7 th International Battery Expo Recycling Conference Efficient

7 th International Battery Expo & Recycling Conference Efficient Energy conversion & storage technologies: Trends and Potential Applications 18 -Feb-21 Advanced Engineering Tata Motors 1

7 th International Battery Expo & Recycling Conference Presentation Over view Insights into future energy options Requirement power sources for sustained mobility Use of power sources in Automotive & Stationary application Fuel Cell Technological gaps required to be filled Redox flow type of Batteries Li-Ion Status & Technological improvements required So. Nic Status & Technological improvements required 18 -Feb-21 Advanced Engineering Tata Motors 2
7 th International Battery Expo & Recycling Conference
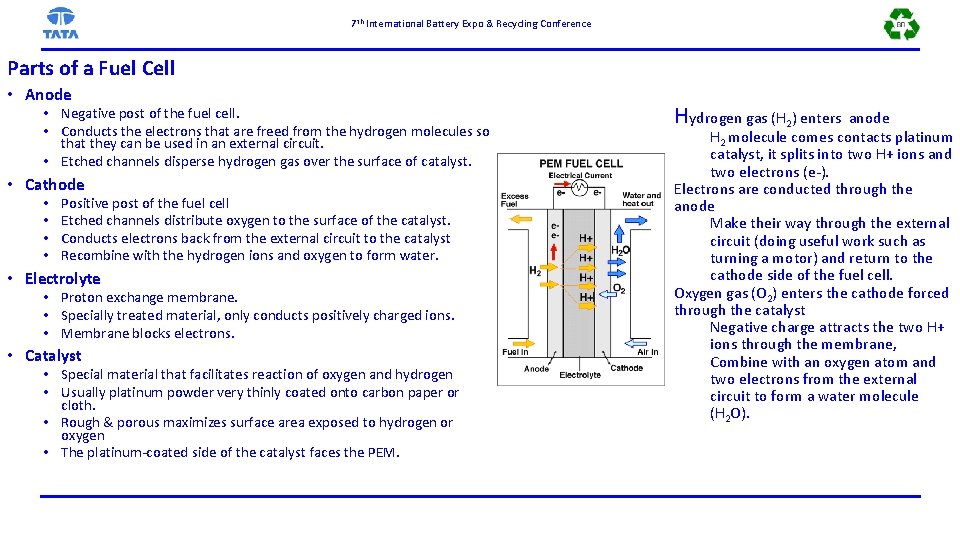
7 th International Battery Expo & Recycling Conference Parts of a Fuel Cell • Anode • Negative post of the fuel cell. • Conducts the electrons that are freed from the hydrogen molecules so that they can be used in an external circuit. • Etched channels disperse hydrogen gas over the surface of catalyst. • Cathode • • Positive post of the fuel cell Etched channels distribute oxygen to the surface of the catalyst. Conducts electrons back from the external circuit to the catalyst Recombine with the hydrogen ions and oxygen to form water. • Electrolyte • Proton exchange membrane. • Specially treated material, only conducts positively charged ions. • Membrane blocks electrons. • Catalyst • Special material that facilitates reaction of oxygen and hydrogen • Usually platinum powder very thinly coated onto carbon paper or cloth. • Rough & porous maximizes surface area exposed to hydrogen or oxygen • The platinum-coated side of the catalyst faces the PEM. Hydrogen gas (H 2) enters anode H 2 molecule comes contacts platinum catalyst, it splits into two H+ ions and two electrons (e-). Electrons are conducted through the anode Make their way through the external circuit (doing useful work such as turning a motor) and return to the cathode side of the fuel cell. Oxygen gas (O 2) enters the cathode forced through the catalyst Negative charge attracts the two H+ ions through the membrane, Combine with an oxygen atom and two electrons from the external circuit to form a water molecule (H 2 O).
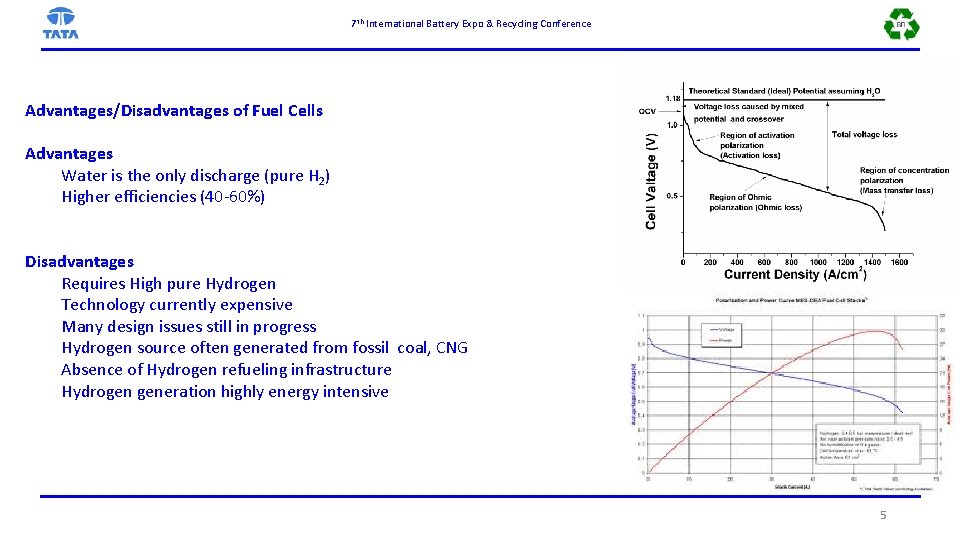
7 th International Battery Expo & Recycling Conference Advantages/Disadvantages of Fuel Cells Advantages Water is the only discharge (pure H 2) Higher efficiencies (40 -60%) Disadvantages Requires High pure Hydrogen Technology currently expensive Many design issues still in progress Hydrogen source often generated from fossil coal, CNG Absence of Hydrogen refueling infrastructure Hydrogen generation highly energy intensive 5
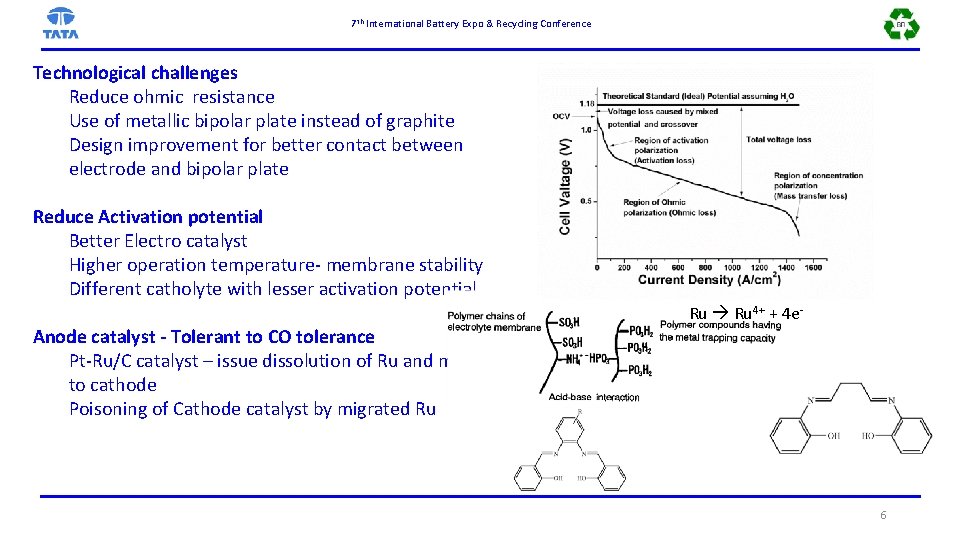
