7 Beers Law and Its Implications for Instrument

7. Beer’s Law and It’s Implications for Instrument Construction

1. Derive Beer’s Law ASSUMPTIONS 1. No light is emitted 2. dx infinitesimal 3. Monochromatic light uniform on the surface, S 4. dn molecules in a section volume 5. Capture cross sectional area is

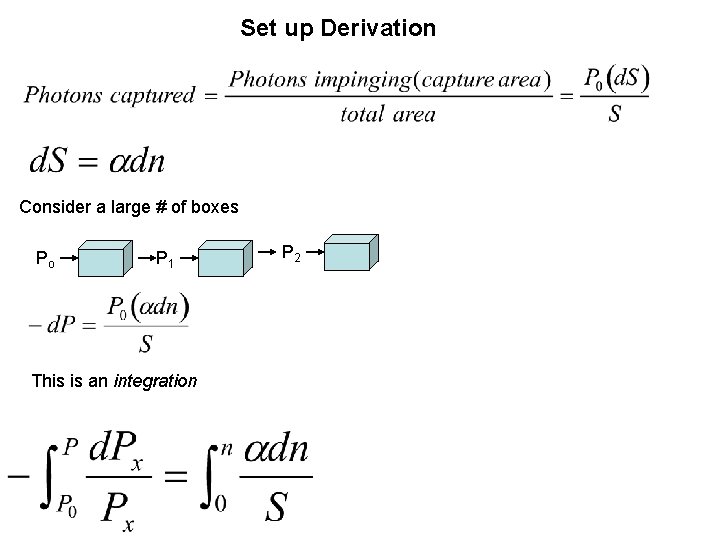
Set up Derivation Consider a large # of boxes Po P 1 This is an integration P 2

Substitutions

What is the absorbance when the light transmitted is 50% of the initial beam in a 2 cm path length cell for a concentration of 10 -3 M?

Deviations 1. Assumed each molecule was independent of the other When will the assumptions fail?

Molecules not independent when: Neighbors experience each other 1. High concentrations 2. High electrolyte 3. Large local fields due to large absorption probability (alpha)

Apparent Instrumental Deviations **polychromatic radiation*** What is the source of polychromatic radiation?

