7 th International Battery Expo Recycling Conference IBRX

7 th International Battery Expo & Recycling Conference IBRX India -- 2015 3 -5 March 2015, Goa, India Lead-Carbon Electrode: Structure, Properties and Mechanism of Processes during Battery Operation Prof. Detchko Pavlov Lead-Acid Batteries Department Institute of Electrochemistry and Energy Systems (IEES) Bulgarian Academy of Sciences (BAS), 1113 Sofia, Bulgaria D. Pavlov, IBRX India – 2015, 3 -5 March 2015, Goa, India

Carbon in negative plates of lead-acid batteries Lead-Carbon negative plates 1 Addition of Carbon to NAM 2 Half plate Pb Half plate Carbon Ultrabattery D. Pavlov, IBRX India – 2015, 3 -5 March 2015, Goa, India 3 All Carbon negative plate Supercapacitor 2

Proposed mechanisms of carbon action • Carbon enhances the overall conductivity of NAM K. Nakamura, M. Shiomi, K. Takahashi, M. Tsubota, J. Power Sources, 59 (1996) 153 • D. Pavlov, IBRX India – 2015, 3 -5 March 2015, Goa, India 3

Contribution of carbon additives to eletrochemical processes in NAM of the lead-acid batteries D. Pavlov, P. Nikolov, ALABC Project No. C 2. 3 “Identification of the mechanism(s) by which certain forms of carbon, when included in the negative active material of a valve-regulated lead–acid battery exposed to high-rate partial-state-of-charge operation, are able to resist sulfation” – Final report 2009 D. Pavlov, T. Rogachev, P. Nikolov, G. Petkova, Mechanism of action of electrochemically active carbons on the processes that take place at the negative plates of lead-acid batteries, J. Power Sources, 191 (2009) 58 -75 D. Pavlov, IBRXIndia–– 2015, 3 -5 March 2015, Goa, India Pavlov, , IBRX Goa, India D. Pavlov 44
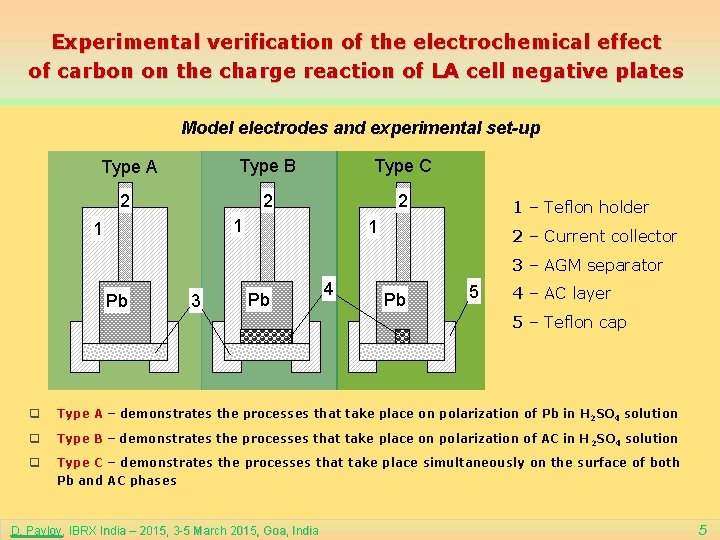