7 th International Battery Expo & Recycling Conference Technological challenges Reduce ohmic resistance Use of metallic bipolar plate instead of graphite Design improvement for better contact between electrode and bipolar plate Reduce Activation potential Better Electro catalyst Higher operation temperature- membrane stability Different catholyte with lesser activation potential Anode catalyst - Tolerant to CO tolerance Pt-Ru/C catalyst – issue dissolution of Ru and migration to cathode Poisoning of Cathode catalyst by migrated Ru Ru 4+ + 4 e- 6

7 th International Battery Expo & Recycling Conference Operation under H 2 starvation condition CO 2 is detected in anode exhaust due to oxidation of carbon Slight decrease in open circuit voltage of the fuel increase in the ohmic resistance and charge transfer resistance – impedance studies increase in the size of the platinum particle size on both anode and cathode due to sintering- X ray diffraction Design of Membrane & Electrode assembly(MEA) for cell reversal Operation of stack under Hydrogen starved conditions lead to failure Membrane inherent characteristics of allowing cross over of reactants Development of hotspots and degradation of MEA Location coolant fluid away from the reaction site – ineffective cooling Aggolomersiation of catalyst particles Improvement of stability and durability Improvement in tolerance for oxidation Cross linking the polymer electrolyte Reduction of dissolution of catalyst Fan Zhou et al International Journal of Hydrogen Energy, Volume 40, Issue 6, 19 February 2015, Pages 2833 -2839 18 -Feb-21 Advanced Engineering Tata Motors 7

7 th International Battery Expo & Recycling Conference Inherent Limitations with PEMFC based on Hydrogen and Air(Oxygen) a. In tolerance to poisonous gases like CO which is also generated during Hydrogen production b. Higher activation losses due to lower temperature operation c. Hydrogen cross over from anode to cathode and combustion results in the hot spots which results in pin holes d. Localized heating and temperature raise in results raise in temperature at catalytic site which renders membrane electrode reversibly damaged e. During operation of fuel cell in anode closed loop mode results in cross over of Nitrogen from cathode to anode due concentration gradient and results in dilution of Hydrogen and leads to starvation and localized cell reversal f. Pressure balancing anode to cathode is very critical for prevention of reactants intermixing and cross over g. Humidification of reactants and control of pressure and temperature of reactants is very critical h. Water recovery is required for rendering fuel cell operation maintenance free i. For removal of heat generated during reaction required coolant flow channel which complicates the design of bipolar plate j. Separate thermal management system is required for controlling the temperature k. Fuel cell stack cannot be operated if there is existence of any pinhole or pinhole formed during operation l. There is faster degradation of fuel cell due to diffusion and the intermixing of fuel and air m. Coolant is flown in the coolant channel which is located away from the reaction site, and cooling is not very effective n. Large quantities of noble metal catalyst is required and cost of the fuel cell stack is also high and not affordable o. Since Hydrogen and air fuel cell system operates at 70 deg C, the radiator system required to be oversized. 18 -Feb-21 Advanced Engineering Tata Motors 8