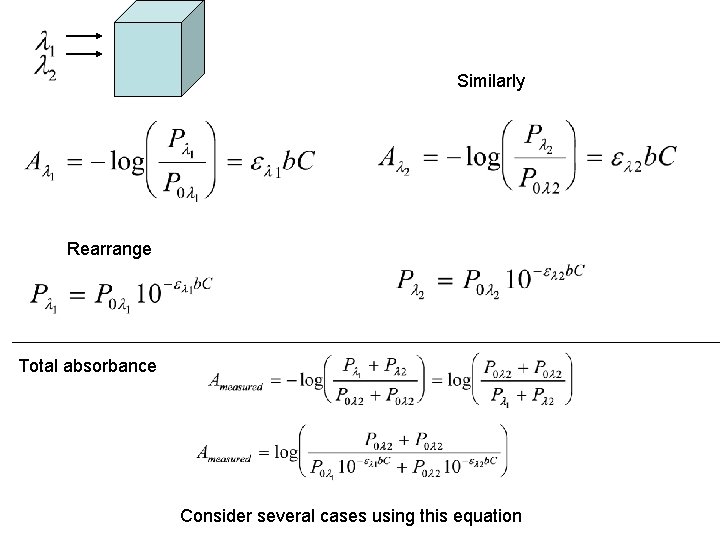
Similarly Rearrange Total absorbance Consider several cases using this equation
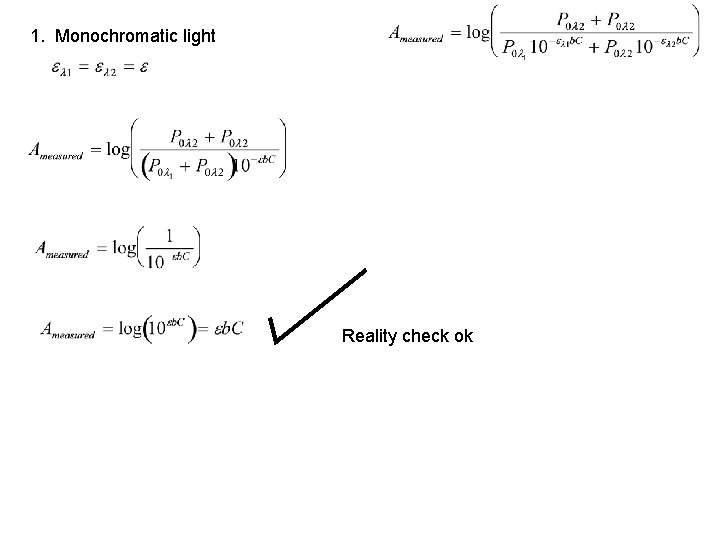
1. Monochromatic light Reality check ok
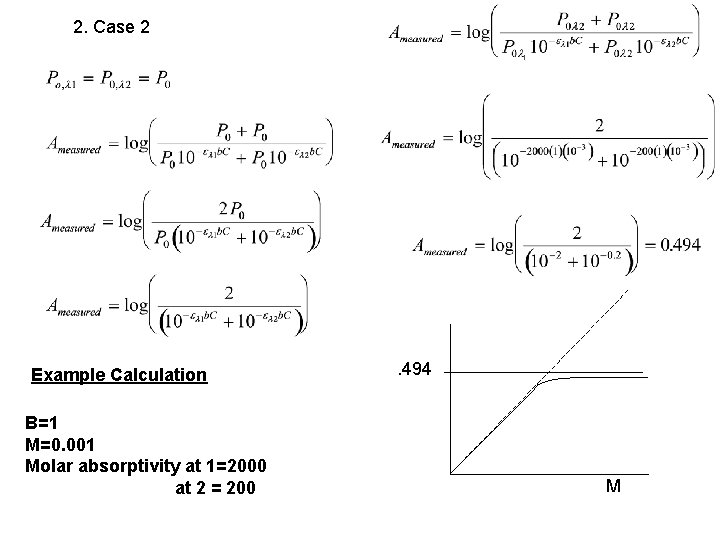
2. Case 2 Example Calculation B=1 M=0. 001 Molar absorptivity at 1=2000 at 2 = 200 . 494 M

When would this situation apply?

