7 5 Molar Mass and Calculations The molar




















- Slides: 23

7. 5 Molar Mass and Calculations The molar mass of an element is useful to convert moles of an element to grams, or grams to moles. For example, 1 mole of sodium has a mass of 22. 99 grams. Learning Goal Calculate the molar mass of a substance given its chemical formula; use molar mass to convert between grams and moles. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Molar Mass The molar mass is • the mass of 1 mole of an element. • the atomic mass expressed in grams. 1 mole C = 12. 01 g 1 mole Li = 6. 941 g Core Chemistry Skill Calculating Molar Mass General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

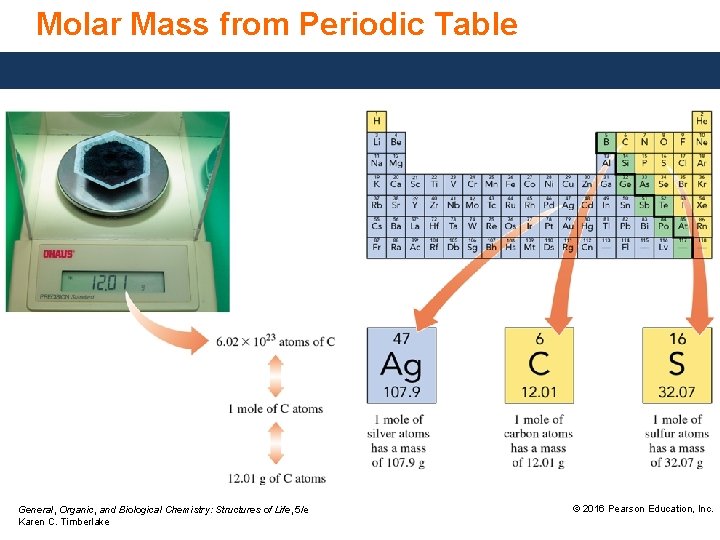
Molar Mass from Periodic Table General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Guide to Calculating Molar Mass of a Compound To calculate the molar mass of a compound, we multiply the molar mass of each element by its subscript in the formula and add the results. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Calculating Molar Mass, Li 2 CO 3 Calculate the molar mass for lithium carbonate, Li 2 CO 3, used to treat bipolar disorder. STEP 1 Obtain the molar mass of each element. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Calculating Molar Mass, Li 2 CO 3 Calculate the molar mass for lithium carbonate, Li 2 CO 3, used to treat bipolar disorder. STEP 2 Multiply each molar mass by the number of moles (subscript) in the formula. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Calculating Molar Mass, Li 2 CO 3 Calculate the molar mass for lithium carbonate, Li 2 CO 3, used to treat bipolar disorder. STEP 3 Calculate the molar mass by adding the masses of the elements. 2 moles Li = 13. 88 g Li 1 mole C = 12. 01 g C 3 moles O = + 48. 00 g O 1 mole Li 2 CO 3 = 73. 89 g General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

1 -Mole Quantities of Substances 1 -mole samples of sulfur, S (32. 07 g); iron, Fe (55. 85 g); salt, Na. Cl (58. 44 g); potassium dichromate, K 2 Cr 2 O 7 (294. 2 g); and sucrose, C 12 H 22 O 11 (342. 3 g) General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

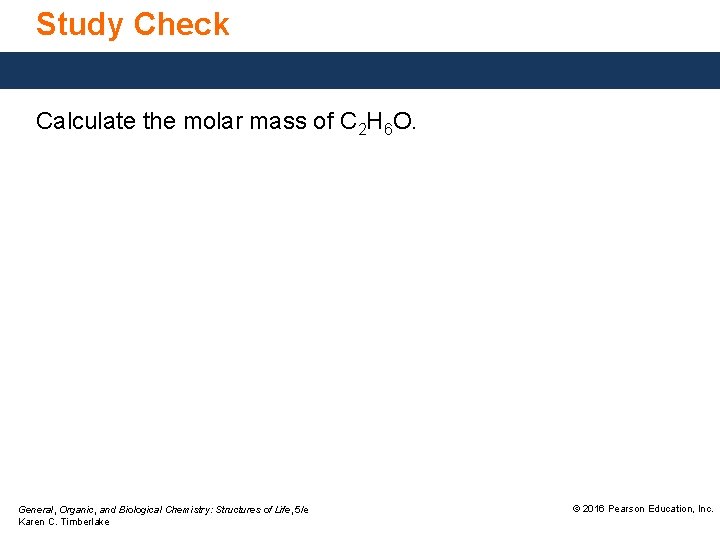
Study Check Calculate the molar mass of C 2 H 6 O. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