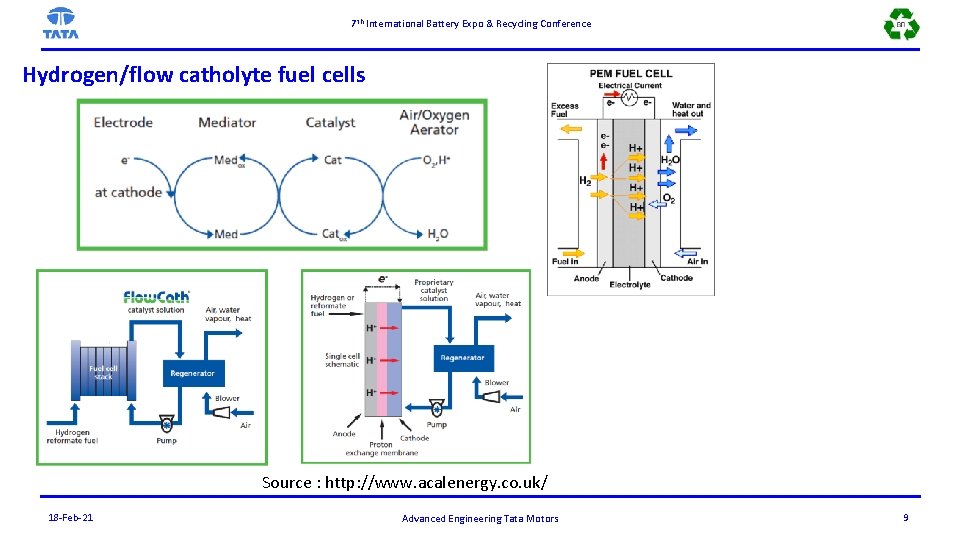
7 th International Battery Expo & Recycling Conference Hydrogen/flow catholyte fuel cells Source : http: //www. acalenergy. co. uk/ 18 -Feb-21 Advanced Engineering Tata Motors 9
7 th International Battery Expo & Recycling Conference 18 -Feb-21 Advanced Engineering Tata Motors 10

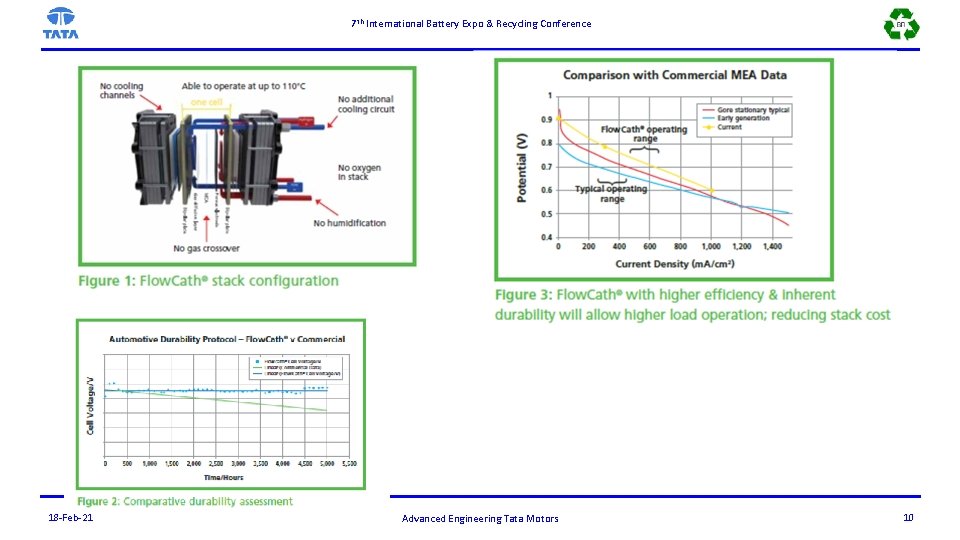
7 th International Battery Expo & Recycling Conference Advantages of PEMFC based on Hydrogen and liquid Redox couple based Fuel cell(ACAL) There is requirement alternate design or alter fuel/oxidant systems. However use of the liquid based Redox couple as catholyte considered for following advantages: a. Platinum required only in the anode and there is no platinum is used for cathode and hence the system is cost effective. Platinum requirements reduced to only 20%. b. Hydrogen cross over reduced significantly from anode to cathode and flowing redox liquid carries away Hydrogen. Even if there is combustion hot spots on membrane is not allowed to formed as the heat generated is removed by flowing liquid c. Unlike conventional fuel cell localized heating and temperature raise is not allowed in Hydrogen-redox based fuel cell d. During operation of fuel cell in anode closed loop anode, there is no crossover of Hydrogen from cathode to anode due concentration gradient and associated problems eliminated. e. Pressure balancing anode to cathode is not very critical f. Humidification of reactants is not required and temperature of reactants may be allowed go up to 110 deg. C. g. Membrane is self-humidified and water recovery is not required for rendering fuel cell operation maintenance free h. There is no separate coolant required and it simplifies the design of bipolar plate i. Separate thermal management system is not required for controlling the temperature j. Fuel cell stack can also be operated if there is existence of any pinhole or pinhole is formed during operation k. Fuel cell system is simplified and durable due to better thermal management l. Liquid is flown in cathode flow channel which removes effectively from the reaction site, and cooling is very effective 18 -Feb-21 Advanced Engineering Tata Motors 11

7 th International Battery Expo & Recycling Conference Because of these above mentioned reasons, Fuel cell stacks of ACAL will be durable longer. However, there are following technological challenges have to overcome: 1. Design of generator for regeneration of cathloyte(reduced form to oxidised form) 2. Water cross over and flooding of anode 3. Periodic purging for draining crossed over water 4. Design of anode for reduction liquid cross over from cathode. 5. Replacement of ions on polymer electrolyte exchange membrane by ions from the catholyte and Contamination ions in polymer electrolyte membrane and resulting in the reduced performance.
