7 1 DESCRIBING REACTIONS SC 912 P 8

7. 1 DESCRIBING REACTIONS SC. 912. P. 8. 2, SC. 912. P. 10. 2

7. 1 LEARNING GOALS 1 Understand Law of Conservation of Mass. 2 Why must chemical equations be balanced? 3 4 What is a mole and why is it used. How to calculate the mass of a reactant or product in a chemical reaction.

CHEMICAL EQUATIONS • When a substance undergoes a chemical change a chemical reaction is taking place. • During a chemical reaction, the reactants change into products. Reactants Products • Reactants are substances that undergo change. • Products are the new substances formed as a result of that

CHEMICAL EQUATIONS • A chemical equation is a representation of a chemical reaction in which the reactants and products are expressed as formulas. Carbon + Oxygen Carbon dioxide • Written in a chemical formula: C + O (Reactants) CO (Products)

CHEMICAL EQUATIONS – CONSERVATION OF MASS • During chemical reactions, the mass of the products is always equal to the mass of the reactants. This principle is the law of conservation of mass. • Law of Conservation of Mass – mass is neither created nor destroyed in a chemical reaction. • In order to show that mass is conserved during a reaction, a chemical equation must be balanced.
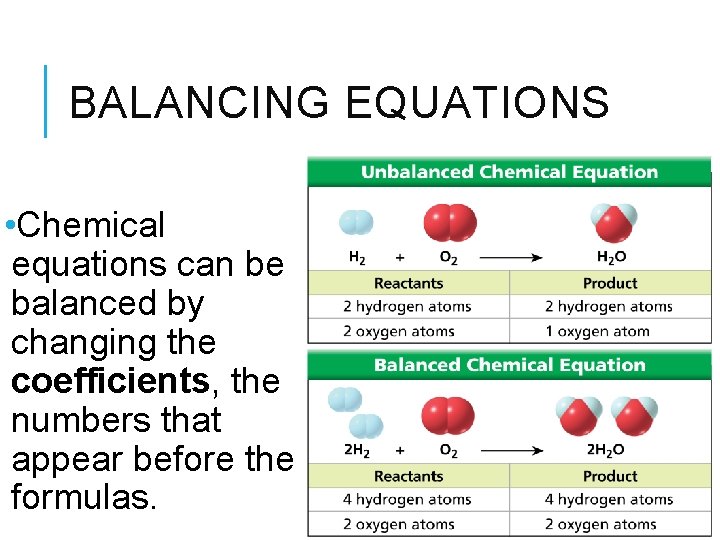
BALANCING EQUATIONS • Chemical equations can be balanced by changing the coefficients, the numbers that appear before the formulas.

BALANCING EQUATIONS PRACTICE • Write a balanced equation for the following reactions: 1. Copper + Oxygen Copper (II) Oxide (Cu. O) • Balance the following equations: 1. H 2 O 2 H 2 O + O 2 2 H 2 O 2 2 H 2 O + O 2 2 Cu + O 2 2 Cu. O 2. Hydrogen + Chloride Hydrogen Chloride (HCl) H 2 + Cl 2 2 HCl 2. Mg + HCl H 2 + Mg. Cl 2 Mg + 2 HCl H 2 + Mg. Cl 2

COUNTING WITH MOLES • Because chemical reactions often involve large numbers of small particles chemists, use a counting unit called the mole to measure amounts of a substance. • A mole (mol) is an amount of a substance that contains approximately 6. 02 × 1023 particles.

COUNTING WITH MOLES • The mass of one mole of a substance is called a molar mass. • For an element, the molar mass is the same as its atomic mass expressed in grams. • The molar mass of Carbon is 12 grams. • The molar mass of sulfur is 32. 1 grams.

COUNTING WITH MOLES • The molar mass of a compound can be calculated by adding up the atomic masses of its component atoms. • Example: Carbon Dioxide (CO 2) 1 carbon atom (12 amu) + 2 oxygen atoms (2 x 16 amu = 32 amu).

