7 Carbohydrates and Glycobiology 2017 W H Freeman

7 | Carbohydrates and Glycobiology © 2017 W. H. Freeman and Company

Energy storage Structural materials and more…. Carbohydrates are the most abundant biological molecule. (C·H 2 O)n (n≥ 3) Chapter 8 Opener

Carbohydrates • Named so because many have formula Cn(H 2 O)n • Produced from CO 2 and H 2 O via photosynthesis in plants • Range from as small as glyceraldehyde (Mw = 90 g/mol) to as large as amylopectin (Mw > 200, 000 g/mol) • Fulfill a variety of functions, including: – energy source and energy storage – structural component of cell walls and exoskeletons – informational molecules in cell-cell signaling • Can be covalently linked with proteins and lipids

Carbohydrates • Basic nomenclature: – number of carbon atoms in the carbohydrate + -ose – example: three carbons = triose • Common functional groups: – All carbohydrates initially had a carbonyl functional group. – aldehydes = aldose – ketones = ketose
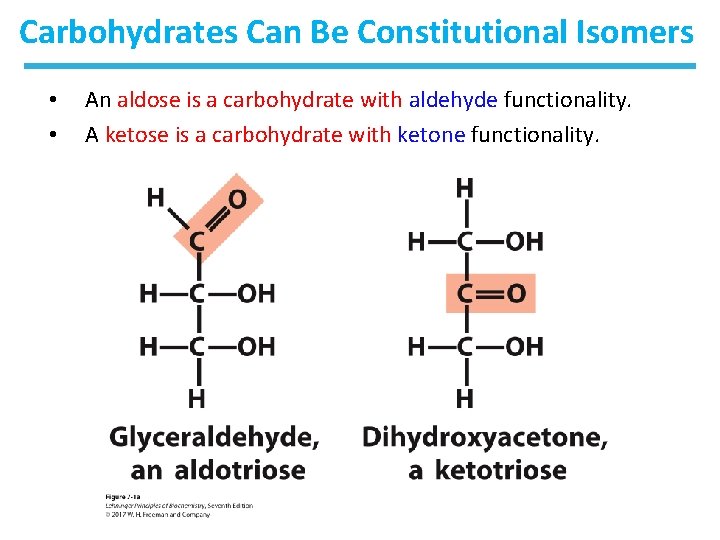
Carbohydrates Can Be Constitutional Isomers • • An aldose is a carbohydrate with aldehyde functionality. A ketose is a carbohydrate with ketone functionality.
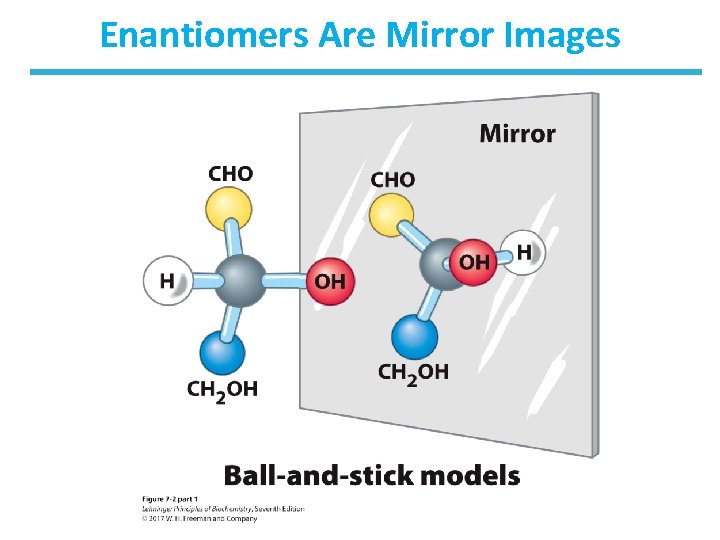
Enantiomers Are Mirror Images

Carbohydrates Can Be Stereoisomers • Enantiomers – stereoisomers that are nonsuperimposable mirror images • In sugars that contain many chiral centers, only the one that is most distant from the carbonyl carbon is designated as d (right) or l (left). • d and l isomers of a sugar are enantiomers. – For example, l- and d-glucose have the same water solubility. • Most hexoses in living organisms are d stereoisomers. • Some simple sugars occur in the l form, such as l-arabinose.
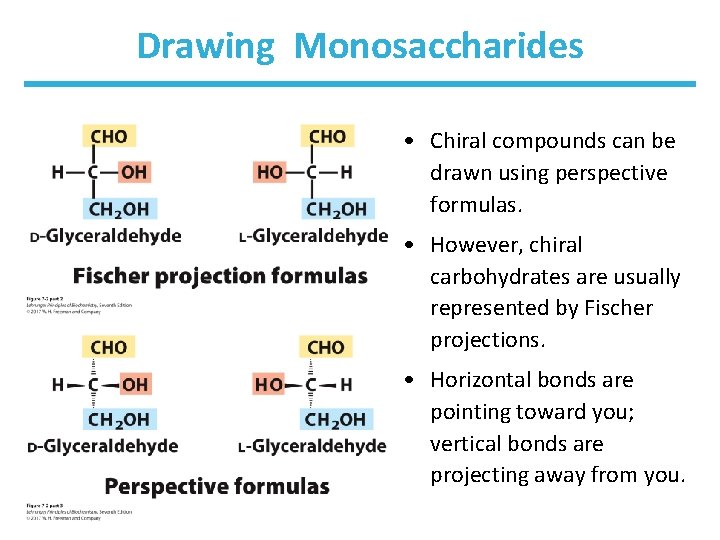
Drawing Monosaccharides • Chiral compounds can be drawn using perspective formulas. • However, chiral carbohydrates are usually represented by Fischer projections. • Horizontal bonds are pointing toward you; vertical bonds are projecting away from you.

Carbohydrates Can Be Stereoisomers • Epimers are stereoisomers that differ at only one chiral center. • Epimers are NOT mirror images, and therefore are NOT enantiomers. • Epimers are diastereomers; diastereomers have different physical properties (i. e. , water solubility, melting temp). • example: D-Threose is the C-2 epimer of D-erythrose. • Both are D sugars because they both have the same orientation around the last chiral carbon in the chain.
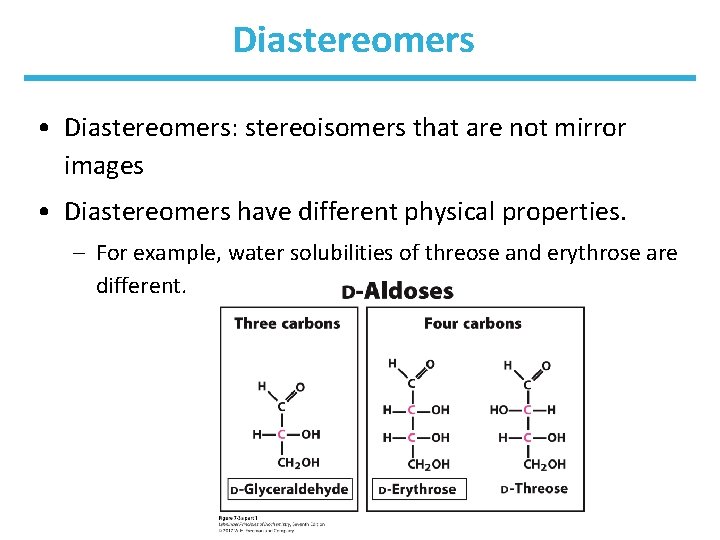
Diastereomers • Diastereomers: stereoisomers that are not mirror images • Diastereomers have different physical properties. – For example, water solubilities of threose and erythrose are different.
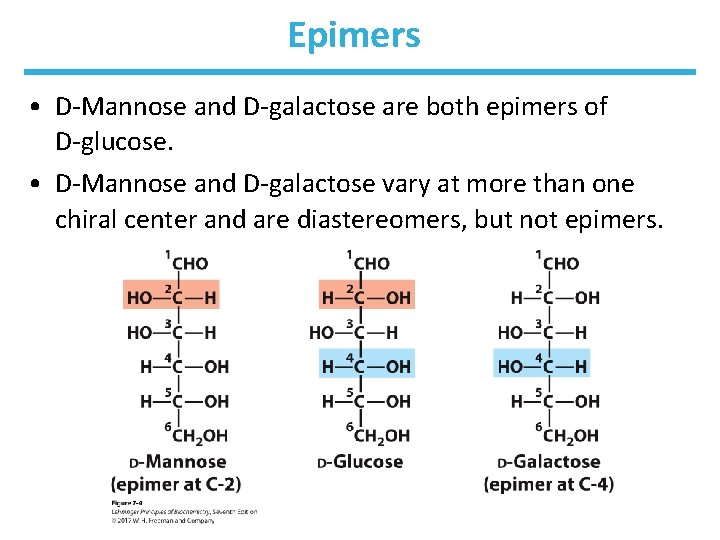
Epimers • D-Mannose and D-galactose are both epimers of D-glucose. • D-Mannose and D-galactose vary at more than one chiral center and are diastereomers, but not epimers.