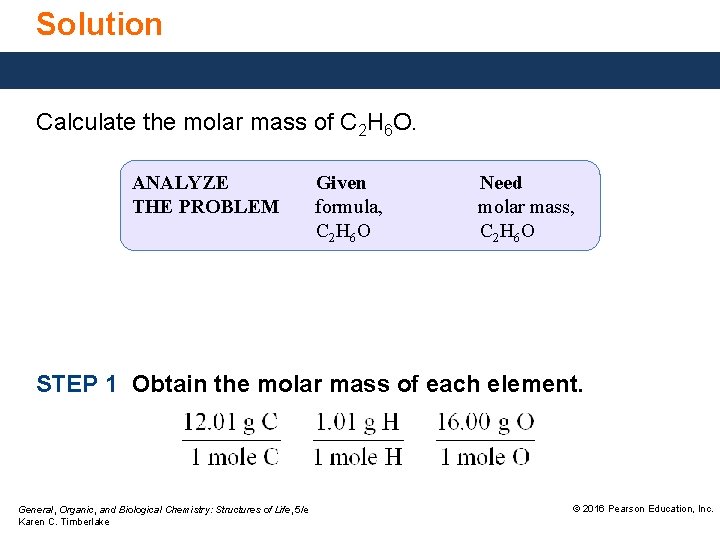
Solution Calculate the molar mass of C 2 H 6 O. ANALYZE THE PROBLEM Given formula, C 2 H 6 O Need molar mass, C 2 H 6 O STEP 1 Obtain the molar mass of each element. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Solution Calculate the molar mass of C 2 H 6 O. STEP 2 Multiply each molar mass by the number of moles (subscripts) in the formula. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Solution Calculate the molar mass of C 2 H 6 O. STEP 3 Calculate the molar mass by adding the masses of the elements. 2 moles C = 24. 02 g C 6 moles H = 6. 06 g H 1 mole O = + 16. 00 g O 1 mole C 2 H 6 O = 46. 08 g General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Calculations Using Molar Mass Molar mass conversion factors • are fractions (ratios) written from the molar mass. • relate grams and moles of an element or compound. • for methane, CH 4, used in gas stoves and gas heaters, is 1 mole of CH 4 = 16. 05 g of CH 4 (molar mass equality) Conversion factors: General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Guide to Calculating Moles from Mass or Mass from Moles General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Converting Mass to Moles of Na. Cl A box of table salt, Na. Cl, contains 737 g of Na. Cl. How many moles of Na. Cl are in the box? Table salt is sodium chloride, Na. Cl. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Converting Mass to Moles of Na. Cl A box of table salt, Na. Cl, contains 737 g of Na. Cl. How many moles of Na. Cl are in the box? STEP 1 Obtain the molar mass of each element. State the given and needed quantities. STEP 2 Write a plan to convert moles to grams or grams to moles. grams of Na. Cl Molar mass moles of Na. Cl General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Converting Mass to Moles of Na. Cl A box of table salt, Na. Cl, contains 737 g of Na. Cl. How many moles of Na. Cl are in the box? STEP 3 Determine the molar mass and write conversion factors. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Converting Mass to Moles of Na. Cl A box of table salt, Na. Cl, contains 737 g of Na. Cl. How many moles of Na. Cl are in the box? STEP 4 Set up the problem to convert grams to moles. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Study Check A sample of water has a mass of 59. 8 grams. How many moles of water are in the sample? General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Solution A sample of water has a mass of 59. 8 grams. How many moles of water are in the sample? STEP 1 State the given and needed quantities. STEP 2 Write a plan to convert grams to moles. grams of H 2 O Molar mass moles of H 2 O General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Solution A sample of water has a mass of 59. 8 grams. How many moles of water are in the sample? STEP 3 Determine the molar mass and write conversion factors. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Solution A sample of water has a mass of 59. 8 grams. How many moles of water are in the sample? STEP 4 Set up the problem to convert grams to moles. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Map: Mass–Moles–Particles General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.