7 5 AcidBase Neutralization Reactions Neutralization When acids

7. 5 Acid-Base Neutralization Reactions

Neutralization • When acids and bases are mixed together the reaction is called neutralization. The products of these reactions are often solutions of new compounds in water. Neutralization reactions are a special case of the double displacement reaction. • AB + CD AD + CB

Neutralization • As an acid is added to a base the base is gradually consumed. When all the base has reacted, the result is a neutral solution of a salt and water. A salt is a compound made up of a positive and negative ion (ionic compound)
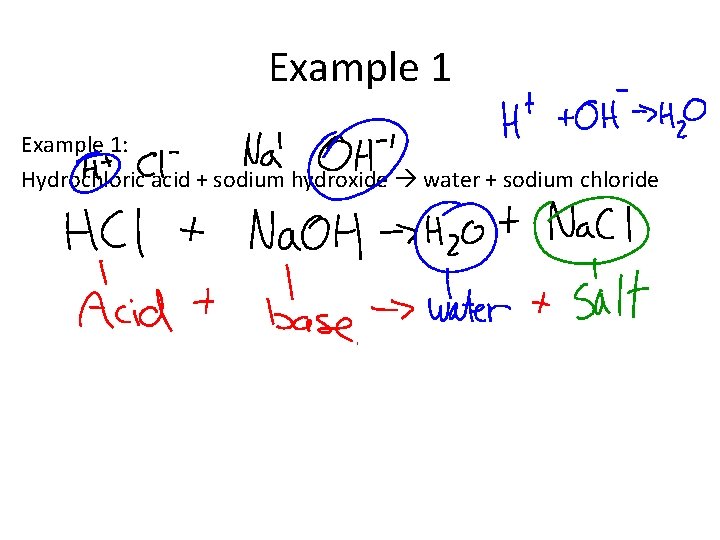
Example 1: Hydrochloric acid + sodium hydroxide water + sodium chloride

Example 1 Example 2: Sulfuric acid + potassium hydroxide water + potassium chloride

Acids + Bases • When an acid and base are combined they react in a _________ reaction. • Acids donate ______ cations • Bases donate ____ anions • ________ is this type of reaction ________.

Acids + Bases acid + base water + ionic compound For example… HCl(aq) + KOH(aq) H 2 O(l) + KCl(aq)

Why is this important? • Treating chemical spills – 150’ 000 L of sulfuric acid in Englehart, Ontario. 2007. – Treated with calcium oxide

Why is this important? • Medicine – Stomach antacids

Acids and metals • When an acid reacts with a metal, it produces a compound with the cation of the metal and the anion of the acid, and hydrogen gas. • Ex. Hydrochloric acid + zinc hydrogen gas + zinc chloride



Acids + Bases • When an acid and base are combined they react in a _________ reaction. • Acids donate ______ cations • Bases donate ____ anions • ________ is this type of reaction ________.

Acids + Bases acid + base water + ionic compound For example… HCl(aq) + KOH(aq) H 2 O(l) + KCl(aq)

Why is this important? • Treating chemical spills – 150’ 000 L of sulfuric acid in Englehart, Ontario. 2007. – Treated with calcium oxide

Why is this important? • Medicine – Stomach antacids

Acids and metals • When an acid reacts with a metal, it produces a compound with the cation of the metal and the anion of the acid, and hydrogen gas. • Ex. Hydrochloric acid + zinc hydrogen gas + zinc chloride



Acids + Bases acid + base water + ionic compound For example… HCl(aq) + KOH(aq) H 2 O(l) + KCl(aq)

Why is this important? • Treating chemical spills – 150’ 000 L of sulfuric acid in Englehart, Ontario. 2007. – Treated with calcium oxide

Why is this important? • Medicine – Stomach antacids

Acids and metals • When an acid reacts with a metal, it produces a compound with the cation of the metal and the anion of the acid, and hydrogen gas. • Ex. Hydrochloric acid + zinc hydrogen gas + zinc chloride
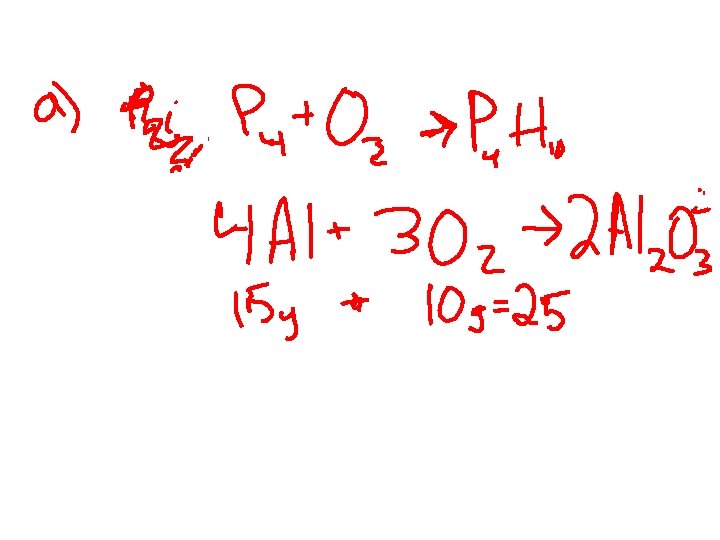

WORK RIGHT NOW • #jk GO • Page 281 – 1, 2 (test hint) 3 a) test hint 4) 5) (this one’s a brain tickler) 6, 7 (need to read text)
- Slides: 25