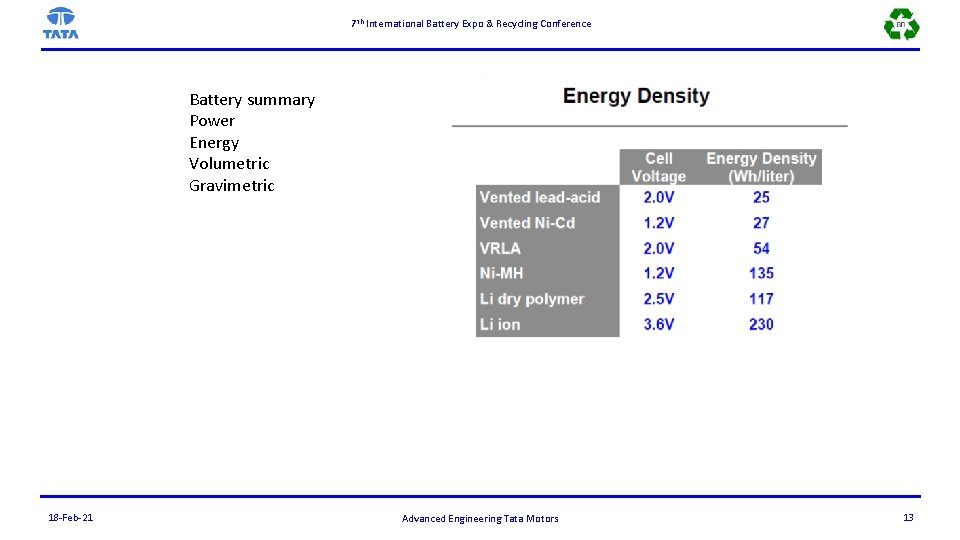
7 th International Battery Expo & Recycling Conference Battery summary Power Energy Volumetric Gravimetric 18 -Feb-21 Advanced Engineering Tata Motors 13
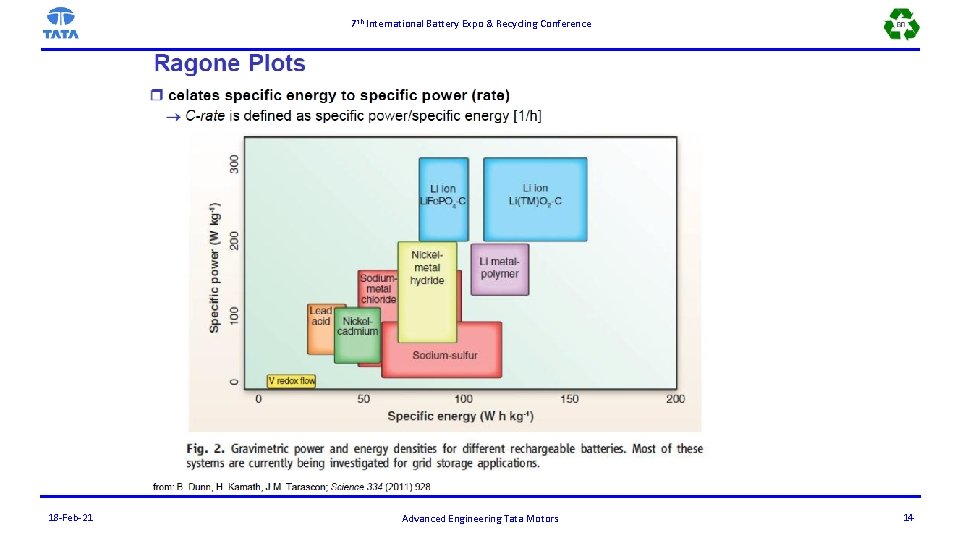
7 th International Battery Expo & Recycling Conference 18 -Feb-21 Advanced Engineering Tata Motors 14
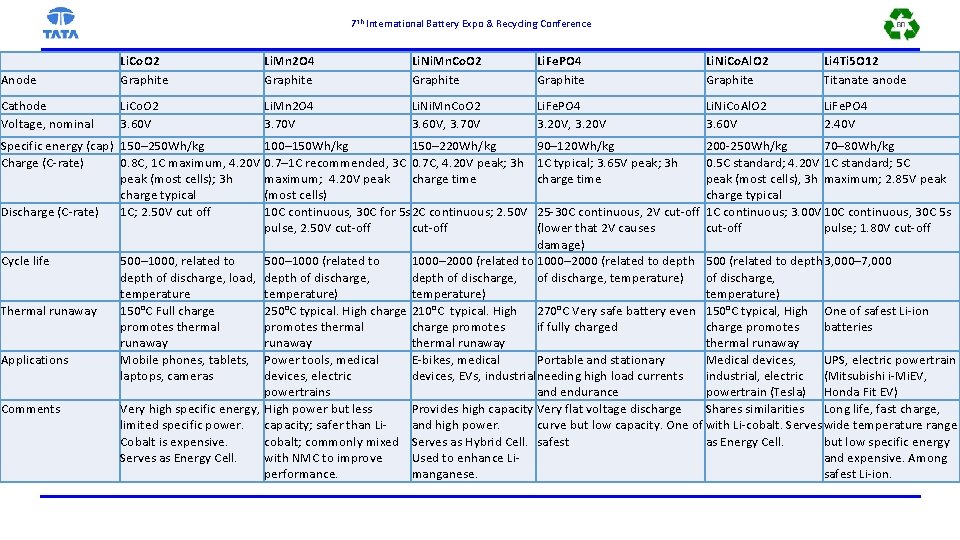
7 th International Battery Expo & Recycling Conference Anode Li. Co. O 2 Graphite Li. Mn 2 O 4 Graphite Li. Ni. Mn. Co. O 2 Graphite Li. Fe. PO 4 Graphite Li. Ni. Co. Al. O 2 Graphite Li 4 Ti 5 O 12 Titanate anode Cathode Voltage, nominal Li. Co. O 2 3. 60 V Li. Mn 2 O 4 3. 70 V Li. Ni. Mn. Co. O 2 3. 60 V, 3. 70 V Li. Fe. PO 4 3. 20 V, 3. 20 V Li. Ni. Co. Al. O 2 3. 60 V Li. Fe. PO 4 2. 40 V Specific energy (cap) 150– 250 Wh/kg 100– 150 Wh/kg 150– 220 Wh/kg Charge (C-rate) 0. 8 C, 1 C maximum, 4. 20 V 0. 7– 1 C recommended, 3 C 0. 7 C, 4. 20 V peak; 3 h peak (most cells); 3 h maximum; 4. 20 V peak charge time charge typical (most cells) Discharge (C-rate) 1 C; 2. 50 V cut off 10 C continuous, 30 C for 5 s 2 C continuous; 2. 50 V pulse, 2. 50 V cut-off Cycle life Thermal runaway Applications Comments 90– 120 Wh/kg 1 C typical; 3. 65 V peak; 3 h charge time 200 -250 Wh/kg 70– 80 Wh/kg 0. 5 C standard; 4. 20 V 1 C standard; 5 C peak (most cells), 3 h maximum; 2. 85 V peak charge typical 25 -30 C continuous, 2 V cut-off 1 C continuous; 3. 00 V 10 C continuous, 30 C 5 s (lower that 2 V causes cut-off pulse; 1. 80 V cut-off damage) 500– 1000, related to 500– 1000 (related to 1000– 2000 (related to depth 500 (related to depth 3, 000– 7, 000 depth of discharge, load, depth of discharge, temperature) of discharge, temperature) 150°C Full charge 250°C typical. High charge 210°C typical. High 270°C Very safe battery even 150°C typical, High One of safest Li-ion promotes thermal charge promotes if fully charged charge promotes batteries runaway thermal runaway Mobile phones, tablets, Power tools, medical E-bikes, medical Portable and stationary Medical devices, UPS, electric powertrain laptops, cameras devices, electric devices, EVs, industrial needing high load currents industrial, electric (Mitsubishi i-Mi. EV, powertrains and endurance powertrain (Tesla) Honda Fit EV) Very high specific energy, High power but less Provides high capacity Very flat voltage discharge Shares similarities Long life, fast charge, limited specific power. capacity; safer than Liand high power. curve but low capacity. One of with Li-cobalt. Serves wide temperature range Cobalt is expensive. cobalt; commonly mixed Serves as Hybrid Cell. safest as Energy Cell. but low specific energy Serves as Energy Cell. with NMC to improve Used to enhance Liand expensive. Among performance. manganese. safest Li-ion.
