specific region of enzyme that binds w/ substrate 1. Usually only a few AA w/in sequence b. Complimentary in shape to substrate 1. Provides the specificity C. Induced fit model 1. Active site not rigid 2. Interactions btwn substrate chemical groups & R groups a. Creates a “snug” fit
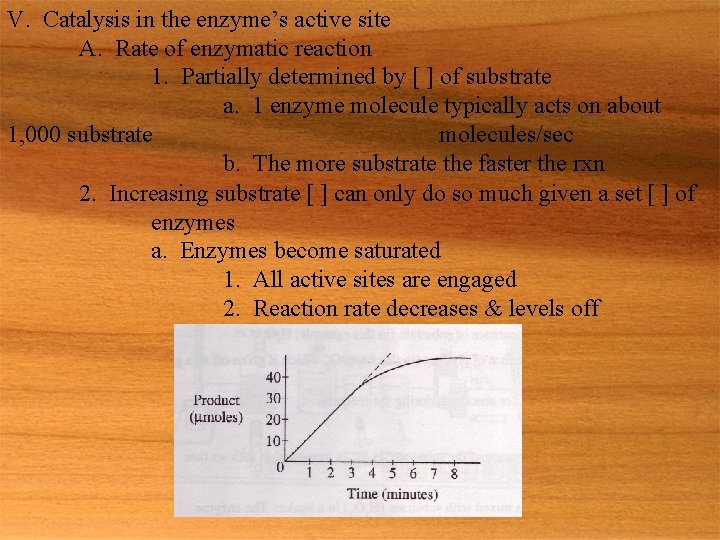
V. Catalysis in the enzyme’s active site A. Rate of enzymatic reaction 1. Partially determined by [ ] of substrate a. 1 enzyme molecule typically acts on about 1, 000 substrate molecules/sec b. The more substrate the faster the rxn 2. Increasing substrate [ ] can only do so much given a set [ ] of enzymes a. Enzymes become saturated 1. All active sites are engaged 2. Reaction rate decreases & levels off
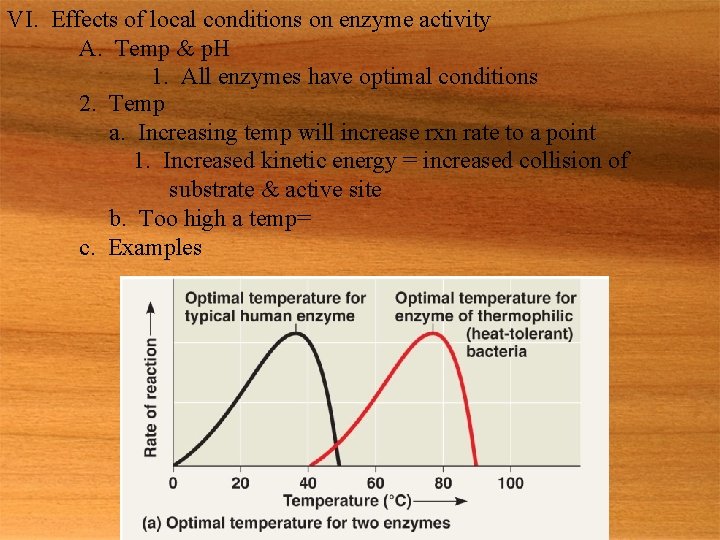
VI. Effects of local conditions on enzyme activity A. Temp & p. H 1. All enzymes have optimal conditions 2. Temp a. Increasing temp will increase rxn rate to a point 1. Increased kinetic energy = increased collision of substrate & active site b. Too high a temp= c. Examples
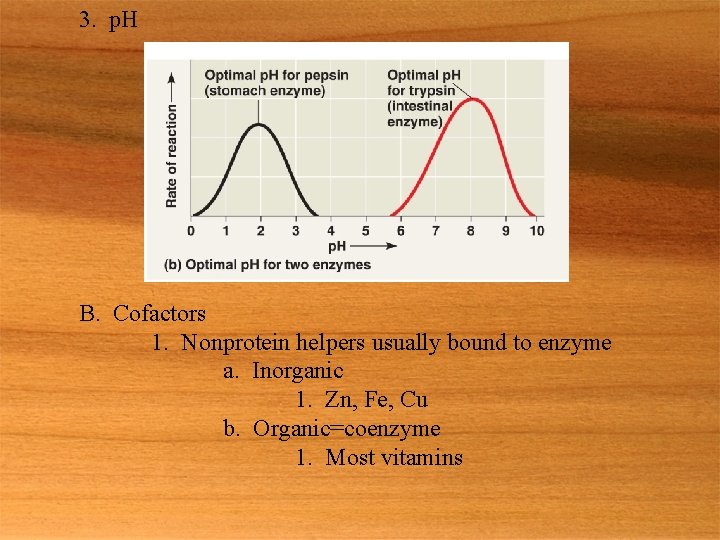
3. p. H B. Cofactors 1. Nonprotein helpers usually bound to enzyme a. Inorganic 1. Zn, Fe, Cu b. Organic=coenzyme 1. Most vitamins