MOLE-MASS CONVERSIONS • Once you know the molar mass of a substance, you can convert moles of that substance into mass, or mass of that substance into moles. • The molar mass of CO 2 is 44 grams, which means that one mole of CO 2 has a mass of 44 grams.
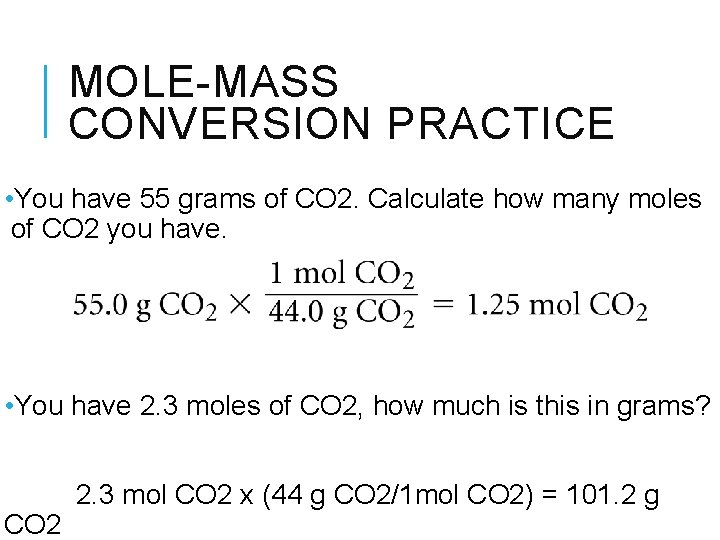
MOLE-MASS CONVERSION PRACTICE • You have 55 grams of CO 2. Calculate how many moles of CO 2 you have. • You have 2. 3 moles of CO 2, how much is this in grams? CO 2 2. 3 mol CO 2 x (44 g CO 2/1 mol CO 2) = 101. 2 g
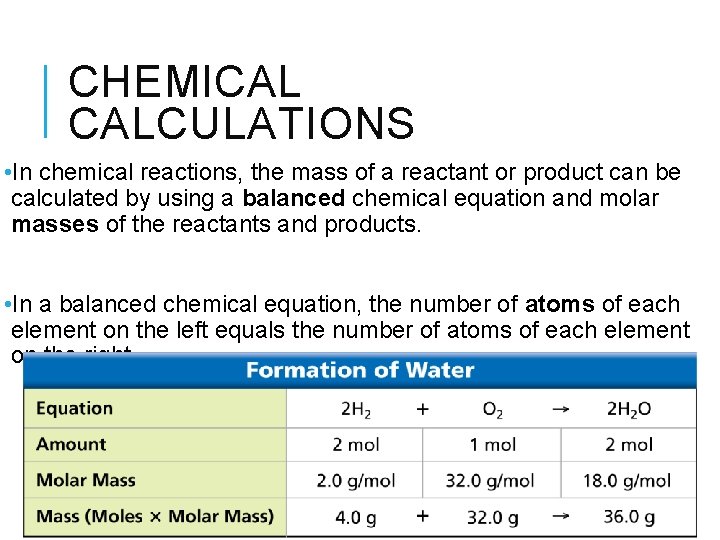
CHEMICAL CALCULATIONS • In chemical reactions, the mass of a reactant or product can be calculated by using a balanced chemical equation and molar masses of the reactants and products. • In a balanced chemical equation, the number of atoms of each element on the left equals the number of atoms of each element on the right.

CONVERTING MASS TO MOLES • Lets work on calculating how much oxygen is required to make 144 grams of water. • Begin with a balanced chemical equation for the reaction: 2 H 2 + O 2 2 H 2 O • Determine how many moles of water you are trying to make by converting the given mass of water into moles.

USING MOLE RATIOS • Write the conversion factors, or mole ratios. • Calculate how many moles of oxygen are required to produce eight moles of water.

CONVERTING MOLES TO MASS • Convert moles of O 2 to grams of O 2 by using the molar mass of O 2 as a conversion factor. • To produce 144 grams of H 2 O, you must supply 128 grams of O 2.
- Slides: 16