Common Carbohydrates in Biochemistry • • • Ribose is the standard five-carbon sugar. Glucose is the standard six-carbon sugar. Galactose is an epimer of glucose. Mannose is an epimer of glucose. Fructose is the ketose form of glucose.
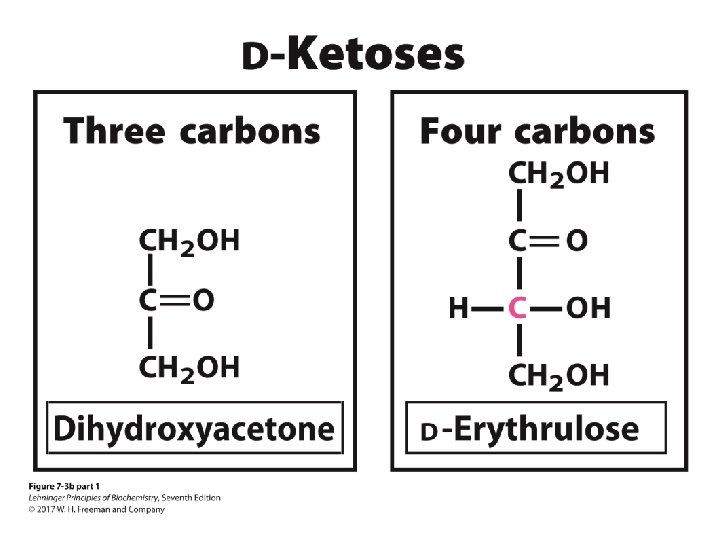
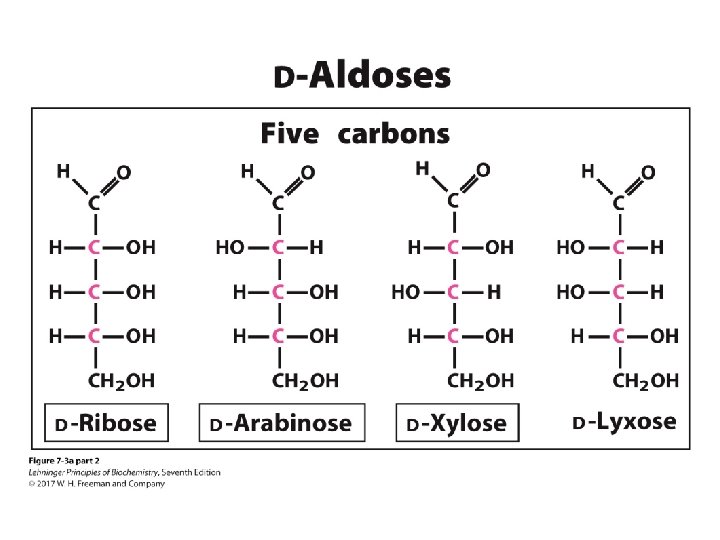
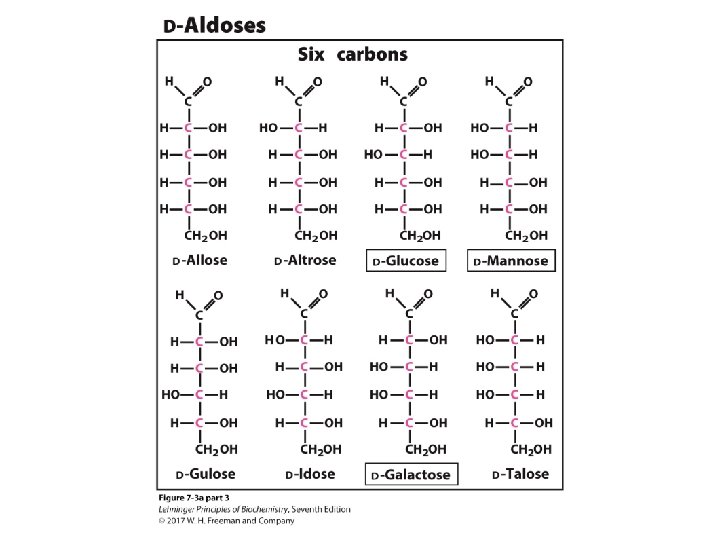
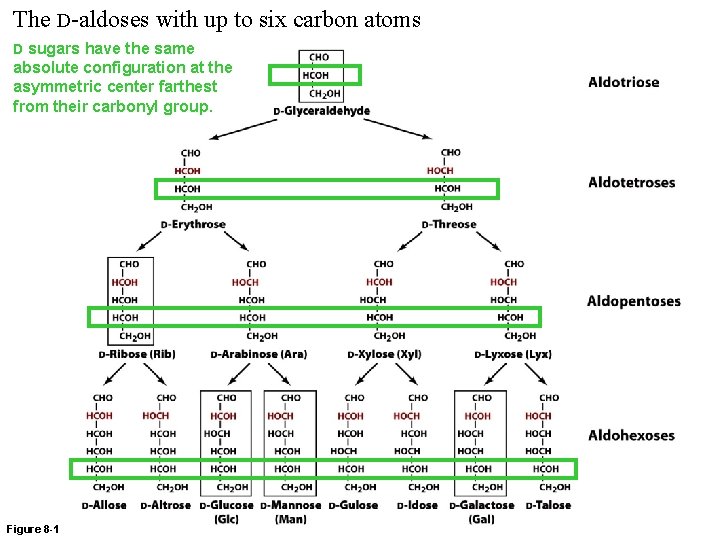
The D-aldoses with up to six carbon atoms D sugars have the same absolute configuration at the asymmetric center farthest from their carbonyl group. Figure 8 -1
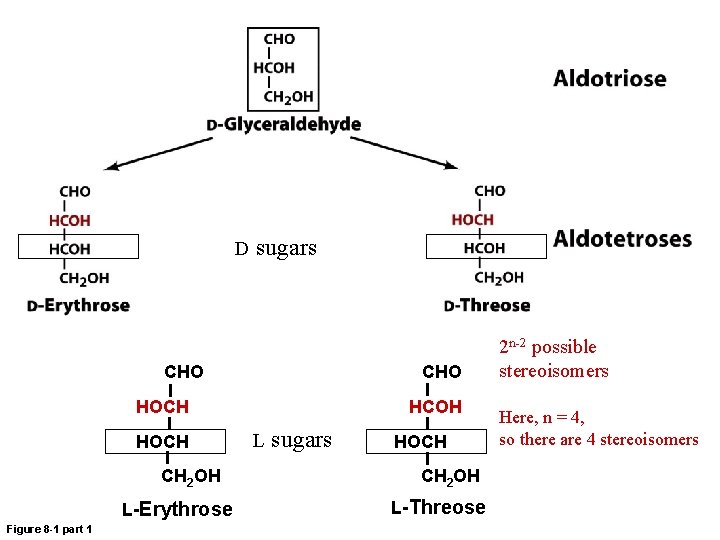
D sugars CHO HOCH CH 2 OH L-Erythrose Figure 8 -1 part 1 HCOH L sugars HOCH CH 2 OH L-Threose 2 n-2 possible stereoisomers Here, n = 4, so there are 4 stereoisomers
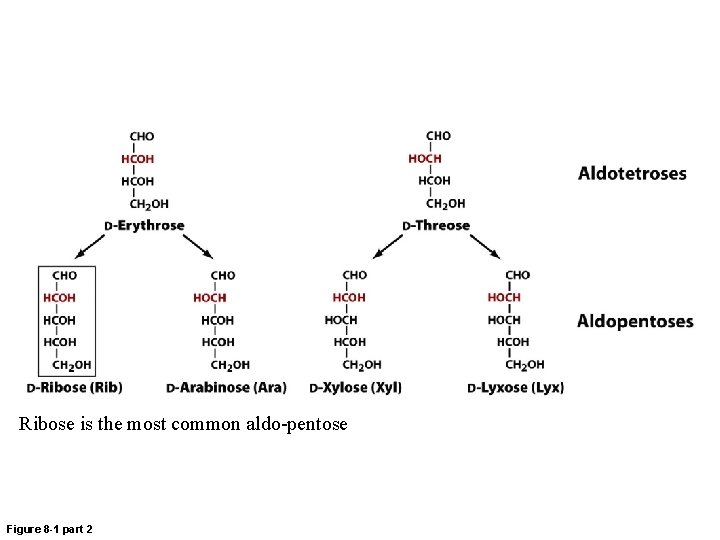
Ribose is the most common aldo-pentose Figure 8 -1 part 2
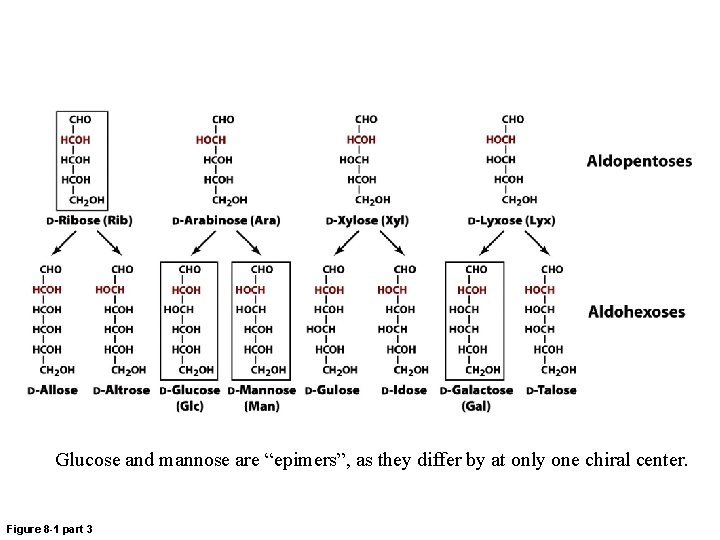
Glucose and mannose are “epimers”, as they differ by at only one chiral center. Figure 8 -1 part 3
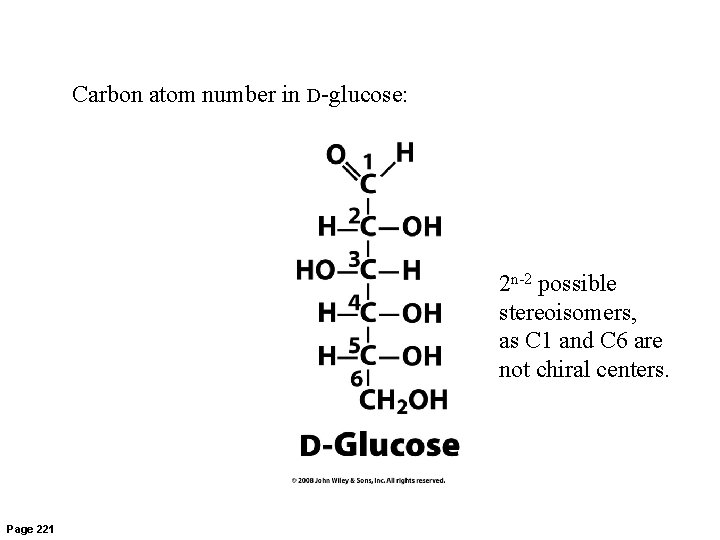
Carbon atom number in D-glucose: 2 n-2 possible stereoisomers, as C 1 and C 6 are not chiral centers. Page 221
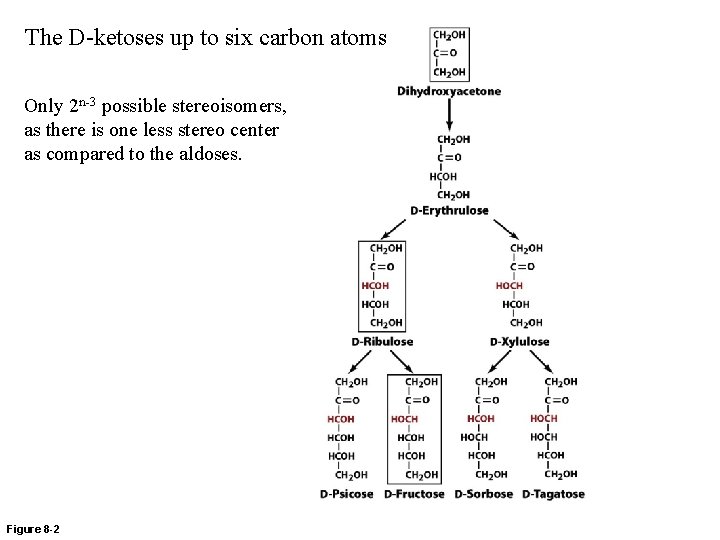
The D-ketoses up to six carbon atoms Only 2 n-3 possible stereoisomers, as there is one less stereo center as compared to the aldoses. Figure 8 -2
These ketoses will be seen again when we cover to metabolism. Figure 8 -2
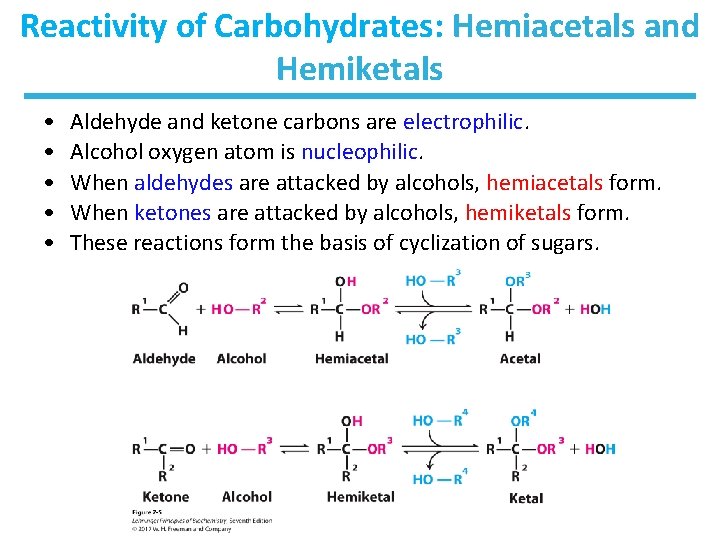
Reactivity of Carbohydrates: Hemiacetals and Hemiketals • • • Aldehyde and ketone carbons are electrophilic. Alcohol oxygen atom is nucleophilic. When aldehydes are attacked by alcohols, hemiacetals form. When ketones are attacked by alcohols, hemiketals form. These reactions form the basis of cyclization of sugars.
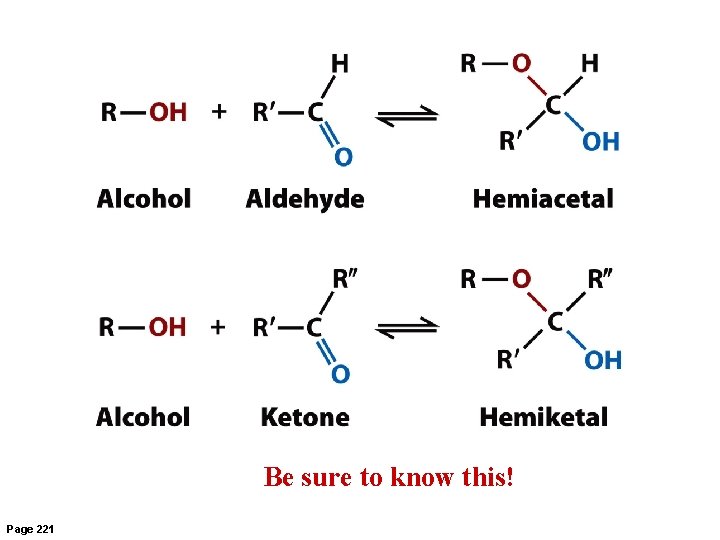
Be sure to know this! Page 221
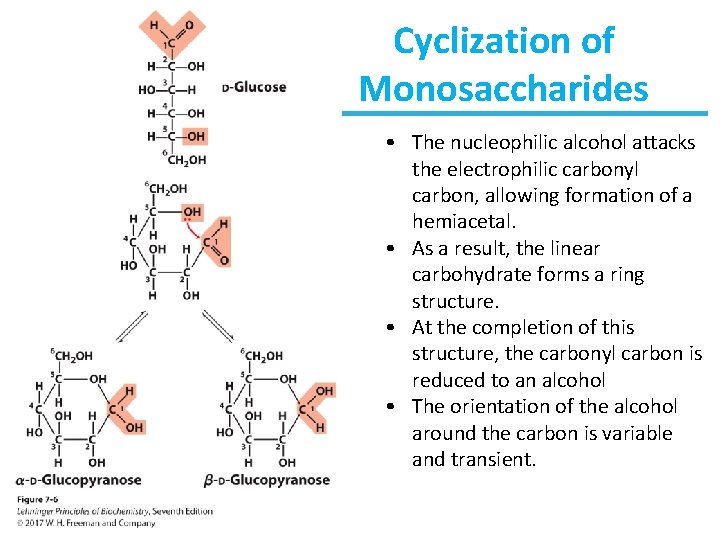
Cyclization of Monosaccharides • The nucleophilic alcohol attacks the electrophilic carbonyl carbon, allowing formation of a hemiacetal. • As a result, the linear carbohydrate forms a ring structure. • At the completion of this structure, the carbonyl carbon is reduced to an alcohol • The orientation of the alcohol around the carbon is variable and transient.
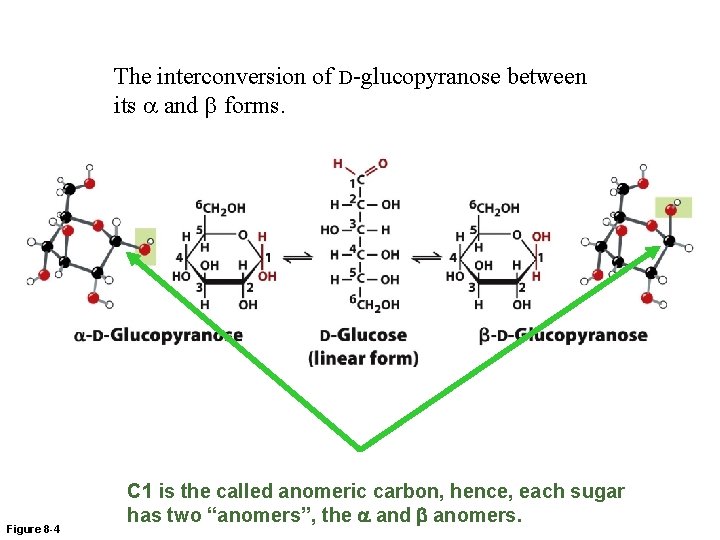
The interconversion of D-glucopyranose between its a and b forms. Figure 8 -4 C 1 is the called anomeric carbon, hence, each sugar has two “anomers”, the a and b anomers.