Experimental verification of the electrochemical effect of carbon on the charge reaction of LA cell negative plates Model electrodes and experimental set-up Type A Type B Type C 2 2 2 1 1 1 – Teflon holder 1 2 – Current collector 3 – AGM separator Pb 3 Pb 4 Pb 5 4 – AC layer 5 – Teflon cap q Type A – demonstrates the processes that take place on polarization of Pb in H 2 SO 4 solution q Type B – demonstrates the processes that take place on polarization of AC in H 2 SO 4 solution q Type C – demonstrates the processes that take place simultaneously on the surface of both Pb and AC phases D. Pavlov, IBRX India – 2015, 3 -5 March 2015, Goa, India 5
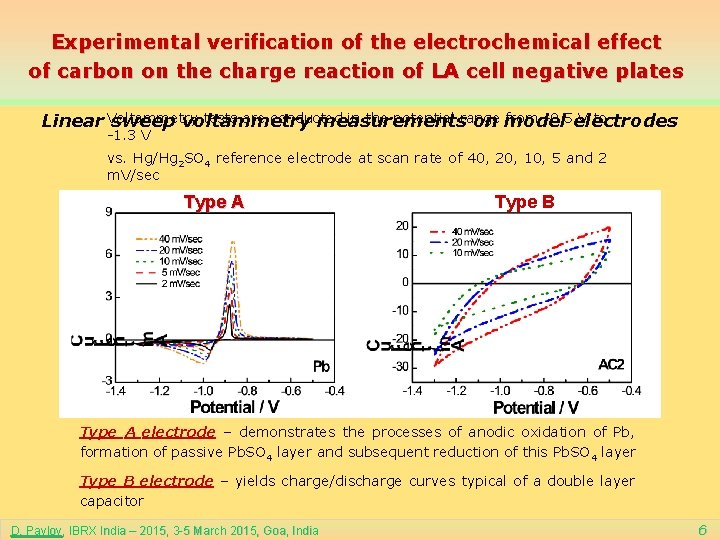
Experimental verification of the electrochemical effect of carbon on the charge reaction of LA cell negative plates tests are conducted in the potential range from -0. 5 electrodes V to Linear Voltammetry sweep voltammetry measurements on model -1. 3 V vs. Hg/Hg 2 SO 4 reference electrode at scan rate of 40, 20, 10, 5 and 2 m. V/sec Type A Type B Type A electrode – demonstrates the processes of anodic oxidation of Pb, formation of passive Pb. SO 4 layer and subsequent reduction of this Pb. SO 4 layer Type B electrode – yields charge/discharge curves typical of a double layer capacitor D. Pavlov, IBRX India – 2015, 3 -5 March 2015, Goa, India 6
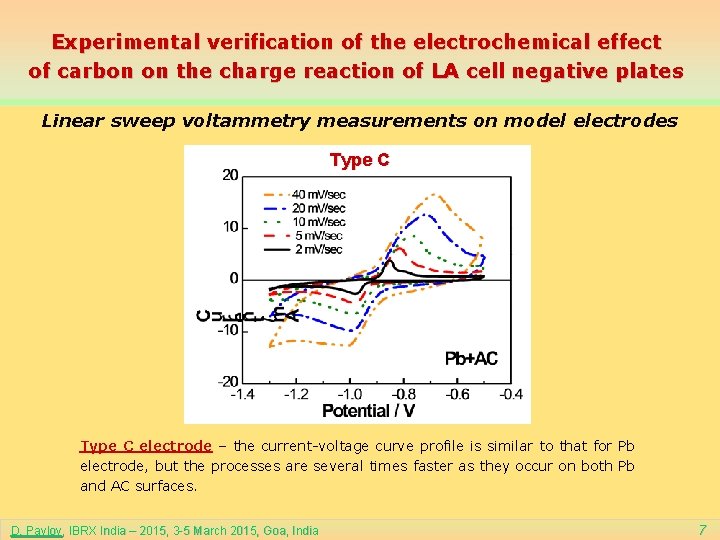
Experimental verification of the electrochemical effect of carbon on the charge reaction of LA cell negative plates Linear sweep voltammetry measurements on model electrodes Type C electrode – the current-voltage curve profile is similar to that for Pb electrode, but the processes are several times faster as they occur on both Pb and AC surfaces. D. Pavlov, IBRX India – 2015, 3 -5 March 2015, Goa, India 7
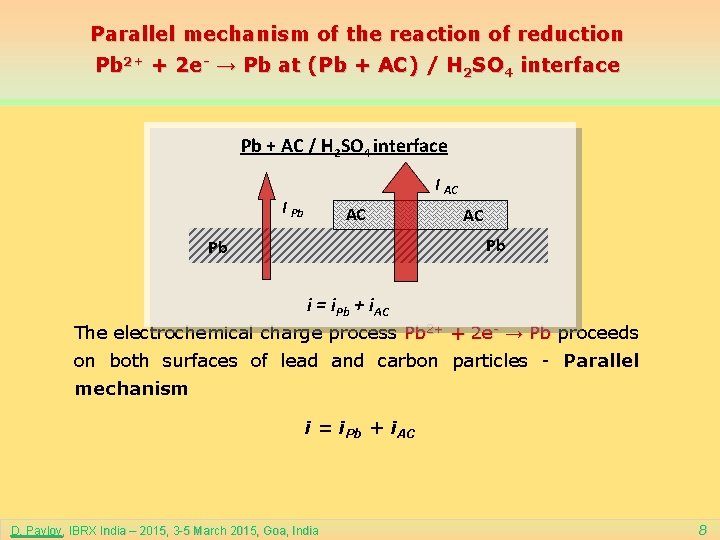
Parallel mechanism of the reaction of reduction Pb 2+ + 2 e- → Pb at (Pb + AC) / H 2 SO 4 interface Pb + AC / H 2 SO 4 interface I AC I Pb AC AC Pb Pb i = i. Pb + i. AC The electrochemical charge process Pb 2+ + 2 e- → Pb proceeds on both surfaces of lead and carbon particles - Parallel mechanism i = i. Pb + i. AC D. Pavlov, IBRX India – 2015, 3 -5 March 2015, Goa, India 8