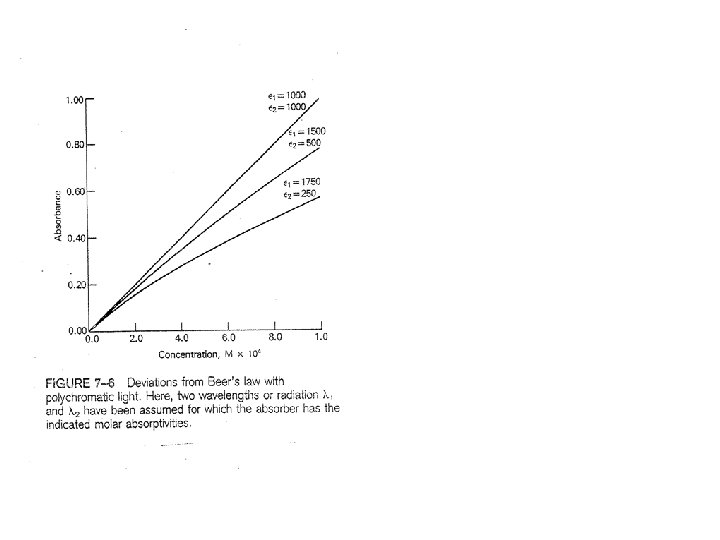

3. Stray Light Example Calculation Stray light is 0. 5% of total light What happens when light at 1 is strongly absorbed? The maximum absorbance the Instrument is capable of measuring is 2. 303
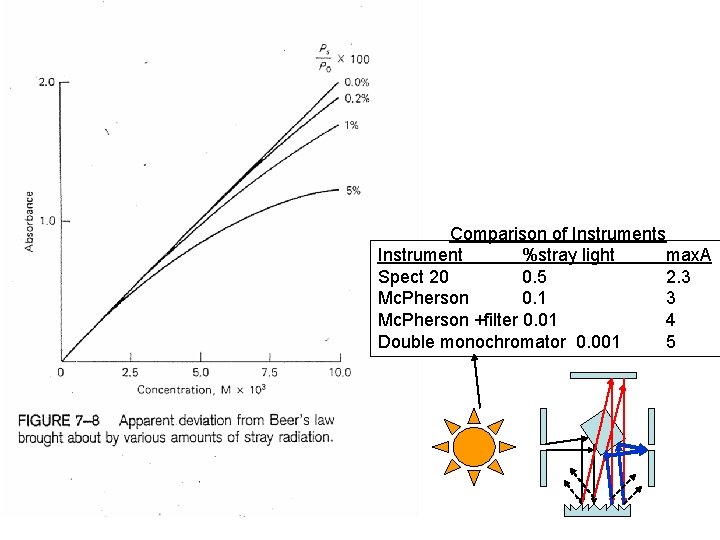
Comparison of Instruments Instrument %stray light max. A Spect 20 0. 5 2. 3 Mc. Pherson 0. 1 3 Mc. Pherson +filter 0. 01 4 Double monochromator 0. 001 5

Physical Dimensions: 89. 1 mm x 63. 3 mm x 34. 4 mm Weight: 190 grams Detector: Sony ILX 511 linear silicon CCD array Detector range: 200 -1100 nm Pixels: 2048 pixels Pixel size: 14 μm x 200 μm Pixel well depth: ~62, 500 electrons Sensitivity: 75 photons/count at 400 nm; 41 photons/count at 600 nm Czerny-Turner Focal length: 42 mm input; 68 mm output construction Entrance aperture: 5, 10, 25, 50, 100 or 200 µm wide slits or fiber (no slit) Design: f/4, Symmetrical crossed Czerny-Turner Grating options: 14 different gratings, UV through Shortwave NIR Detector collection lens option: Yes, L 2 OFLV filter options: OFLV-200 -850; OFLV-350 -1000 Other bench filter options: Longpass OF-1 filters Collimating and focusing mirrors: Standard or SAG+ UV enhanced window: Yes, UV 2 Fiber optic connector: SMA 905 to 0. 22 numerical aperture single-strand optical fiber Spectroscopic Wavelength range: Grating dependent Optical resolution: ~0. 3 -10. 0 nm FWHM Signal-to-noise ratio: 250: 1 (at full signal) A/D resolution: 12 bit Dark noise: 3. 2 RMS counts Dynamic range: 2 x 10^8 (system); 1300: 1 for a single acquisition Integration time: 3 ms to 65 seconds Stray light: <0. 05% at 600 nm; <0. 10% at 435 nm Corrected linearity: What would be Electronics Power consumption: The maximum Data transfer speed: A this could measure? >99. 8% 90 m. A @ 5 VDC Full scans to memory every 13 ms with USB 2. 0 or 1. 1 port, 300 ms with serial port

What is the maximum amount of absorbance you can measure if the stray light in an instrument is 8%? If it is 0. 05% at 600 nm as for the Ocean Optics?

1. Where does stray light come from? 2. Is stray light likely to be more important for 200 or for 900 nm light? 3. Is stray light likely to be more or less important near a region where solvent interferes?