Cyclization of Monosaccharides • Pentoses and hexoses readily undergo intramolecular cyclization. • The former carbonyl carbon becomes a new chiral center, called the anomeric carbon. – When the former carbonyl oxygen becomes a hydroxyl group, the position of this group determines if the anomer is or . – If the hydroxyl group is on the opposite side (trans) of the ring as the CH 2 OH moiety, the configuration is . – If the hydroxyl group is on the same side (cis) of the ring as the CH 2 OH moiety, the configuration is .
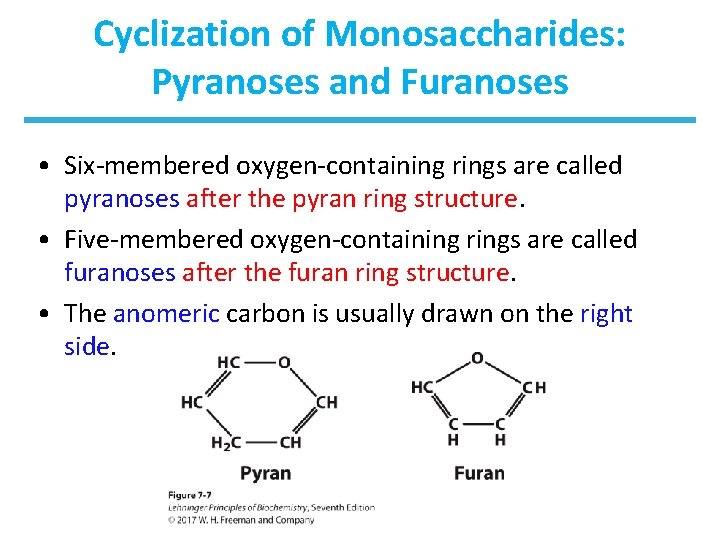
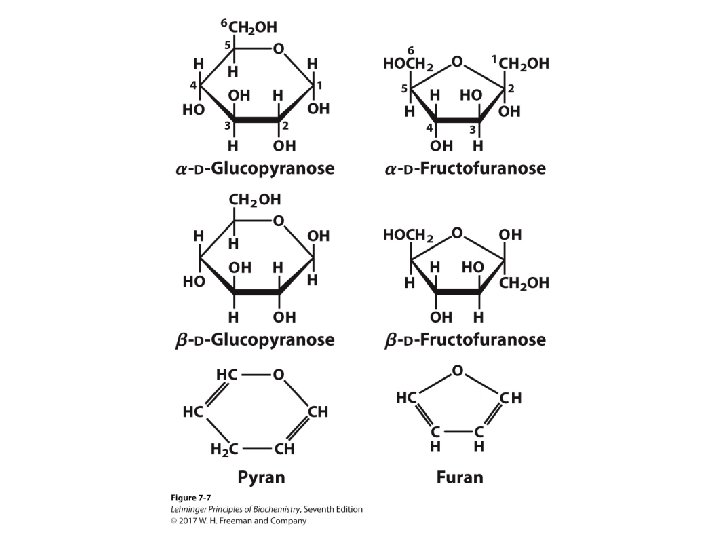
Cyclization of Monosaccharides: Pyranoses and Furanoses • Six-membered oxygen-containing rings are called pyranoses after the pyran ring structure. • Five-membered oxygen-containing rings are called furanoses after the furan ring structure. • The anomeric carbon is usually drawn on the right side.
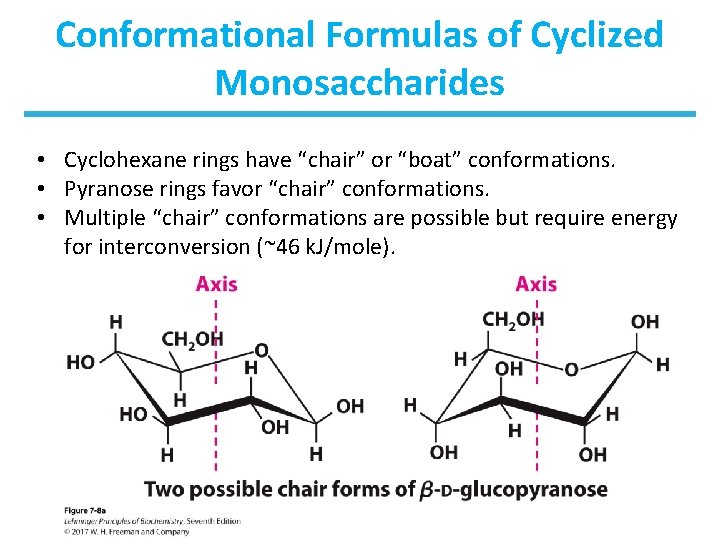
Conformational Formulas of Cyclized Monosaccharides • Cyclohexane rings have “chair” or “boat” conformations. • Pyranose rings favor “chair” conformations. • Multiple “chair” conformations are possible but require energy for interconversion (~46 k. J/mole).
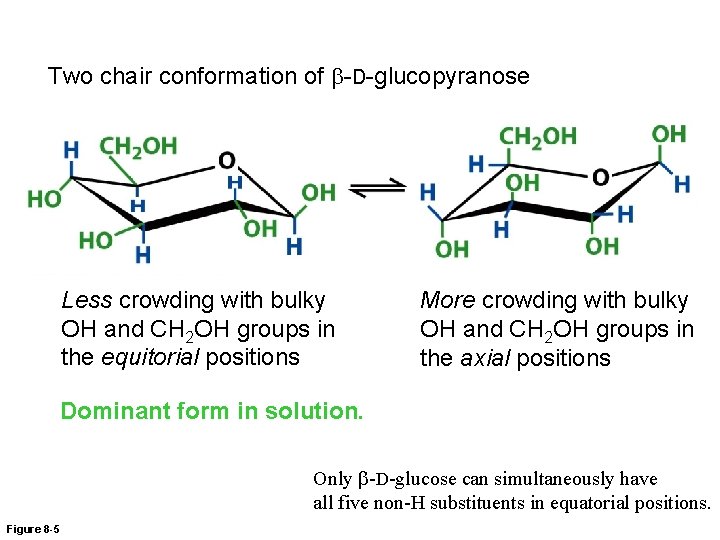
Two chair conformation of b-D-glucopyranose Less crowding with bulky OH and CH 2 OH groups in the equitorial positions More crowding with bulky OH and CH 2 OH groups in the axial positions Dominant form in solution. Only b-D-glucose can simultaneously have all five non-H substituents in equatorial positions. Figure 8 -5
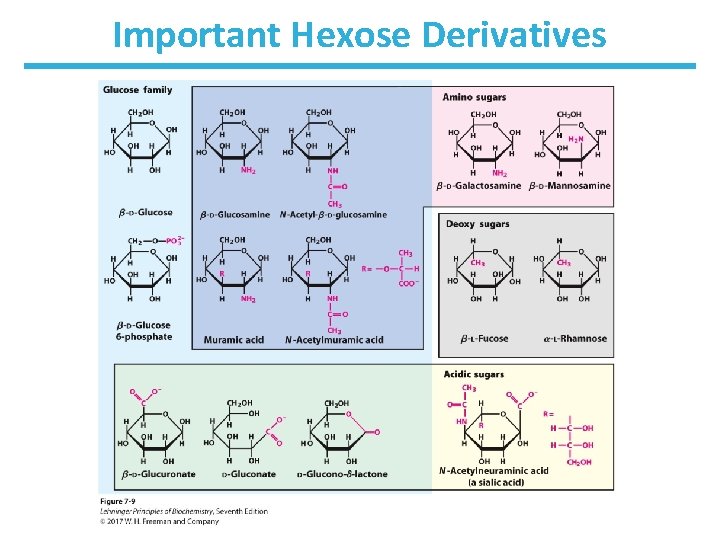
Important Hexose Derivatives
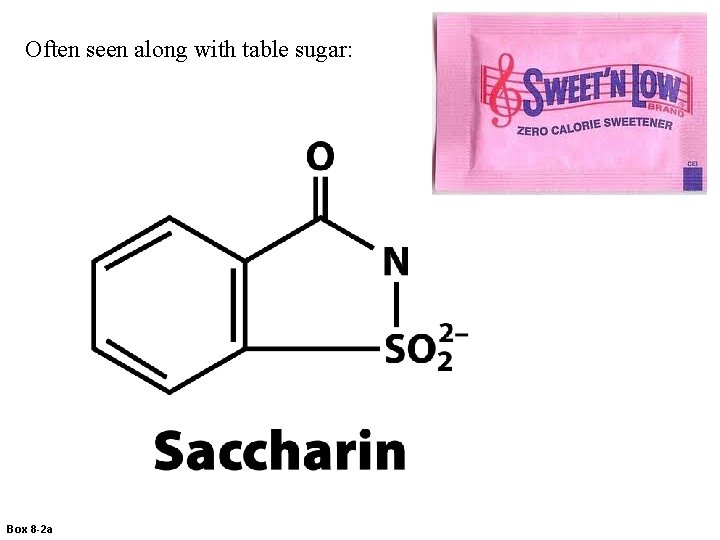
Often seen along with table sugar: Box 8 -2 a
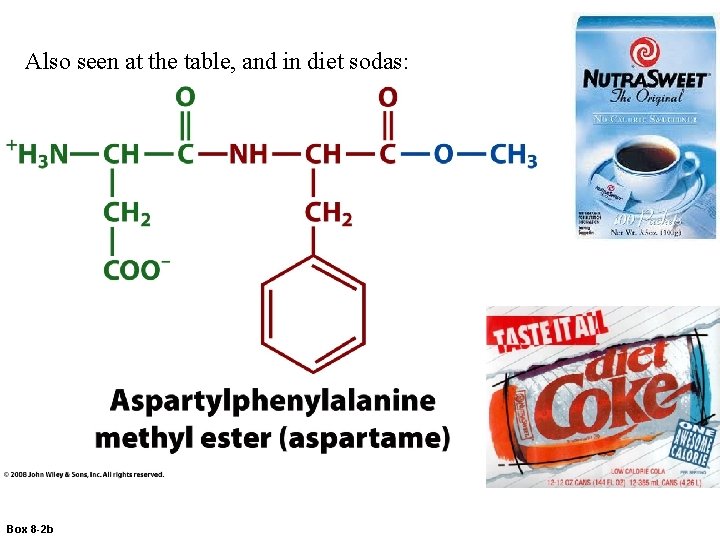
Also seen at the table, and in diet sodas: Box 8 -2 b
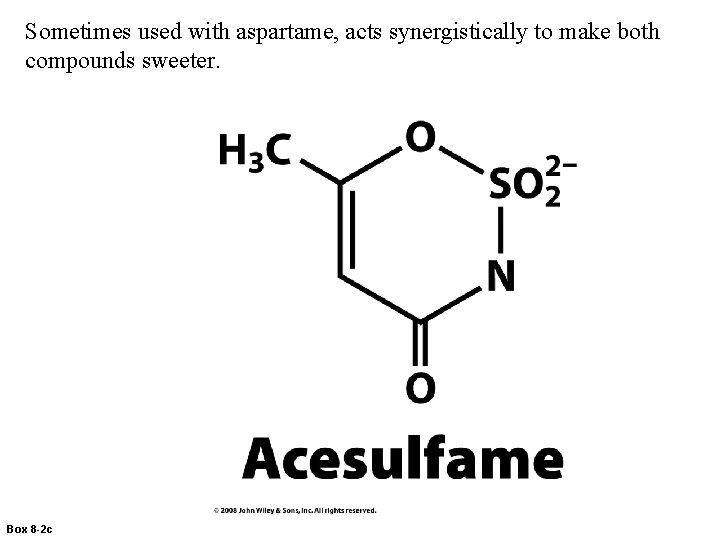
Sometimes used with aspartame, acts synergistically to make both compounds sweeter. Box 8 -2 c
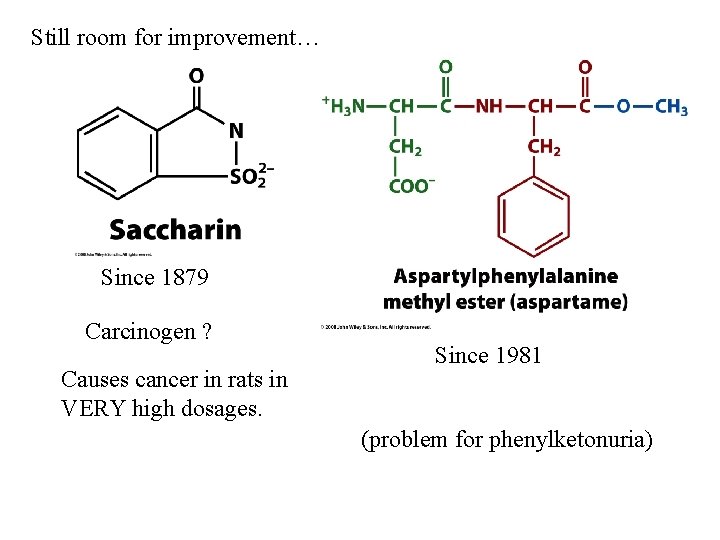
Still room for improvement… Since 1879 Carcinogen ? Causes cancer in rats in VERY high dosages. Since 1981 (problem for phenylketonuria)