Electro-deposition of lead particles onto the carbon surfaces Pb particles Carbon particles Pb branches Carbon particles • Transfer of electrons to Pb 2+ ions proceeds through the Pb/solution and AC/solution interfaces. • Pb nuclei are formed in active centers on the surface of the carbon particles and they grow into Pb branches • Activated carbon particles are incorporated in the structure of NAM. D. Pavlov, IBRX India – 2015, 3 -5 March 2015, Goa, India 9

Periods during which the lead and carbon parts of the negative electrode in lead-acid batteries exhibit active action D. Pavlov, P. Nikolov, Formation of capacitive carbon and electrochemical lead electrode systems at the negative plates of lead-acid batteries and elementary processes on cycling, J. Power Sources, 242 (2013) 380 -399 D. Pavlov, IBRXIndia–– 2015, 3 -5 March 2015, Goa, India Pavlov, , IBRX Goa, India D. Pavlov 10 10
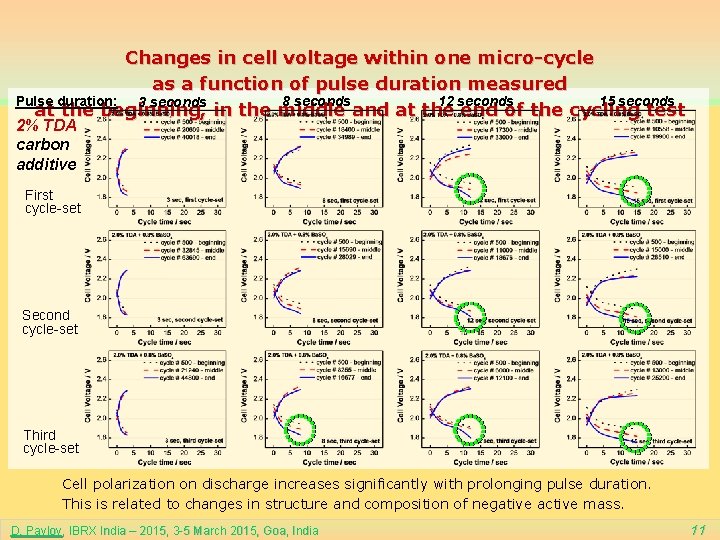
Changes in cell voltage within one micro-cycle as a function of pulse duration measured 8 seconds 12 seconds Pulse duration: 15 seconds 3 seconds at the beginning, in the middle and at the end of the cycling test 2% TDA carbon additive First cycle-set Second cycle-set Third cycle-set Cell polarization on discharge increases significantly with prolonging pulse duration. This is related to changes in structure and composition of negative active mass. D. Pavlov, IBRX India – 2015, 3 -5 March 2015, Goa, India 11
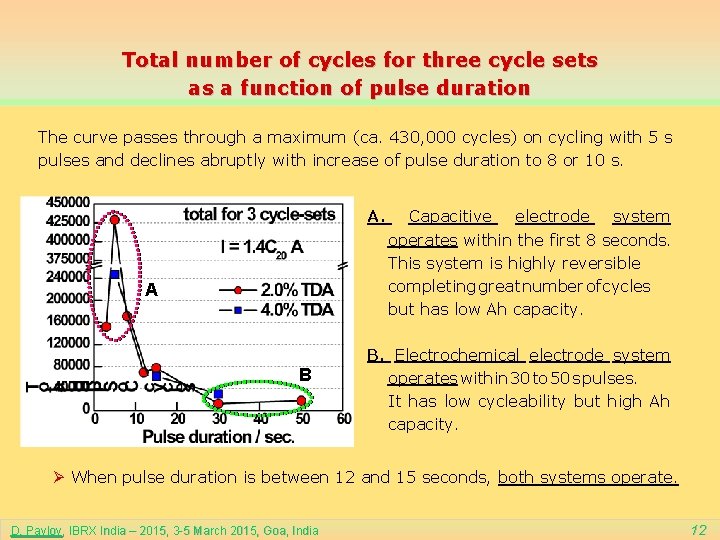
Total number of cycles for three cycle sets as a function of pulse duration The curve passes through a maximum (ca. 430, 000 cycles) on cycling with 5 s pulses and declines abruptly with increase of pulse duration to 8 or 10 s. A. A B Capacitive electrode system operates within the first 8 seconds. This system is highly reversible completing great number of cycles but has low Ah capacity. B. Electrochemical electrode system operates within 30 to 50 s pulses. It has low cycleability but high Ah capacity. Ø When pulse duration is between 12 and 15 seconds, both systems operate. D. Pavlov, IBRX India – 2015, 3 -5 March 2015, Goa, India 12