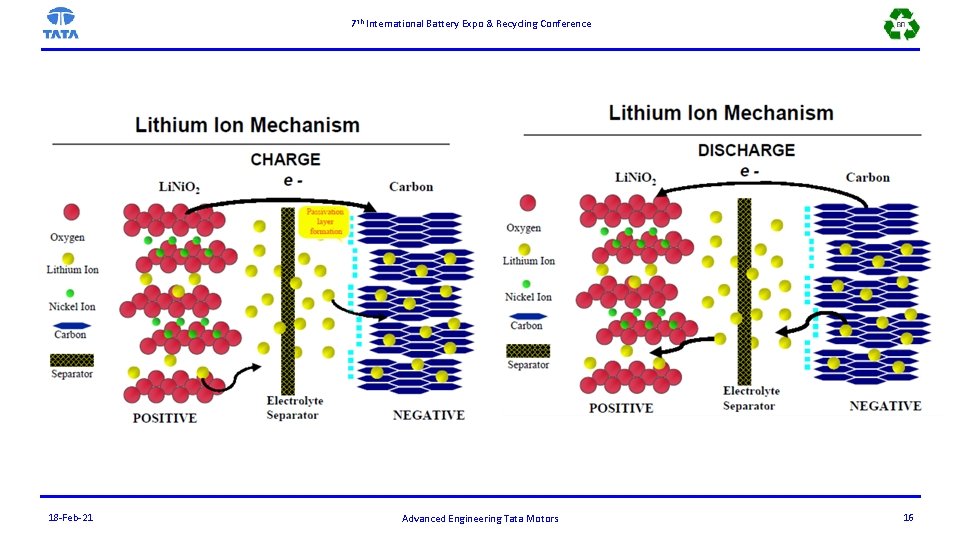
7 th International Battery Expo & Recycling Conference 18 -Feb-21 Advanced Engineering Tata Motors 16
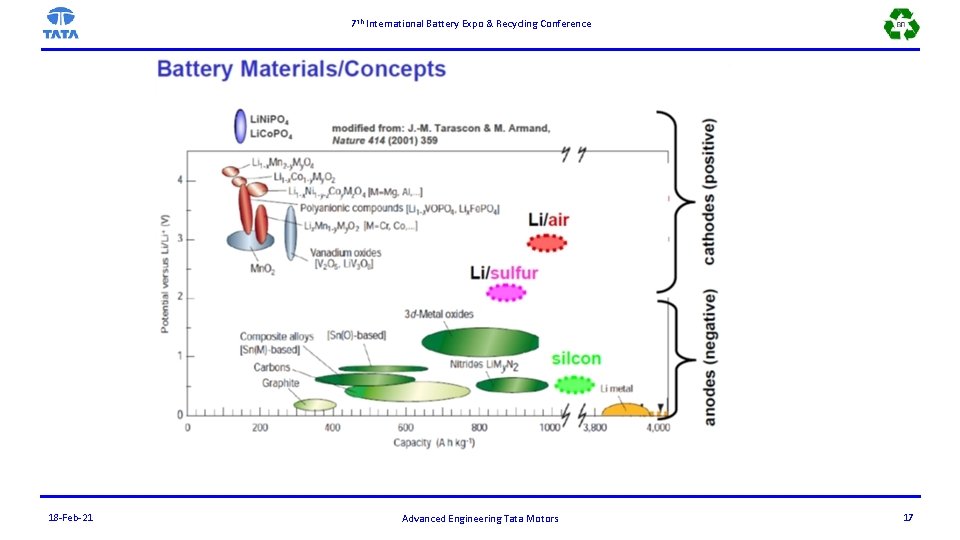
7 th International Battery Expo & Recycling Conference 18 -Feb-21 Advanced Engineering Tata Motors 17

7 th International Battery Expo & Recycling Conference Improvements – Li-Ion Batteries Anode material Lithium metal Lithium –Tin alloy Cathode material
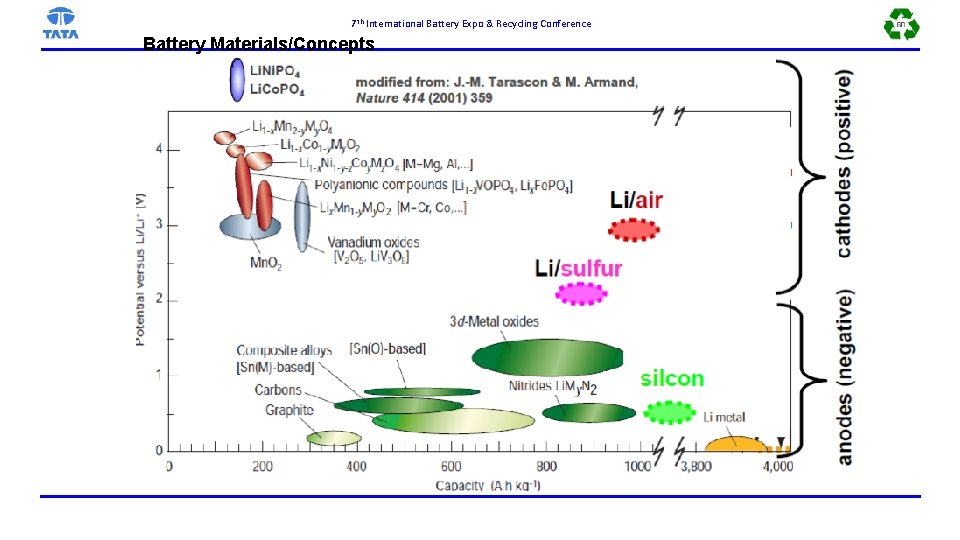
7 th International Battery Expo & Recycling Conference Battery Materials/Concepts
![7 th International Battery Expo & Recycling Conference Battery Specific Energy [Wh/kgelectrodes ] 7 th International Battery Expo & Recycling Conference Battery Specific Energy [Wh/kgelectrodes ]](http://slidetodoc.com/presentation_image_h/00068498a4443aa0d1a48867356ae8ca/image-20.jpg)
7 th International Battery Expo & Recycling Conference Battery Specific Energy [Wh/kgelectrodes ]

7 th International Battery Expo & Recycling Conference Improvement made for improving safety and energy density of Li-Ion Batteries Lithium Solapur batteries
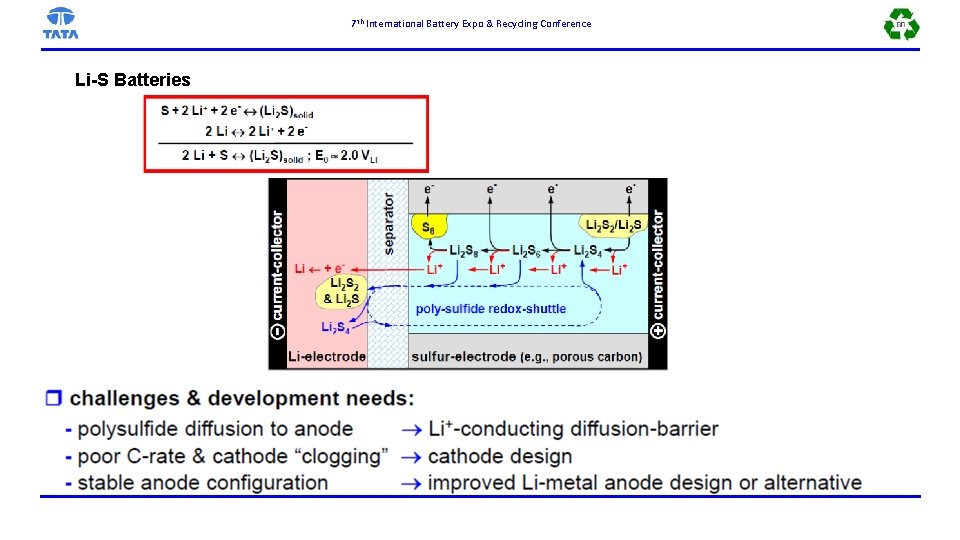
7 th International Battery Expo & Recycling Conference Li-S Batteries

7 th International Battery Expo & Recycling Conference Li-S Batteries: State-of-the-Art
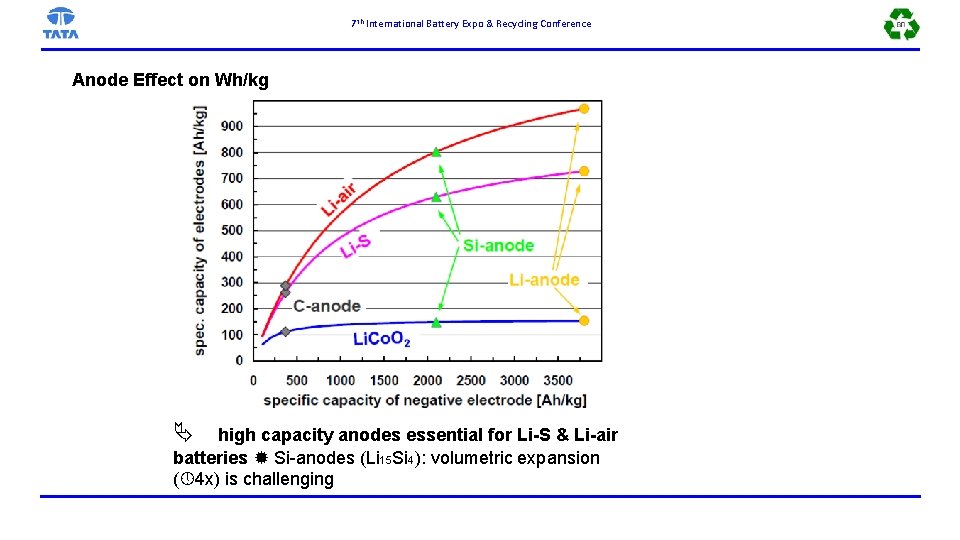
7 th International Battery Expo & Recycling Conference Anode Effect on Wh/kg high capacity anodes essential for Li-S & Li-air batteries Si-anodes (Li 15 Si 4): volumetric expansion ( 4 x) is challenging
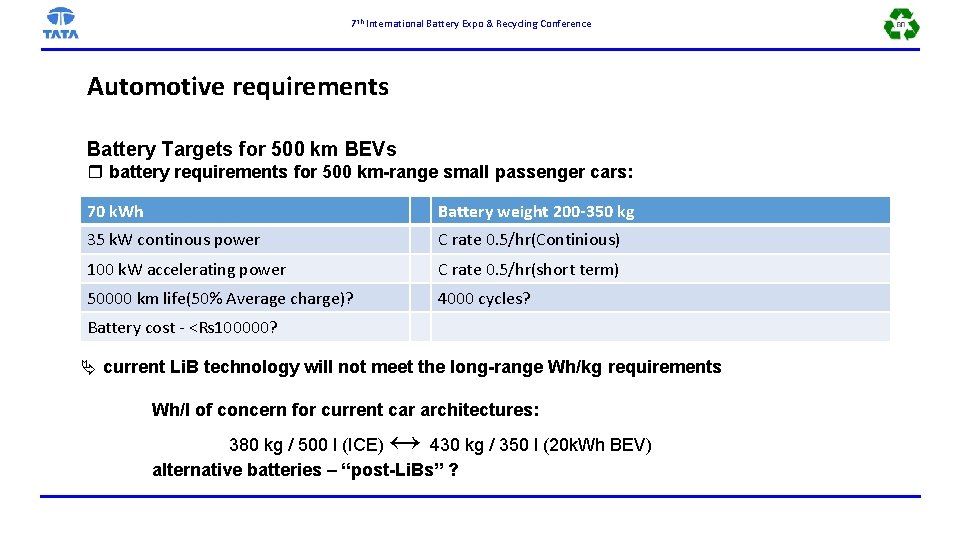