C. Enzyme inhibitors 1. Molecules that selectively inhibit enzyme activity a. Bond to enzyme making inactive 1. Weak bond= reversible inhibition 2. Covalent bond= irreversible inhibition 2. Reversible a. Competitive inhibitors 1. Mimic substrate & compete w/ substrate for active site 2. Decrease enzyme activity 3. Can be overcome by increasing substrate [ ]
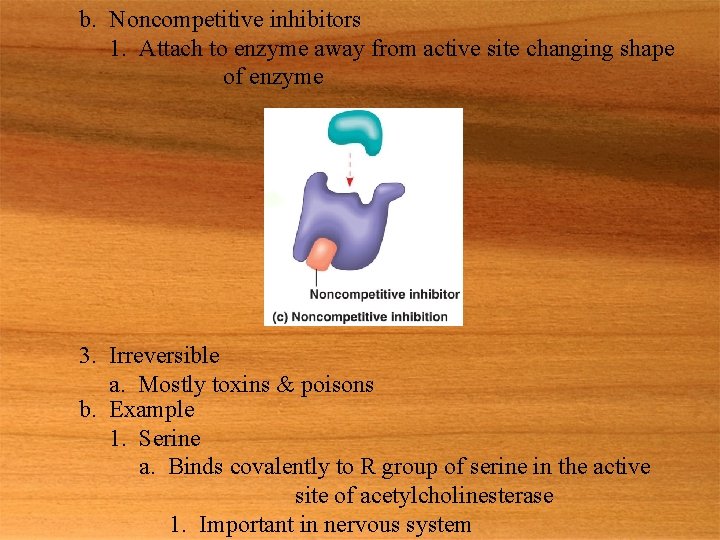
b. Noncompetitive inhibitors 1. Attach to enzyme away from active site changing shape of enzyme 3. Irreversible a. Mostly toxins & poisons b. Example 1. Serine a. Binds covalently to R group of serine in the active site of acetylcholinesterase 1. Important in nervous system

2. DDT 3. Antibiotics a. Penicillin 1. Blocks the active site of an enzyme in bacteria that makes cell wall 4. Important regulators of cellular metabolism 8. 5 Regulation of enzyme activity helps control metabolism I. Allosteric regulation of enzymes A. Allosteric regulation 1. Regulaion of a protein’s function at 1 site by the binding of a regulatory molecule to a separate site 2. Can inhibit or stimulate B. Allosteric activation & inhibition 1. Allossterically controlled enzyme structure a. 2 or more subunits 1. Each has own active site 2. Oscillates btwn. 2 different shapes a. Active or inactive
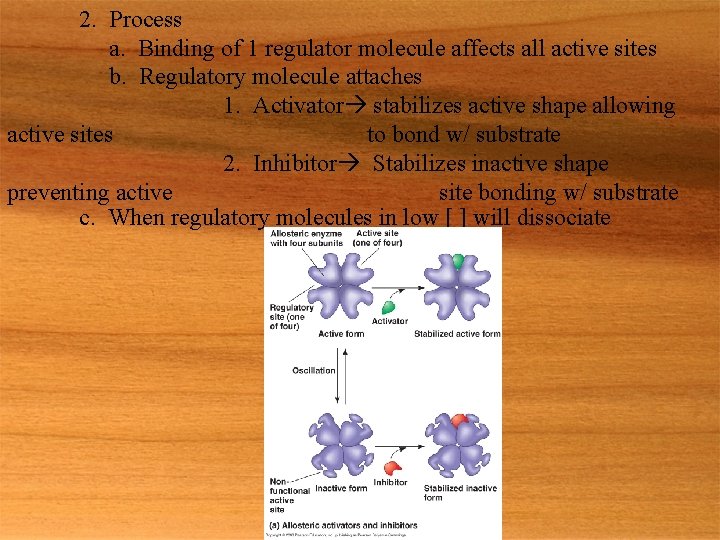