Electrochemical and capacitive electrode systems formed on the negative plates of a lead-acid battery Carbon capacitive system - - Electrochemical lead system + Capacitive system contributes 0. 22% to the 4. 5 Ah capacity of the cell. The electrochemical system has dominating role in the charge–discharge processes. D. Pavlov, IBRX India – 2015, 3 -5 March 2015, Goa, India 13

Influence of expander on electrochemical properties of lead-carbon electrode D. Pavlov, P. Nikolov and T. Rogachev, Influence of expander components on the processes at the negative plates of lead-acid cells on HRPSo. C cycling. Part I. Effect of lignosulfonates and Ba. SO 4 on the processes of charge and discharge of negative plates, J. Power Sources, 195 (2010) 4435 -4443 D. Pavlov, P. Nikolov and T. Rogachev, Influence of expander components on the processes at the negative plates of lead-acid cells on HRPSo. C cycling. Part II. Effect of carbon additives on the processes of charge and discharge of negative plates. Nanotechnological effects, J. Power Sources, 195 (2010) 4444 -4457 D. Pavlov, IBRXIndia–– 2015, 3 -5 March 2015, Goa, India Pavlov, , IBRX Goa, India D. Pavlov 14 14

Carbons used for the investigations and negative paste formulation Specific characteristics: Material Type of material Particle Size BET Surface m 2/g PRINTEX® A PR 1 carbon black 41 nm 45 PRINTEX® U PR 2 carbon black 25 nm 100 PRINTEX® F 85 PR 3 carbon black 16 nm 200 PRINTEX® 90 PR 4 carbon black 14 nm 300 NEGATIVE PASTE FORMULATION H 2 SO 4 / Leady Oxide = 4. 5 % Vanisperse A – 0. 2% or 0. 0% Ba. SO 4 - 0. 8% PR 1 -4 – 0. 2, 0. 5, 1. 0, 1. 5 or 2. 0% Total number of pastes and cells: 40 D. Pavlov, IBRX India – 2015, 3 -5 March 2015, Goa, India 15
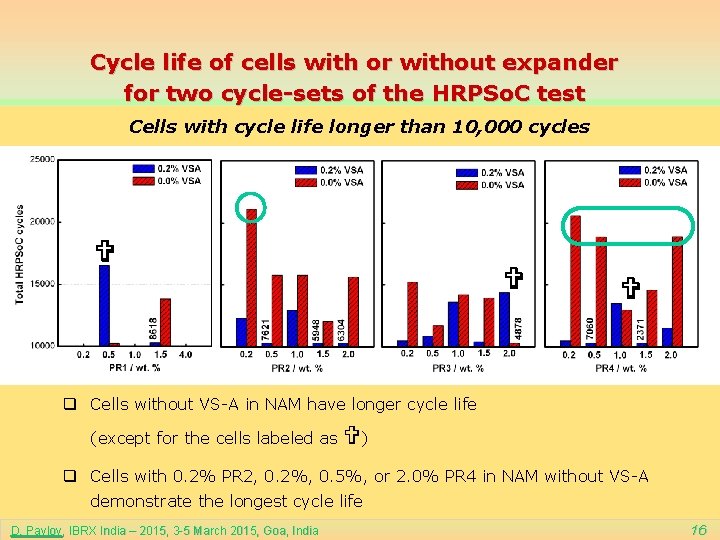
Cycle life of cells with or without expander for two cycle-sets of the HRPSo. C test Cells with cycle life longer than 10, 000 cycles q Cells without VS-A in NAM have longer cycle life (except for the cells labeled as ) q Cells with 0. 2% PR 2, 0. 2%, 0. 5%, or 2. 0% PR 4 in NAM without VS-A demonstrate the longest cycle life D. Pavlov, IBRX India – 2015, 3 -5 March 2015, Goa, India 16