7 th International Battery Expo & Recycling Conference Automotive requirements Battery Targets for 500 km BEVs r battery requirements for 500 km-range small passenger cars: 70 k. Wh Battery weight 200 -350 kg 35 k. W continous power C rate 0. 5/hr(Continious) 100 k. W accelerating power C rate 0. 5/hr(short term) 50000 km life(50% Average charge)? 4000 cycles? Battery cost - <Rs 100000? current Li. B technology will not meet the long-range Wh/kg requirements Wh/l of concern for current car architectures: ↔ 380 kg / 500 l (ICE) 430 kg / 350 l (20 k. Wh BEV) alternative batteries – “post-Li. Bs” ?

7 th International Battery Expo & Recycling Conference Alternate plan for replacement of Li-Ion battery Lithium cost Availability of lithium in India Availability of Sodium Development sodium based Batteries
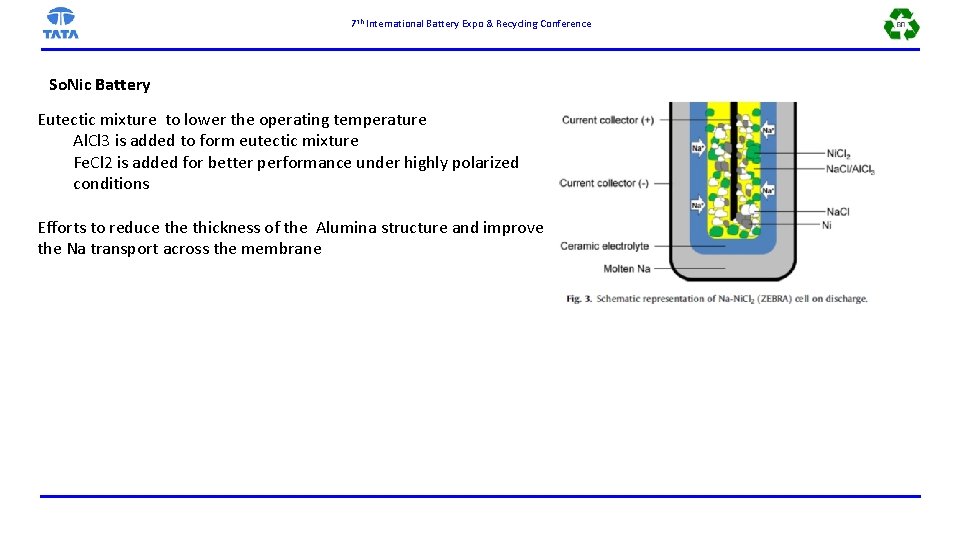
7 th International Battery Expo & Recycling Conference So. Nic Battery Eutectic mixture to lower the operating temperature Al. Cl 3 is added to form eutectic mixture Fe. Cl 2 is added for better performance under highly polarized conditions Efforts to reduce thickness of the Alumina structure and improve the Na transport across the membrane
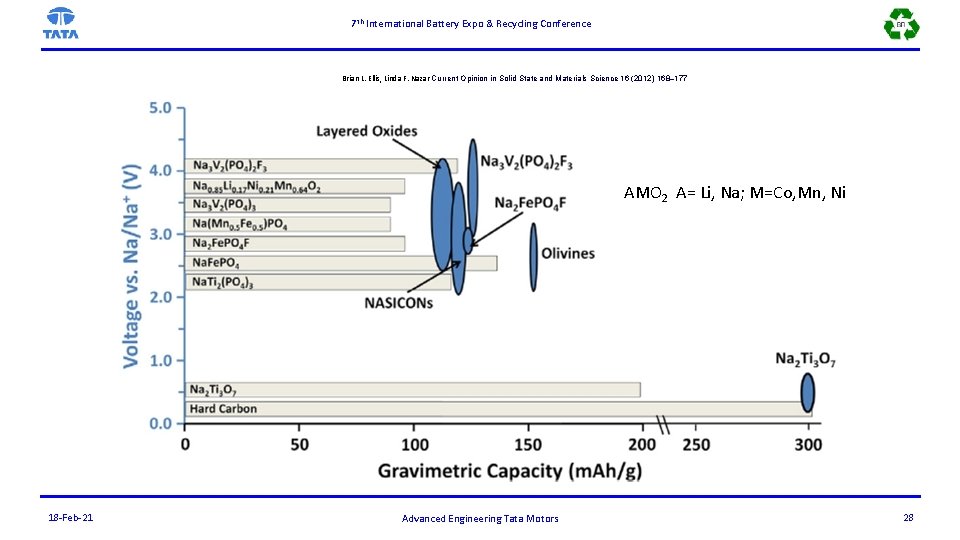
7 th International Battery Expo & Recycling Conference Brian L. Ellis, Linda F. Nazar Current Opinion in Solid State and Materials Science 16 (2012) 168– 177 AMO 2 A= Li, Na; M=Co, Mn, Ni 18 -Feb-21 Advanced Engineering Tata Motors 28
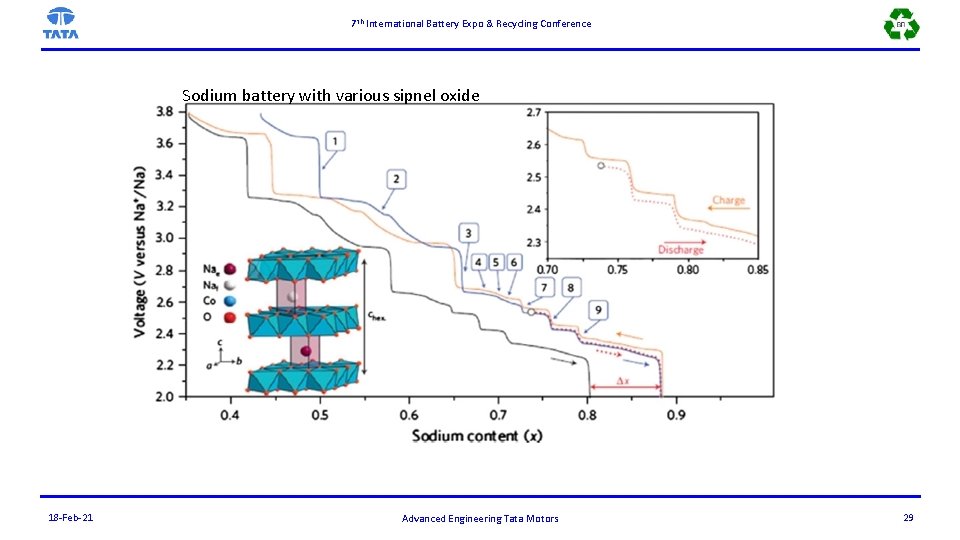
7 th International Battery Expo & Recycling Conference Sodium battery with various sipnel oxide 18 -Feb-21 Advanced Engineering Tata Motors 29
7 th International Battery Expo & Recycling Conference 18 -Feb-21 Advanced Engineering Tata Motors 30

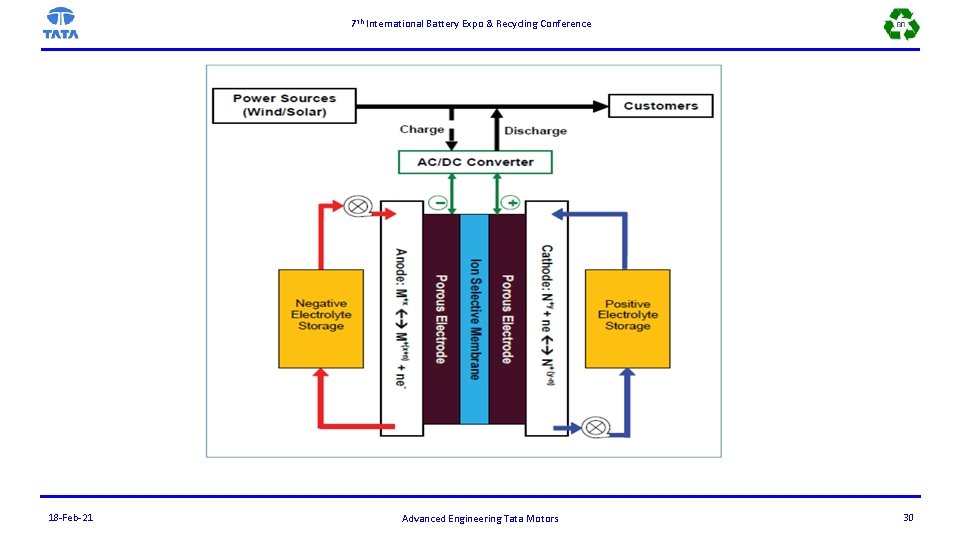