Reducing Sugars • Reducing sugars have a free anomeric carbon. – – Aldehyde can reduce Cu 2+ to Cu+ (Fehling’s test). Aldehyde can reduce Ag+ to Ag 0 (Tollens’ test). It allows detection of reducing sugars, such as glucose. Modern detection techniques use colorimetric and electrochemical tests.
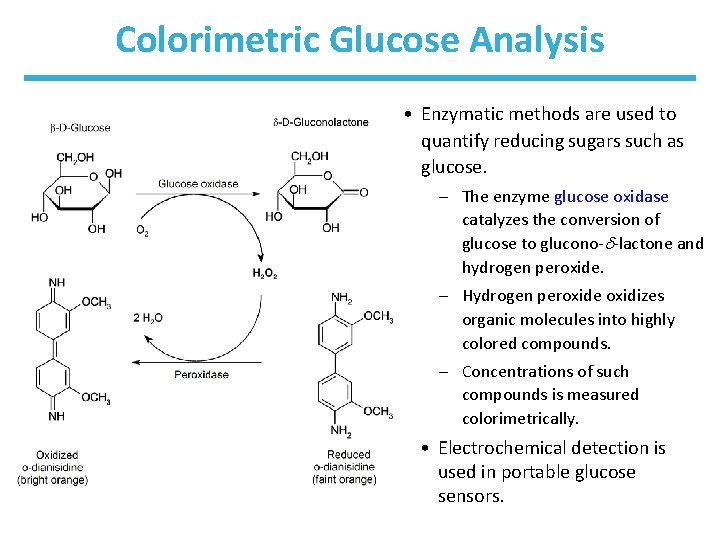
Colorimetric Glucose Analysis • Enzymatic methods are used to quantify reducing sugars such as glucose. – The enzyme glucose oxidase catalyzes the conversion of glucose to glucono- -lactone and hydrogen peroxide. – Hydrogen peroxide oxidizes organic molecules into highly colored compounds. – Concentrations of such compounds is measured colorimetrically. • Electrochemical detection is used in portable glucose sensors.