Influence of Carbon additives on the pore system of the negative active mass and on the chemical and electrochemical reactions D. Pavlov, P. Nikolov, T. Rogachev, Influence of carbons on the structure of the negative active material of lead-acid batteries and on battery performance, J. Power Sources, 196 (2011) 5155 -5167 D. Pavlov, P. Nikolov, Formation of capacitive carbon and electrochemical lead electrode systems at the negative plates of lead-acid batteries and elementary processes on cycling, J. Power Sources, 242 (2013) 380 -399 D. Pavlov, IBRXIndia–– 2015, 3 -5 March 2015, Goa, India Pavlov, , IBRX Goa, India D. Pavlov 17 17

Specific BET surface NAM Influence of TDAarea and of AC 3 a function of and TDA median or AC 3 carbon contentof NAM on totalaspore volume pore radius Total pore volume Median pore radius (volume) TDA cm 3. g-1 mm AC 3 cm 3. g-1 mm 0. 5% 0. 1357 7. 25 0. 5% 0. 1305 1. 10 2. 0% 0. 1370 2. 44 1. 0% 0. 1326 0. 55 AC 3 reduces the pore radius down to submicron dimensions TDA BET surface / m 2. g-1 AC 3 0. 5 % 2. 0 % 0. 5 % 1. 0 % 5. 80 25. 20 4. 70 8. 75 D. Pavlov, IBRX India – 2015, 3 -5 March 2015, Goa, India 18
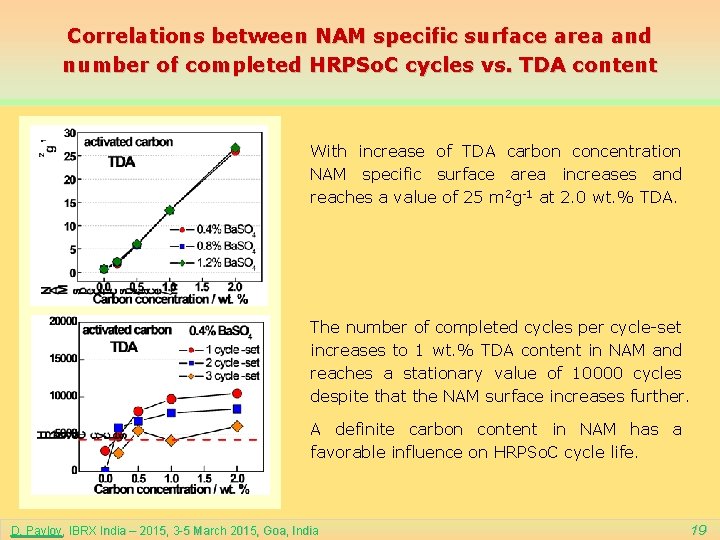
Correlations between NAM specific surface area and number of completed HRPSo. C cycles vs. TDA content With increase of TDA carbon concentration NAM specific surface area increases and reaches a value of 25 m 2 g-1 at 2. 0 wt. % TDA. The number of completed cycles per cycle-set increases to 1 wt. % TDA content in NAM and reaches a stationary value of 10000 cycles despite that the NAM surface increases further. A definite carbon content in NAM has a favorable influence on HRPSo. C cycle life. D. Pavlov, IBRX India – 2015, 3 -5 March 2015, Goa, India 19