7 th International Battery Expo & Recycling Conference Advantages and Disadvantages Electroactive materials stored externally -separation of the power and energy requirements. The electrodes can be designed to have optimal power acceptance and delivery properties (e. g. , catalytic, electrical, and transport) Furthermore, the electrodes do not undergo physical and chemical changes during operation (because they do not contain active materials), thus leading to more stable and durable performance. Therefore, engineered microstructures developed to optimize performance can be maintained over the lifetime of the device: with longer lifetimes, the capital costs of the battery system can be amortized over a longer period, and with a wider state-of-charge operating window, the quantity of active material required to deliver power over the entire required duration of discharge can be minimized. 18 -Feb-21 Advanced Engineering Tata Motors 31
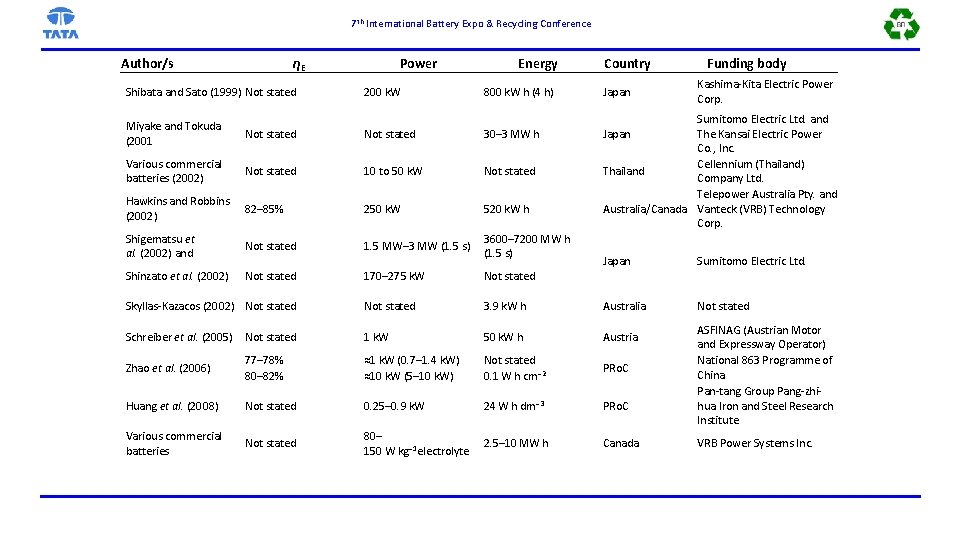
7 th International Battery Expo & Recycling Conference Author/s ηE Shibata and Sato (1999) Not stated Power 200 k. W Energy 800 k. W h (4 h) Miyake and Tokuda (2001 Not stated 30– 3 MW h Various commercial batteries (2002) Not stated 10 to 50 k. W Not stated Hawkins and Robbins (2002) 82– 85% 250 k. W 520 k. W h Shigematsu et al. (2002) and Not stated 1. 5 MW– 3 MW (1. 5 s) 3600– 7200 MW h (1. 5 s) Shinzato et al. (2002) Not stated Country Japan Funding body Kashima-Kita Electric Power Corp. Sumitomo Electric Ltd. and Japan The Kansai Electric Power Co. , Inc. Cellennium (Thailand) Thailand Company Ltd. Telepower Australia Pty. and Australia/Canada Vanteck (VRB) Technology Corp. Japan Sumitomo Electric Ltd. Not stated 170– 275 k. W Not stated Skyllas-Kazacos (2002) Not stated 3. 9 k. W h Australia Schreiber et al. (2005) Not stated 1 k. W 50 k. W h Austria Zhao et al. (2006) 77– 78% 80– 82% ≈1 k. W (0. 7– 1. 4 k. W) ≈10 k. W (5– 10 k. W) Not stated 0. 1 W h cm− 2 PRo. C Huang et al. (2008) Not stated 0. 25– 0. 9 k. W 24 W h dm− 3 PRo. C Various commercial batteries Not stated 80– 150 W kg− 1 electrolyte 2. 5– 10 MW h Canada ASFINAG (Austrian Motor and Expressway Operator) National 863 Programme of China Pan-tang Group Pang-zhihua Iron and Steel Research Institute VRB Power Systems Inc.