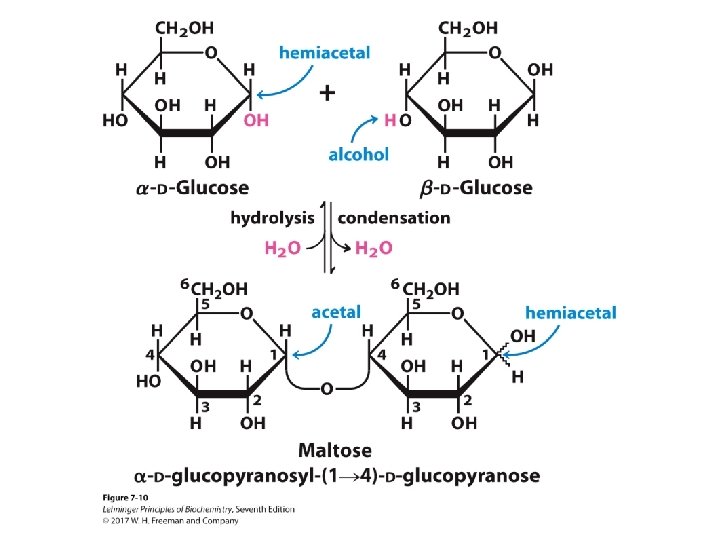
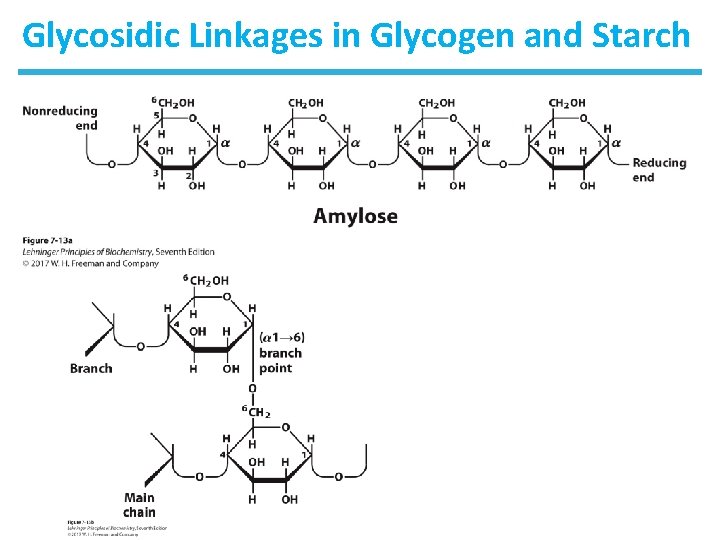
The Glycosidic Bond • Two sugar molecules can be joined via a glycosidic bond between an anomeric carbon and a hydroxyl carbon. • The glycosidic bond (an acetal) between monomers is more stable and less reactive than the hemiacetal at the second monomer. – The second monomer, with the hemiacetal, is reducing. – The anomeric carbon involved in the glycosidic linkage is nonreducing. • Disacharides can be named by the organization and linkage or a common name. – The disaccharide formed upon condensation of two glucose molecules via a 1 4 bond is described as α-D-glucopyranosyl(1 4)-D-glucopyranose. – The common name for this disaccharide is maltose.
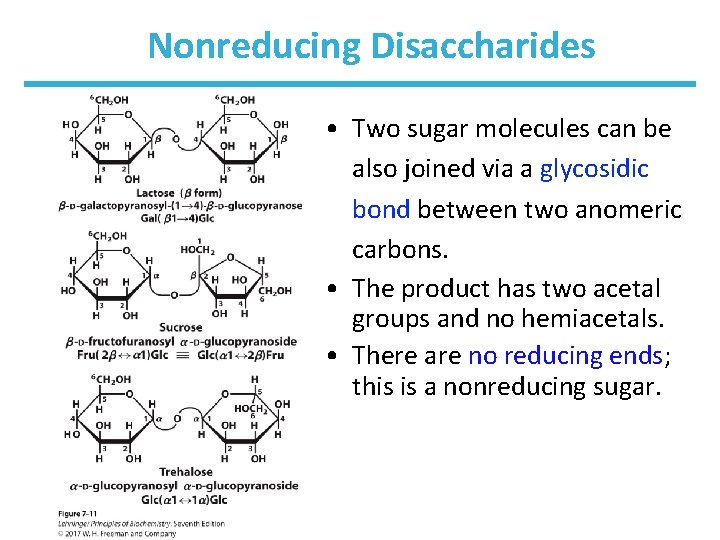
Nonreducing Disaccharides • Two sugar molecules can be also joined via a glycosidic bond between two anomeric carbons. • The product has two acetal groups and no hemiacetals. • There are no reducing ends; this is a nonreducing sugar.