Correlations between NAM specific surface area and number of completed HRPSo. C cycles vs. AC 3 content With increase of AC 3 carbon concentration NAM specific surface area reaches a value of 20 m 2 g-1 at 2. 0 wt. % AC 3 Cycleability increases abruptly from 900 cycles for cells without AC 3 to 9200 cycles for cells with 0. 2 wt. % AC 3. Then it declines with further increase of carbon content down to 2700 cycles at 2% AC 3 in NAM. The size of the carbon particles and the way they are incorporated in NAM structure determine the reversibility of the electrochemical processes and the number of cycles per cycle-set. D. Pavlov, IBRX India – 2015, 3 -5 March 2015, Goa, India 20
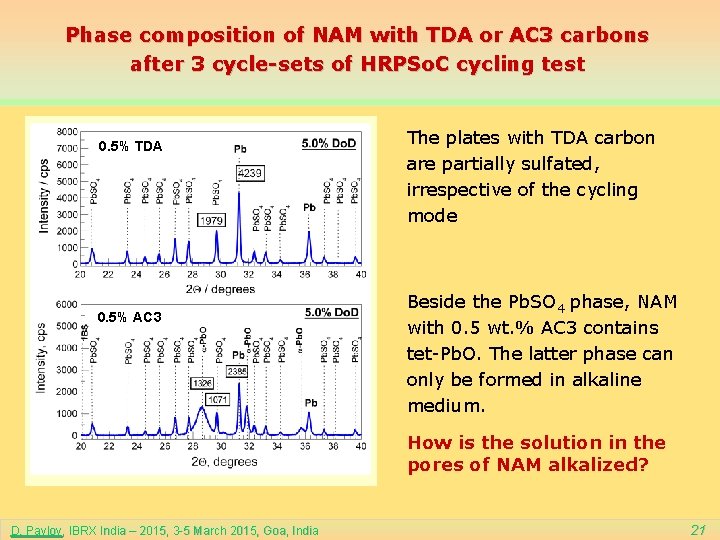
Phase composition of NAM with TDA or AC 3 carbons after 3 cycle-sets of HRPSo. C cycling test 0. 5% TDA 0. 5% AC 3 The plates with TDA carbon are partially sulfated, irrespective of the cycling mode Beside the Pb. SO 4 phase, NAM with 0. 5 wt. % AC 3 contains tet-Pb. O. The latter phase can only be formed in alkaline medium. How is the solution in the pores of NAM alkalized? D. Pavlov, IBRX India – 2015, 3 -5 March 2015, Goa, India 21

Mechanism of formation of Pb. SO 4 crystals in the wide pores of NAM Carbon additives affect the formation of wide pores in NAM Free access of H 2 SO 4 in the wide pores H+ NAM H+ Pb 2+ Pb. SO 4 H+ H 2 SO 4 H+ Pb. SO 4 NAM Formation of Pb. SO 4 The free access of the big SO 42 - and HSO 4 - ions into the wide pores assures the formation of Pb. SO 4 crystals. D. Pavlov, IBRX India – 2015, 3 -5 March 2015, Goa, India 22

Mechanism of formation of tet-Pb. O in the membrane sized pores of NAM a) H 2 O dissociates and H+ ions migrate from the pores b) Pb(OH)2 and tet-Pb. O form in NAM NAM H+ Pb 2+ + - H + OH H 2 O Pb 2+ Pb(OH)2 NAM Pb(OH)2 -Pb. O + H 2 O H+ + OHNAM When anodic current flows through NAM, Pb 2+ ions are formed. These ions charge the solution in the pores with positive charges. SO 42 - ions can not diffuse to neutralize the positively charged solution. Its electroneutralization involves dissociation of water in the pore solution H 2 O H+ + OHThe H+ ions migrate from the NAM pores to the bulk solution. Thus the solution in the pores is alkalized and Pb(OH)2 and Pb. O are formed. 1. D. Pavlov, R. Popova, Electrochim. Acta, 15 (1970) 1483 -1491. 2. D. Pavlov, Electrochim. Acta, 23 (1978) 845 -854. 3. D. Pavlov, P. Nikolov, J. Power Sources, 242 (2013) 380 -399. D. Pavlov, IBRX India – 2015, 3 -5 March 2015, Goa, India 23