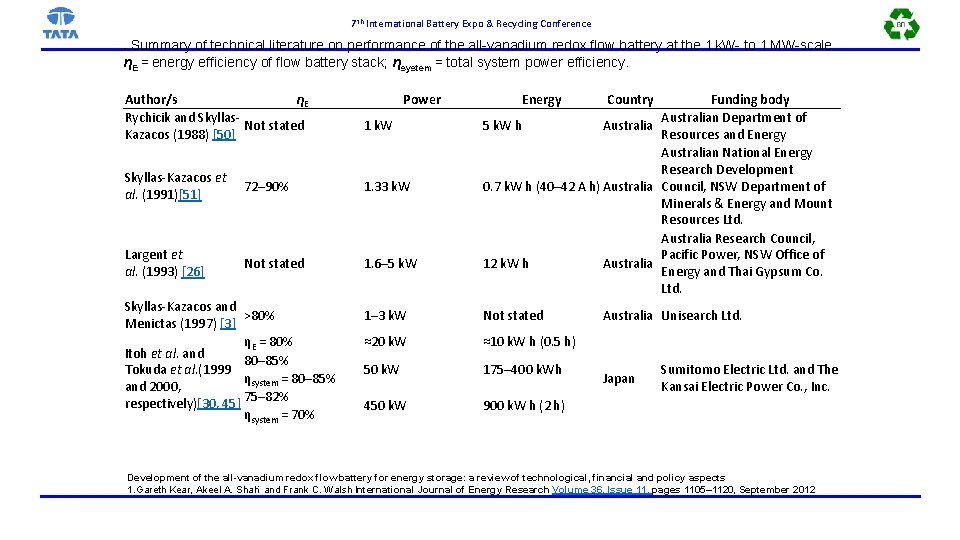
7 th International Battery Expo & Recycling Conference . Summary of technical literature on performance of the all-vanadium redox flow battery at the 1 k. W- to 1 MW-scale. ηE = energy efficiency of flow battery stack; ηsystem = total system power efficiency. Author/s ηE Rychicik and Skyllas. Not stated Kazacos (1988) [50] Power 1 k. W Skyllas-Kazacos et al. (1991)[51] 72– 90% 1. 33 k. W Largent et al. (1993) [26] Not stated 1. 6– 5 k. W Skyllas-Kazacos and >80% Menictas (1997) [3] ηE = 80% Itoh et al. and 80– 85% Tokuda et al. (1999 ηsystem = 80– 85% and 2000, respectively)[30, 45] 75– 82% ηsystem = 70% Energy Funding body Australian Department of 5 k. W h Australia Resources and Energy Australian National Energy Research Development 0. 7 k. W h (40– 42 A h) Australia Council, NSW Department of Minerals & Energy and Mount Resources Ltd. Australia Research Council, Pacific Power, NSW Office of 12 k. W h Australia Energy and Thai Gypsum Co. Ltd. 1– 3 k. W Not stated ≈20 k. W ≈10 k. W h (0. 5 h) 50 k. W 175– 400 k. Wh 450 k. W 900 k. W h (2 h) Country Australia Unisearch Ltd. Japan Sumitomo Electric Ltd. and The Kansai Electric Power Co. , Inc. Development of the all-vanadium redox flow battery for energy storage: a review of technological, financial and policy aspects 1. Gareth Kear, Akeel A. Shah* and Frank C. Walsh International Journal of Energy Research Volume 36, Issue 11, pages 1105– 1120, September 2012

7 th International Battery Expo & Recycling Conference Thanks
- Slides: 34