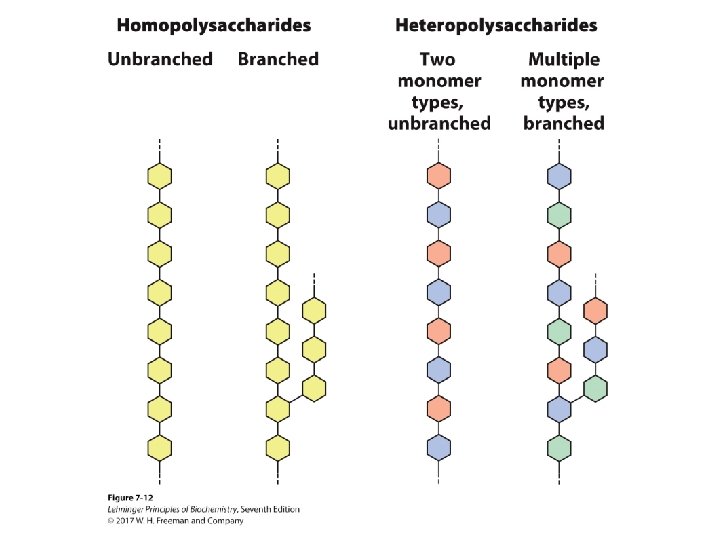
Polysaccharides • Natural carbohydrates are usually found as polymers. • These polysaccharides can be: – homopolysaccharides (one monomer unit) – heteropolysaccharides (multiple monomer units) – linear (one type of glycosidic bond) – branched (multiple types of glycosidic bonds) • Polysaccharides do not have a defined molecular weight. – This is in contrast to proteins because, unlike proteins, no template is used to make polysaccharides. – Polysaccharides are often in a state of flux; monomer units are added and removed as needed by the organism.

Homopolymers of Glucose: Glycogen • Glycogen is a branched homopolysaccharide of glucose. – Glucose monomers form ( 1 4) linked chains. – There are branch points with ( 1 6) linkers every 8– 12 residues. – Molecular weight reaches several millions. – It functions as the main storage polysaccharide in animals.

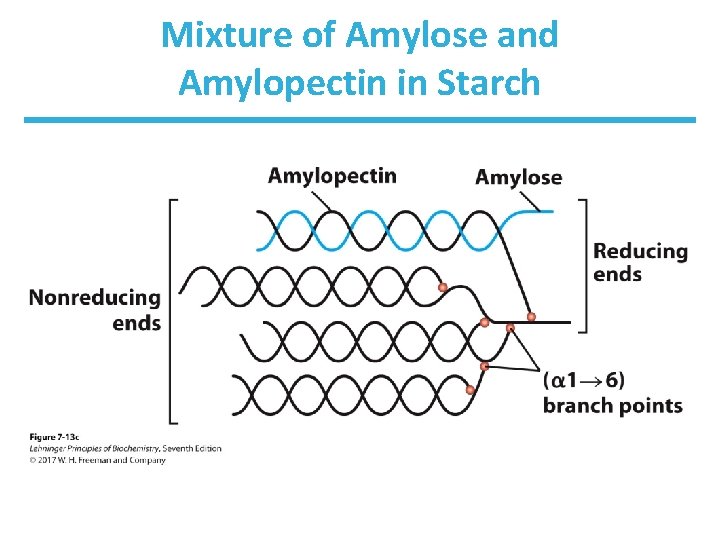
Homopolymers of Glucose: Starch • Starch is a mixture of two homopolysaccharides of glucose. • Amylose is an unbranched polymer of ( 1 4) linked residues. • Amylopectin is branched like glycogen, but the branch points with ( 1 6) linkers occur every 24 – 30 residues. • Molecular weight of amylopectin is up to 200 million. • Starch is the main storage polysaccharide in plants.
Glycosidic Linkages in Glycogen and Starch
Mixture of Amylose and Amylopectin in Starch

Metabolism of Glycogen and Starch • Glycogen and starch are insoluble due to their high molecular weight and often form granules in cells. • Granules contain enzymes that synthesize and degrade these polymers. • Glycogen and amylopectin have one reducing end but many nonreducing ends. • Enzymatic processing occurs simultaneously in many nonreducing ends.

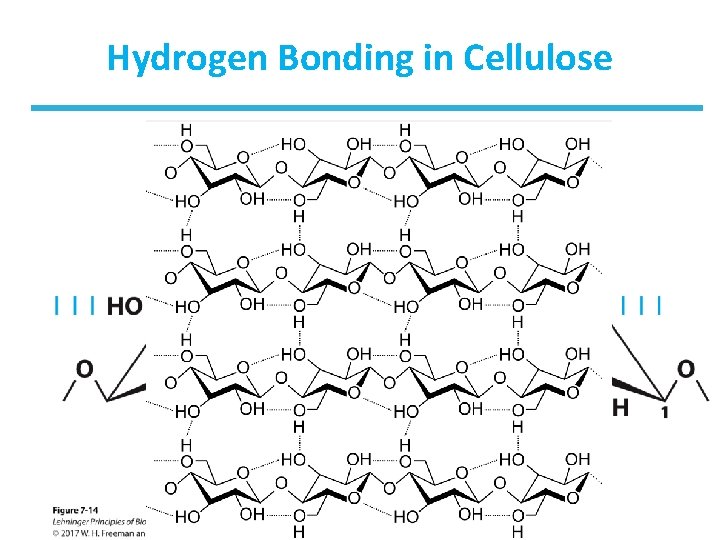
Homopolymers of Glucose: Cellulose • Cellulose is a linear homopolysaccharide of glucose. – Glucose monomers form ( 1 4) linked chains. – Hydrogen bonds form between adjacent monomers. – There additional H-bonds between chains. – Structure is tough and water insoluble. – It is the most abundant polysaccharide in nature. – Cotton is nearly pure fibrous cellulose.
Hydrogen Bonding in Cellulose

Cellulose Metabolism • The fibrous structure and water insolubility make cellulose a difficult substrate to act upon. • Most animals cannot use cellulose as a fuel source because they lack the enzyme to hydrolyze ( 1 4) linkages. • Fungi, bacteria, and protozoa secrete cellulase, which allows them to use wood as source of glucose. • Ruminants and termites live symbiotically with microorganisms that produce cellulase and are able to absorb the freed glucose into their bloodstreams. • Cellulases hold promise in the fermentation of biomass into biofuels.

Q: What do these two have in common? A: They both have a “sugary” shell 100% N-acetylglucosamine. 80% sucrose 10% glucose 10% fructose
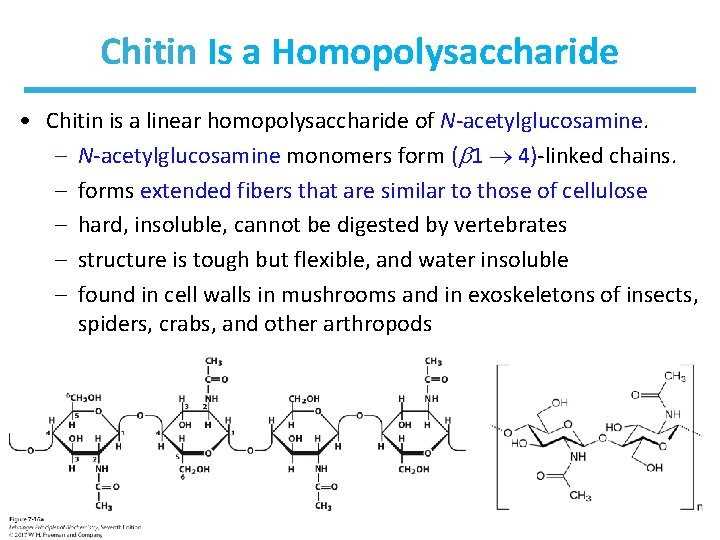
Chitin Is a Homopolysaccharide • Chitin is a linear homopolysaccharide of N-acetylglucosamine. – N-acetylglucosamine monomers form ( 1 4)-linked chains. – forms extended fibers that are similar to those of cellulose – hard, insoluble, cannot be digested by vertebrates – structure is tough but flexible, and water insoluble – found in cell walls in mushrooms and in exoskeletons of insects, spiders, crabs, and other arthropods

Agar and Agarose • Agar is a branched heteropolysaccharide composed of agarose and agaropectin. • Agar serves as a component of cell wall in some seaweeds. • Agar solutions form gels that are commonly used in the laboratory as a surface for growing bacteria. • Agarose solutions form gels that are commonly used in the laboratory for separation DNA by electrophoresis.
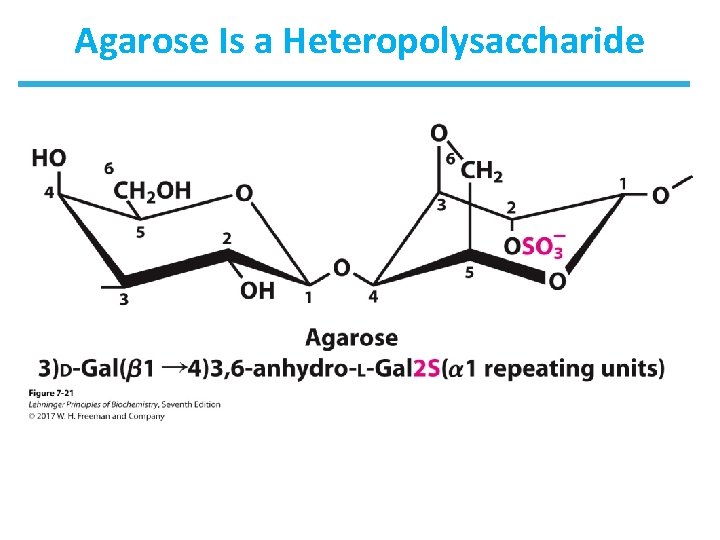
Agarose Is a Heteropolysaccharide