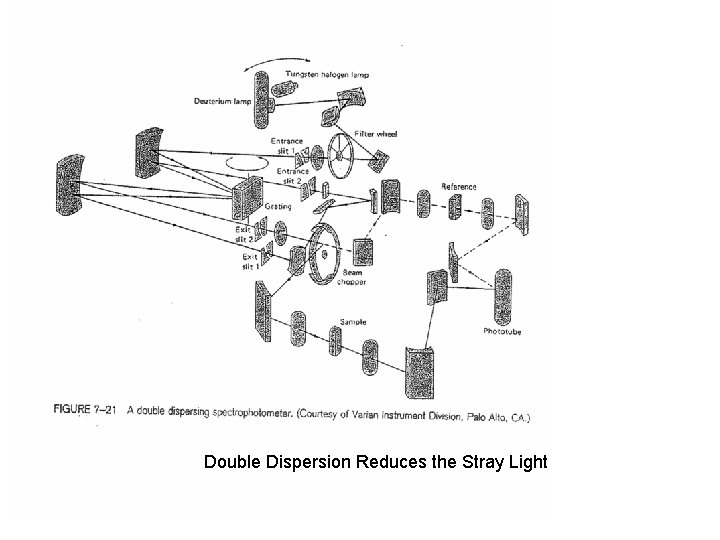
Double Dispersion Reduces the Stray Light
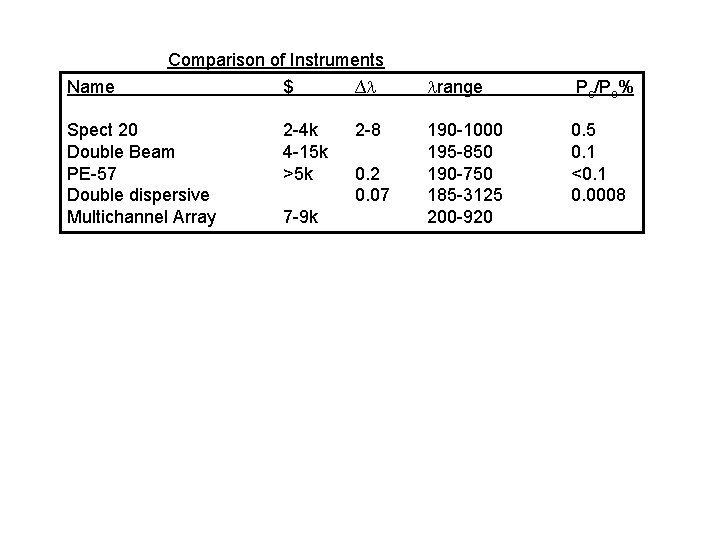
Comparison of Instruments Name $ ∆ range Ps/Po% Spect 20 Double Beam PE-57 Double dispersive Multichannel Array 2 -4 k 4 -15 k >5 k 2 -8 190 -1000 195 -850 190 -750 185 -3125 200 -920 0. 5 0. 1 <0. 1 0. 0008 7 -9 k 0. 2 0. 07

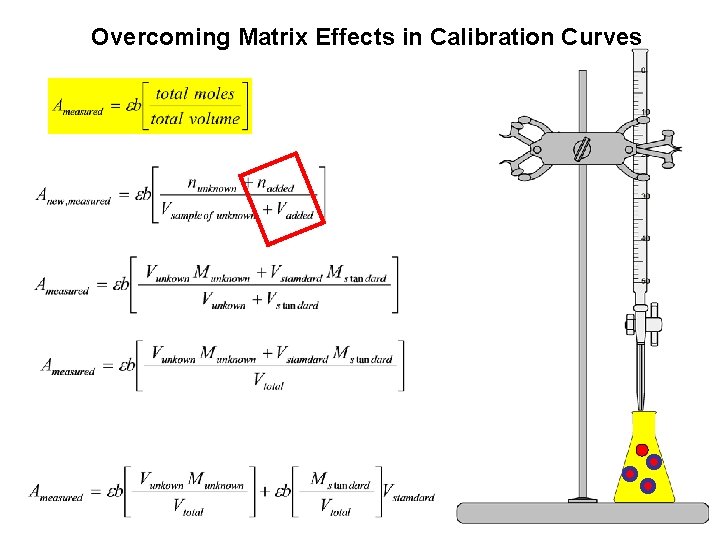
Beer’s Law and Standard Additions

QUANTITATION 1. Wide chromophore range (universality) -extended by color forming reactions for example complexation 2. Good sensitivity 3. Selectivity 4. Accuracy 5. Ease