Conclusions 1. Тwo electrical systems operate in the negative active mass with addition of electrochemically active carbon: a) an electrochemical lead system, and b) a capacitive carbon system 2. The carbon system comprises charging and discharging of the electric double layer and electrochemical processes of formation and growth of lead particles proceed in active centers on the carbon surface. Thus, a lead-carbon electrode is formed. 3. Most often, addition of lignosulfonate to the negative active material does not improve the electrochemical parameters of the lead-carbon electrode. 4. When the carbon particles are big in size, they form large pores in NAM allowing free access of the SO 42 - ions and Pb. SO 4 particles are formed during electrode discharge. 5. When carbon particles are small in size, pores of membrane sizes may be formed in NAM and Pb. O particles will form during electrode discharge. Thus, carbon additives exert an influence on the electrochemical reactions that proceed on the lead-carbon electrodes. D. Pavlov, IBRX India – 2015, 3 -5 March 2015, Goa, India 24

Thank you for your attention! D. Pavlov, IBRXIndia–– 2015, 3 -5 March 2015, Goa, India Pavlov, , IBRX Goa, India D. Pavlov 25 25

D. Pavlov, IBRX India – 2015, 3 -5 March 2015, Goa, India 26

Publications on influence of carbon additives to NAM on lead-acid battery performance D. Pavlov, T. Rogachev, P. Nikolov, G. Petkova, Mechanism of action of electrochemically active carbons on the processes that take place at the negative plates of lead-acid batteries, J. Power Sources, 191 (2009) 58 -75 D. Pavlov, P. Nikolov and T. Rogachev, Influence of expander components on the processes at the negative plates of lead-acid cells on HRPSo. C cycling. Part I. Effect of lignosulfonates and Ba. SO 4 on the processes of charge and discharge of negative plates, J. Power Sources, 195 (2010) 4435 -4443 D. Pavlov, P. Nikolov and T. Rogachev, Influence of expander components on the processes at the negative plates of lead-acid cells on HRPSo. C cycling. Part II. Effect of carbon additives on the processes of charge and discharge of negative plates. Nanotechnological effects, J. Power Sources, 195 (2010) 4444 -4457 D. Pavlov, P. Nikolov, T. Rogachev, Influence of carbons on the structure of the negative active material of lead-acid batteries and on battery performance, J. Power Sources, 196 (2011) 5155 -5167 D. Pavlov, How carbon can extend the cycle life of lead acid batteries, The Batteries International, Issue 84, Summer 2012, p. 43 -49 D. Pavlov, P. Nikolov, Formation of capacitive carbon and electrochemical lead electrode systems at the negative plates of lead-acid batteries and elementary processes on cycling, J. Power Sources, 242 (2013) 380 -399 D. Pavlov, IBRX India – 2015, 3 -5 March 2015, Goa, India 27

Problems to batteries for HEV applications Lead-acid batteries faced two technical problems with their introduction of lead-acid batteries to HEV: a) improvement of the charge acceptance on HRPSo. C operation; b) inhibition of the lead electrode sulfatization. These problems were significantly solved with creation of the lead-carbon electrode. D. Pavlov, IBRX India – 2015, 3 -5 March 2015, Goa, India 28