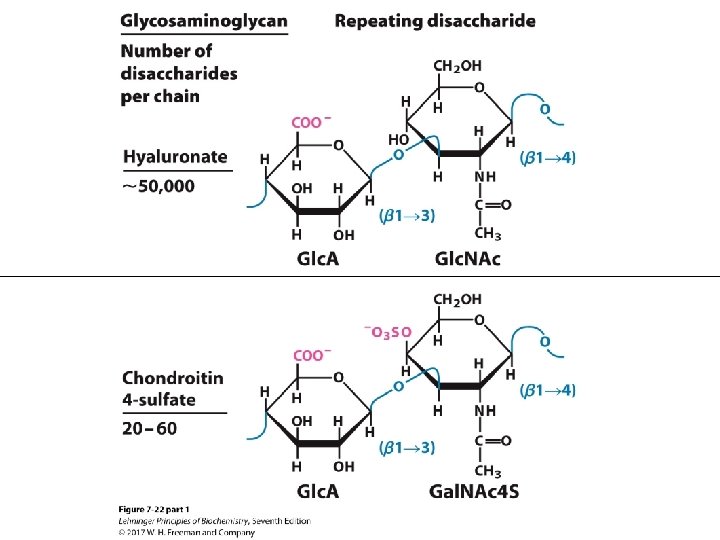
Glycosaminoglycans • Linear polymers of repeating disaccharide units • One monomer is either: – N-acetyl-glucosamine or – N-acetyl-galactosamine • Negatively charged – uronic acids (C 6 oxidation) – sulfate esters • Extended hydrated molecule – minimizes charge repulsion • Forms meshwork with fibrous proteins to form extracellular matrix – connective tissue – lubrication of joints
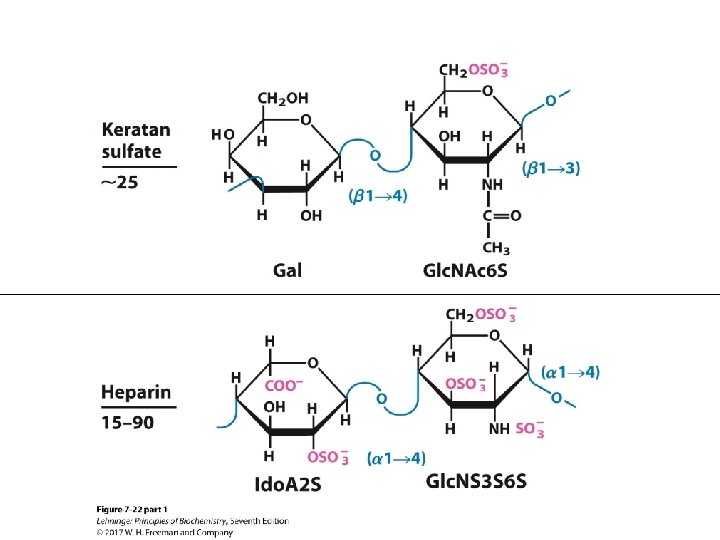

Heparin and Heparan Sulfate • Heparin is linear polymer, 3– 40 k. Da. • Heparan sulfate is heparin-like polysaccharide but attached to proteins. • Highest negative-charge density biomolecules • Prevent blood clotting by activating protease inhibitor antithrombin • Binding to various cells regulates development and formation of blood vessels. • Can also bind to viruses and bacteria and decrease their virulence
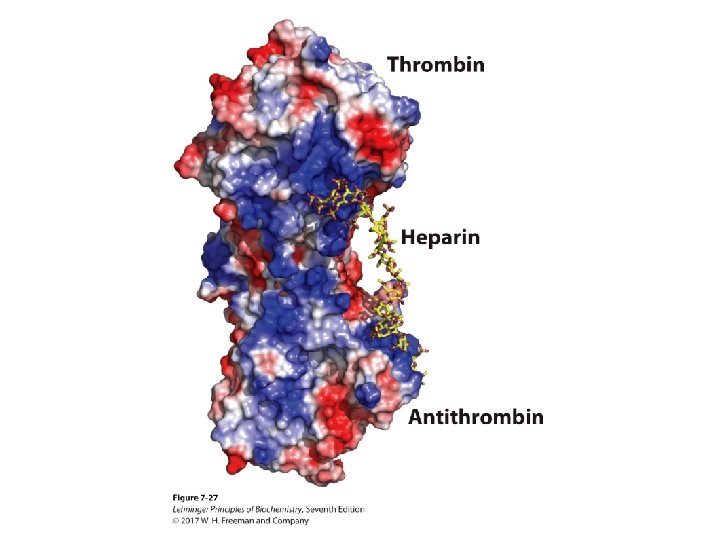
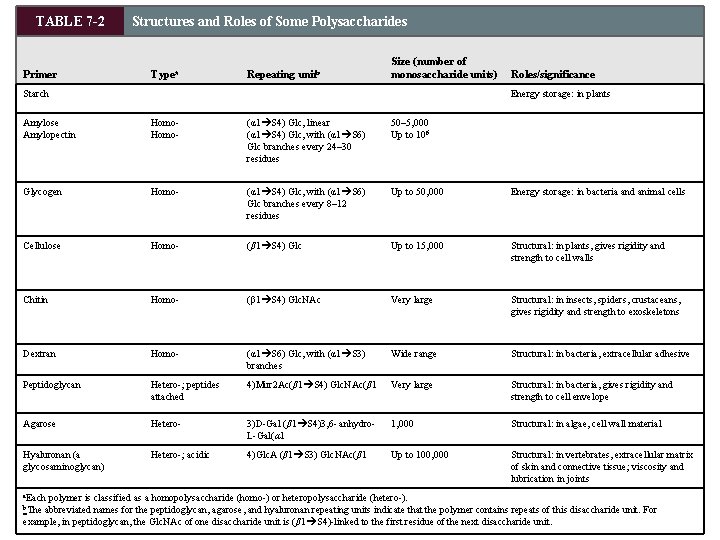
TABLE 7 -2 Primer Structures and Roles of Some Polysaccharides Typea Repeating unitb Size (number of monosaccharide units) Starch Roles/significance Energy storage: in plants Amylose Amylopectin Homo- (α 1 S 4) Glc, linear (α 1 S 4) Glc, with (α 1 S 6) Glc branches every 24– 30 residues 50– 5, 000 Up to 106 Glycogen Homo- (α 1 S 4) Glc, with (α 1 S 6) Glc branches every 8– 12 residues Up to 50, 000 Energy storage: in bacteria and animal cells Cellulose Homo- (β 1 S 4) Glc Up to 15, 000 Structural: in plants, gives rigidity and strength to cell walls Chitin Homo- (β 1 S 4) Glc. NAc Very large Structural: in insects, spiders, crustaceans, gives rigidity and strength to exoskeletons Dextran Homo- (α 1 S 6) Glc, with (α 1 S 3) branches Wide range Structural: in bacteria, extracellular adhesive Peptidoglycan Hetero-; peptides attached 4)Mur 2 Ac(β 1 S 4) Glc. NAc(β 1 Very large Structural: in bacteria, gives rigidity and strength to cell envelope Agarose Hetero- 3)D-Gal (β 1 S 4)3, 6 - anhydro. L-Gal(α 1 1, 000 Structural: in algae, cell wall material Hyaluronan (a glycosaminoglycan) Hetero-; acidic 4)Glc. A (β 1 S 3) Glc. NAc(β 1 Up to 100, 000 Structural: in vertebrates, extracellular matrix of skin and connective tissue; viscosity and lubrication in joints a. Each polymer is classified as a homopolysaccharide (homo-) or heteropolysaccharide (hetero-). abbreviated names for the peptidoglycan, agarose, and hyaluronan repeating units indicate that the polymer contains repeats of this disaccharide unit. For example, in peptidoglycan, the Glc. NAc of one disaccharide unit is (β 1 S 4)-linked to the first residue of the next disaccharide unit. b. The

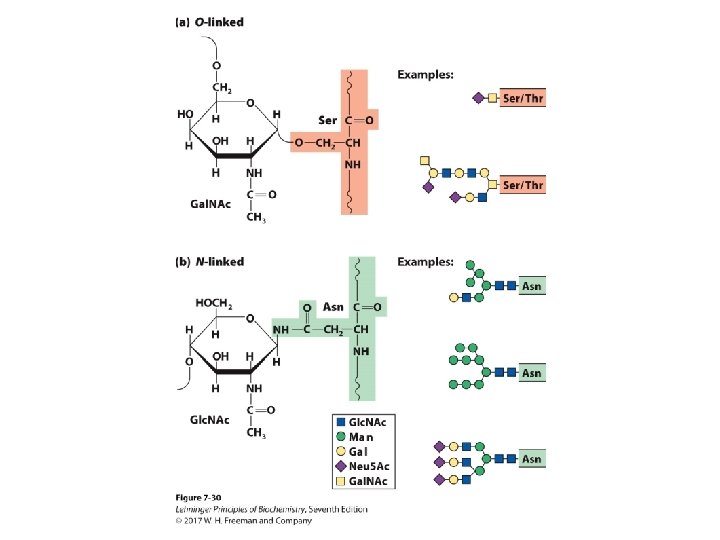
Glycoconjugates: Glycoprotein • A protein with small oligosaccharides attached – Carbohydrate ia attached via its anomeric carbon to amino acids on the protein. • Common connections occur at Ser, Thr, and Asn. – About half of mammalian proteins are glycoproteins. – Only some bacteria glycosylate a few of their proteins. – Carbohydrates play role in protein-protein recognition. – Viral proteins are heavily glycosylated; this helps evade the immune system.