2. Process a. Binding of 1 regulator molecule affects all active sites b. Regulatory molecule attaches 1. Activator stabilizes active shape allowing active sites to bond w/ substrate 2. Inhibitor Stabilizes inactive shape preventing active site bonding w/ substrate c. When regulatory molecules in low [ ] will dissociate
![3. Example a. Catabolic & anabolic pathways 1. High [ ] of ATP inhibits 3. Example a. Catabolic & anabolic pathways 1. High [ ] of ATP inhibits](http://slidetodoc.com/presentation_image_h2/88010975e8b34c0823534a32334a8437/image-22.jpg)
3. Example a. Catabolic & anabolic pathways 1. High [ ] of ATP inhibits catabolic enzymes a. Decreases ATP production 2. High [ ] of ADP activates catabolic enzymes a. Increasing ATP production 4. Cooperativity a. Substrate binding to 1 active site increases affinity for all other active sites 1. Amplifies response to substrate b. Example 1. Hemoglobin (not an enzyme) C. Feedback inhibition 1. Metabolic control in which the end product acts as an inhibitor of an enzyme w/ in the pathway
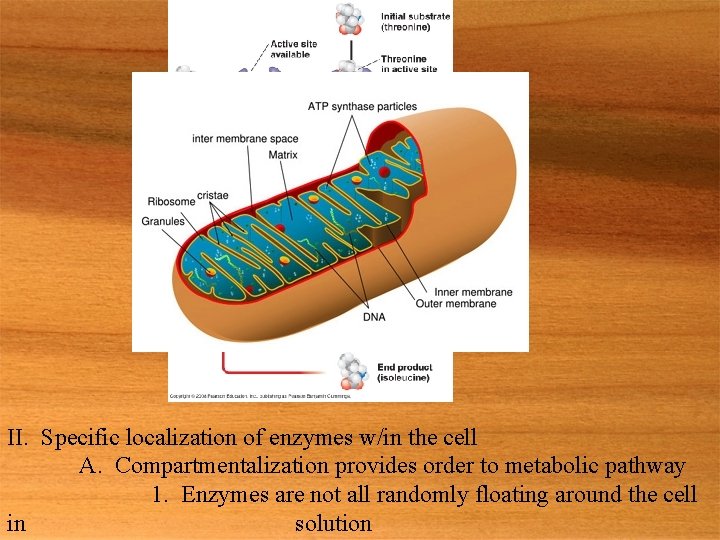
II. Specific localization of enzymes w/in the cell A. Compartmentalization provides order to metabolic pathway 1. Enzymes are not all randomly floating around the cell in solution
- Slides: 23