1. Standard Curves Choose a wavelength where the molar absorptivity does not change where would this be? why choose this wavelength region? Need clear cells and no greasy fingers. Why? Need to control: temperature; p. H; electrolyte/solvent. Why? 2. Standard addition method is useful when matrix (the solution containing the sample analyte) effects complicate matters
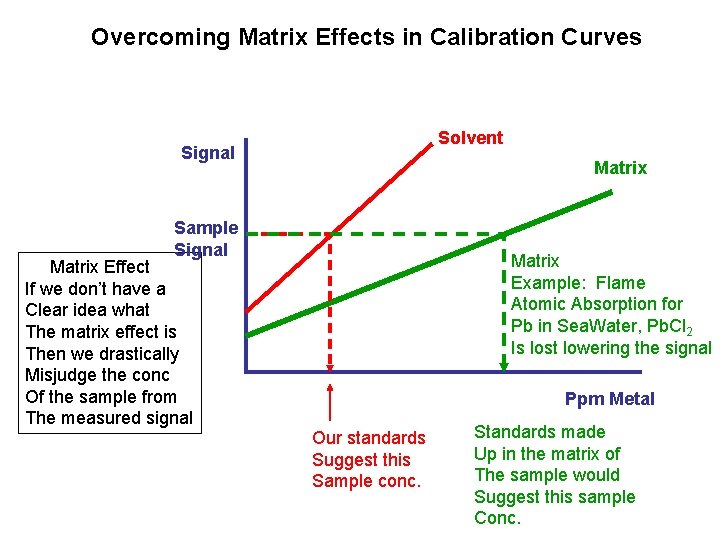
Overcoming Matrix Effects in Calibration Curves Solvent Signal Matrix Sample Signal Matrix Effect If we don’t have a Clear idea what The matrix effect is Then we drastically Misjudge the conc Of the sample from The measured signal Matrix Example: Flame Atomic Absorption for Pb in Sea. Water, Pb. Cl 2 Is lost lowering the signal Ppm Metal Our standards Suggest this Sample conc. Standards made Up in the matrix of The sample would Suggest this sample Conc.
Overcoming Matrix Effects in Calibration Curves
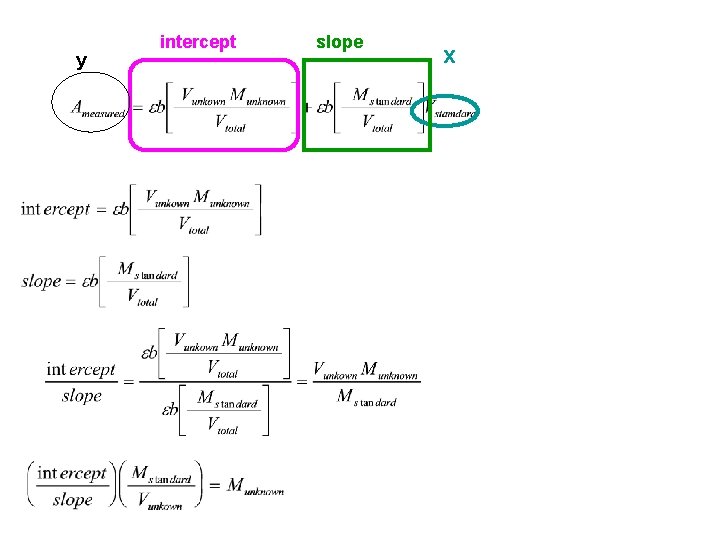
y intercept slope x

M=slope=0. 0 B=intercept= Vunknown= 10 Mstandard=11

You did standard addition for the flame lead analysis. You found: Your unknown volume is 10 m. L and the standard you added is 20 ppb. What is the unknown concentration?

Two Component Spectra measure Must be known Result is two equations in two unknowns – can be solved
- Slides: 34