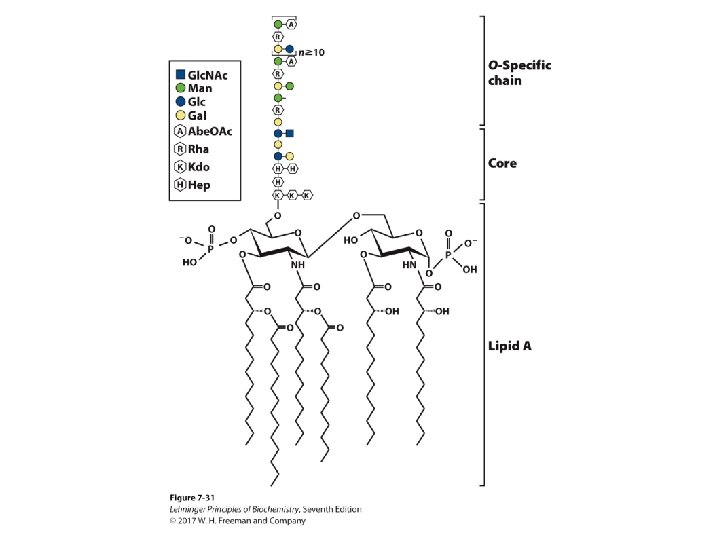
Glycoconjugates: Glycolipids • Lipids with covalently bound oligosaccharide – They are parts of plant and animal cell membranes. – In vertebrates, ganglioside carbohydrate composition determines blood groups. – In gram-negative bacteria, lipopolysaccharides cover the peptidoglycan layer.

Glycoconjugates: Proteoglycans • Sulfated glucoseaminoglycans attached to a large rodshaped protein in cell membrane – syndecans: protein has a single transmembrane domain – glypicans: protein is anchored to a lipid membrane – interact with a variety of receptors from neighboring cells and regulate cell growth
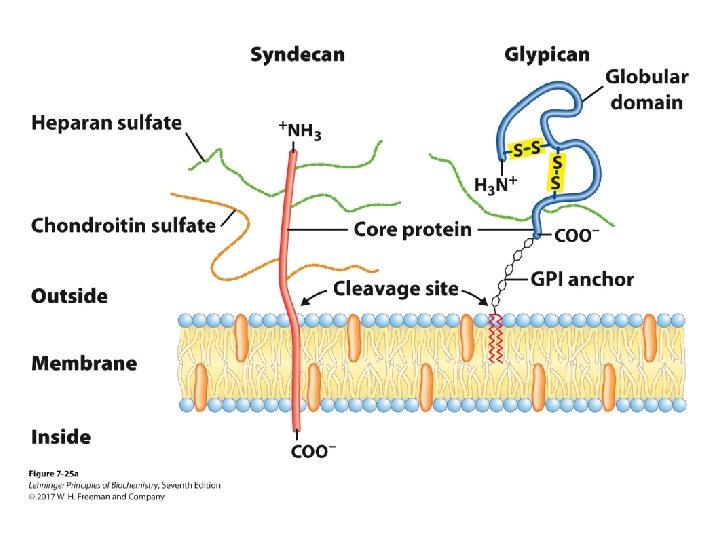
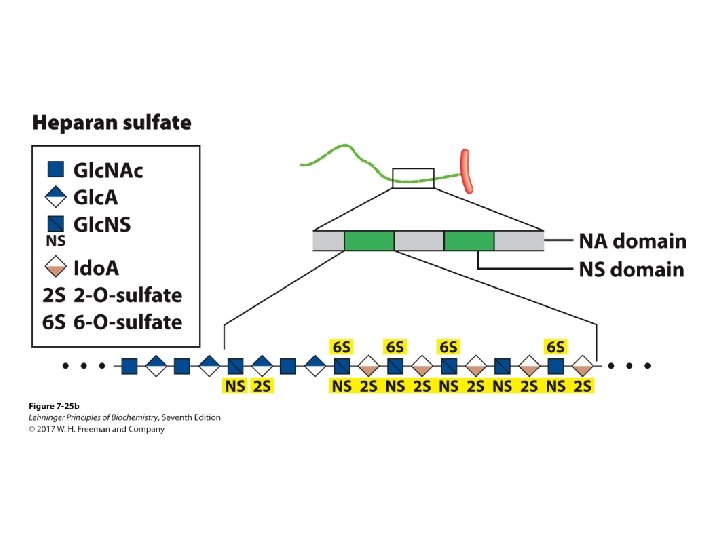

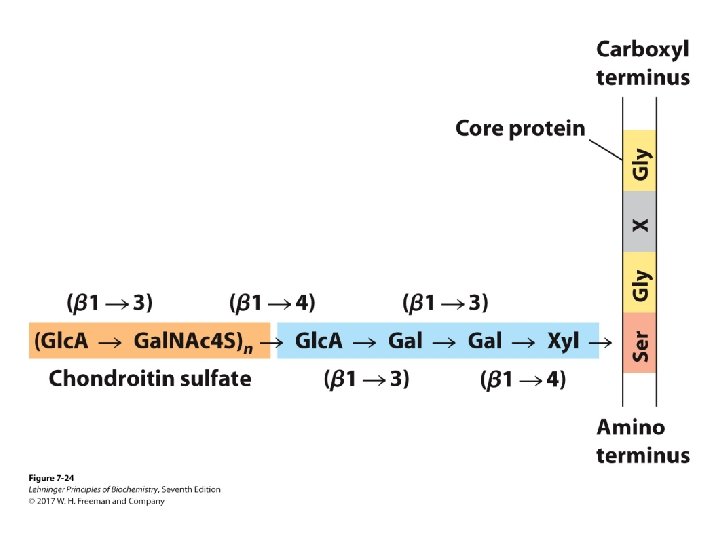
Proteoglycans • Different glycosaminoglycans are linked to the core protein. • Linkage from anomeric carbon of xylose to serine hydroxyl • Our tissues have many different core proteins; aggrecan is the best studied.

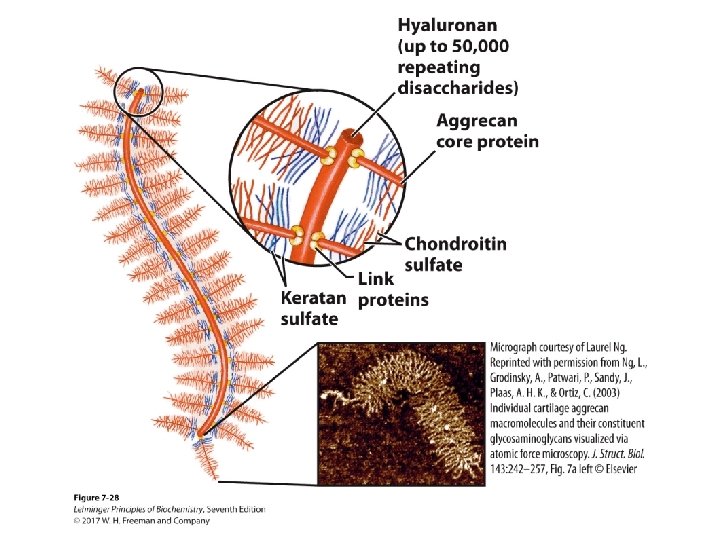
Proteoglycan Aggregates • Hyaluronan and aggrecan form huge (Mr > 2 • 108) noncovalent aggregates. • They hold a lot of water (1000 its weight) and provide lubrication. • Very low friction material • Covers joint surfaces: articular cartilage – reduced friction – load balancing

Extracellular Matrix (ECM) • Material outside the cell • Strength, elasticity, and physical barrier in tissues • Main components – proteoglycan aggregates – collagen fibers – elastin (a fibrous protein) • ECM is a barrier for tumor cells seeking to invade new tissues. – Some tumor cells secrete heparinase that degrades ECM.

Interaction of the Cells with ECM • Some integral membrane proteins are proteoglycans. – syndecans • Other integral membrane proteins are receptors for extracellular proteoglycans. – integrins • These proteins link cellular cytoskeleton to the ECM and transmit signals into the cell to regulate: – – cell growth cell mobility apoptosis wound healing

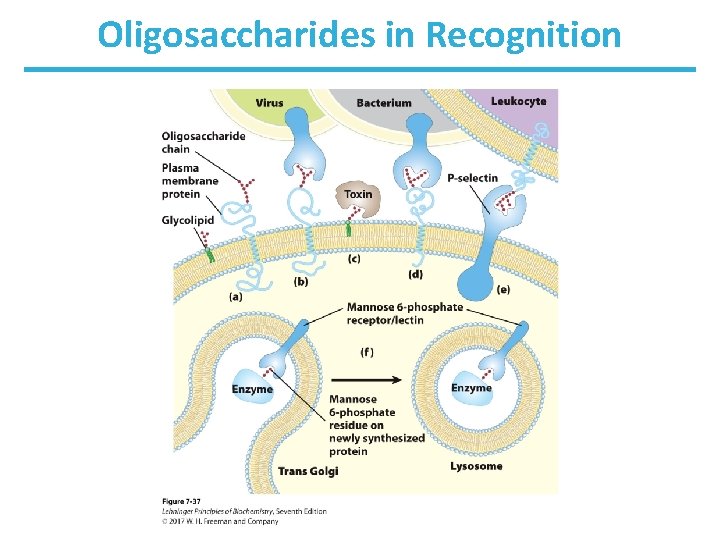
Oligosaccharides in Recognition
Glycoconjugates: Analysis

Chapter 7: Summary In this chapter, we learned about: • • structures of some important monosaccharides structures and properties of disaccharides structures and biological roles of polysaccharides functions of glycosylaminoglycans as structural components of the extracellular matrix • functions glycoconjugates in regulating a variety of biological functions
- Slides: 79