Morphology of NAM with or without expander 1. 5% PR 4 2. 0% PR 4 with Vanisperse A 1. 0% PR 4 2. 0% PR 4 without Vanisperse A Almost the entire Pb surface is covered with carbon particles, when high concentrations of PR 4 are added to the negative paste D. Pavlov, IBRX India – 2015, 3 -5 March 2015, Goa, India 29
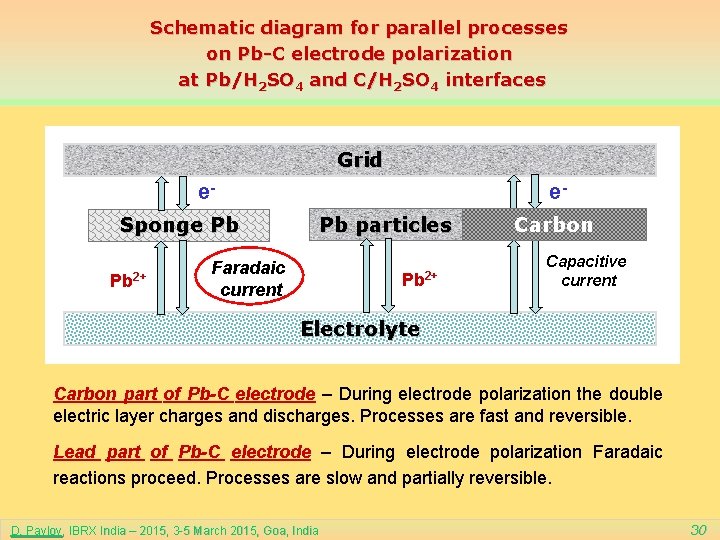
Schematic diagram for parallel processes on Pb-C electrode polarization at Pb/H 2 SO 4 and C/H 2 SO 4 interfaces Grid e- e. Pb particles Sponge Pb Pb 2+ Faradaic current Pb 2+ Carbon Capacitive current Electrolyte Carbon part of Pb-C electrode – During electrode polarization the double electric layer charges and discharges. Processes are fast and reversible. Lead part of Pb-C electrode – During electrode polarization Faradaic reactions proceed. Processes are slow and partially reversible. D. Pavlov, IBRX India – 2015, 3 -5 March 2015, Goa, India 30
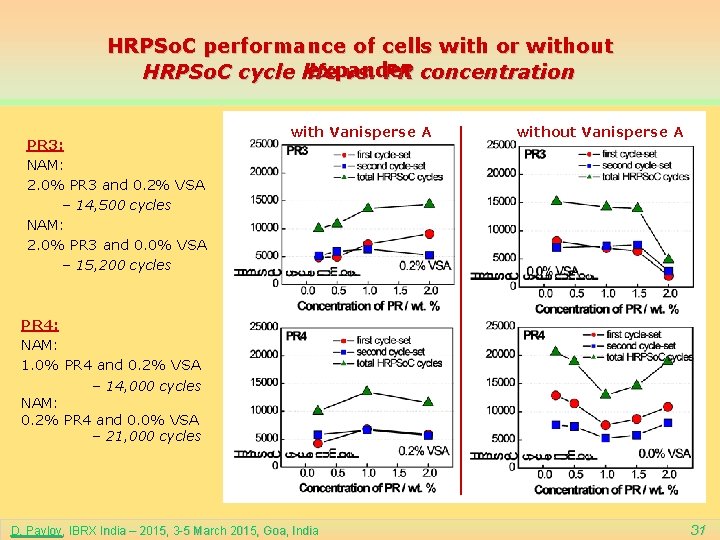
HRPSo. C performance of cells with or without expander HRPSo. C cycle life vs. PR concentration PR 3: NAM: 2. 0% PR 3 and 0. 2% VSA – 14, 500 cycles NAM: 2. 0% PR 3 and 0. 0% VSA – 15, 200 cycles with Vanisperse A without Vanisperse A PR 4: NAM: 1. 0% PR 4 and 0. 2% VSA – 14, 000 cycles NAM: 0. 2% PR 4 and 0. 0% VSA – 21, 000 cycles D. Pavlov, IBRX India – 2015, 3 -5 March 2015, Goa, India 31

Electrochemical and chemical reactions during cycling • In NAM with wide pores • In NAM with membrane sized pores Pb. SO 4 phase is formed Pb. SO 4 and Pb. O phases are formed q Carbon particle size determines the radius of the pores in NAM, which limit the access of SO 42 - ions and determine chemical reactions in the pores. D. Pavlov, IBRX India – 2015, 3 -5 March 2015, Goa, India 32
- Slides: 32