7 1 Chlorhexidine Digulconate for Umbilical Cord Care

7. 1% Chlorhexidine Digulconate for Umbilical Cord Care A summary of global introduction efforts, policy evidence, and product availability

What is chlorhexidine digluconate? • A broad-spectrum antiseptic • Commonly used worldwide for various applications • Excellent safety record with topical application on humans • Routinely practiced in many developed-country settings throughout the 1970 s and 1980 s without adverse health consequences

Evidence for chlorhexidine intervention
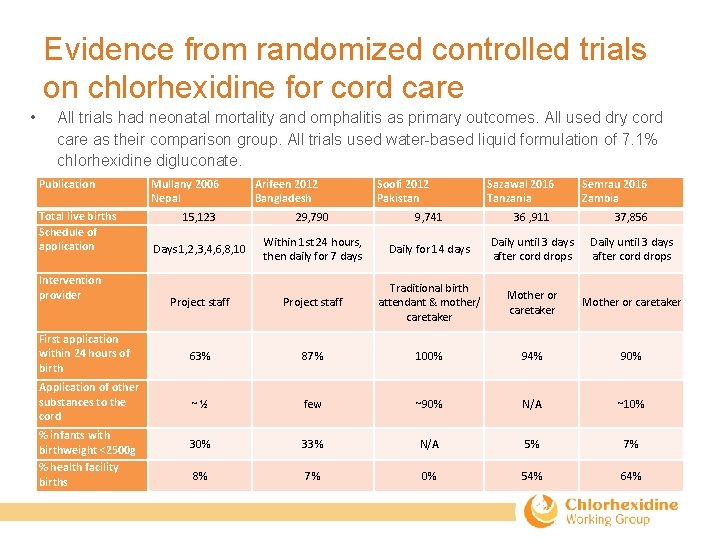
Evidence from randomized controlled trials on chlorhexidine for cord care • All trials had neonatal mortality and omphalitis as primary outcomes. All used dry cord care as their comparison group. All trials used water-based liquid formulation of 7. 1% chlorhexidine digluconate. Publication Total live births Schedule of application Mullany 2006 Nepal Arifeen 2012 Bangladesh Soofi 2012 Pakistan Sazawal 2016 Tanzania Semrau 2016 Zambia 15, 123 29, 790 9, 741 36 , 911 37, 856 Days 1, 2, 3, 4, 6, 8, 10 Within 1 st 24 hours, then daily for 7 days Daily for 14 days Daily until 3 days after cord drops Project staff Traditional birth attendant & mother/ caretaker Mother or caretaker First application within 24 hours of birth 63% 87% 100% 94% 90% Application of other substances to the cord ~½ few ~90% N/A ~10% 33% N/A 5% 7% 8% 7% 0% 54% 64% Intervention provider % infants with birthweight <2500 g % health facility births

Pooled analysis of the RCTs conducted in Bangladesh, Pakistan, and Nepal Results showed: • A 23% reduction in all-cause neonatal mortality compared with the control. • Reduced risk of cord infection by 27%– 56% depending upon severity of infection. • Cord separation time was increased by 1. 7 days over dry cord care. • Washing of umbilical cord with soap and water was not advantageous compared with dry cord care in community settings.
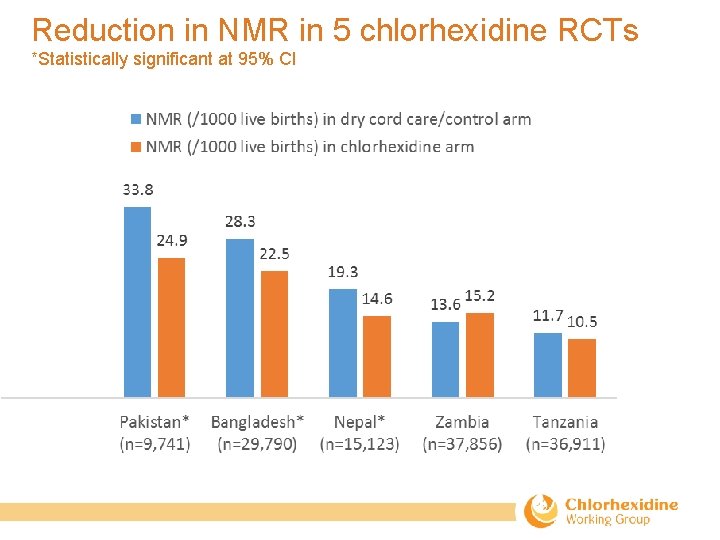
Reduction in NMR in 5 chlorhexidine RCTs *Statistically significant at 95% CI

Evidence summary regarding use of 7. 1% chlorhexidine to the umbilical cord • Use does reduce risk of omphalitis, even in low mortality settings where there is no evidence of impact on mortality risk. • Despite previously reported substantial reductions in South Asia, use did not significantly reduce NMR in study sites in Tanzania or Zambia. – This means that study results have shown an impact on mortality risk in populations with high NMR (35 -40 deaths/1, 000 live births) and have not shown an impact on mortality risk in populations with low NMR (≤ 18 deaths/1, 000 live births). • In high-mortality settings, use reduced deaths regardless of whether infants were born at home or in a facility (Hodgins 2017; Mullany, et al. 2017).

WHO recommendation on postnatal care of the mother and newborn, 2014 • RECOMMENDATION 6: Cord care daily chlorhexidine (7. 1% chlorhexidine digluconate aqueous solution or gel, delivering 4% chlorhexidine) application to the umbilical cord stump during the first week of life is recommended for newborns who are born at home in settings with high neonatal mortality (30 or more neonatal deaths per 1, 000 live births). • Clean, dry cord care is recommended for newborns born in health facilities and at home in low neonatal mortality settings. Use of chlorhexidine in these situations may be considered only to replace application of a harmful traditional substance, such as cow dung, to the cord stump.

Inclusion of chlorhexidine in WHO Model List of Essential Medicines • In 2008, USAID and Health. Tech jointly made the first application to list 7. 1% chlorhexidine digluconate for umbilical cord care. • In 2013, 7. 1% chlorhexidine digluconate was listed in the WHO Model List for Essential Medicines. • Facilitated the inclusion of chlorhexidine for umbilical cord care in national EMLs. • Eliminated confusion with chlorhexidine for other applications.

Programmatic decisions for context-appropriate CHX implementation • Settings for use – Home, facility, or both. – Distribution channels must align with intended setting of use. – ANC, private sector outlets (kiosk, pharmacy, etc. ), TBA, and CHW. • Regimen: Single-day vs. multiple-day application – Begin application as early as possible (within first 24 hours). – Consider cultural practices around cord care. • Dosage form (gel vs. liquid) – Ensure proper use of product by selecting containers that are packaged differently from other newborn medicines commonly available in the country. – Avoid forms of primary packaging that could easily be mistaken for eye or ear care products. – Conduct formative research on preference as part of larger market research if possible.
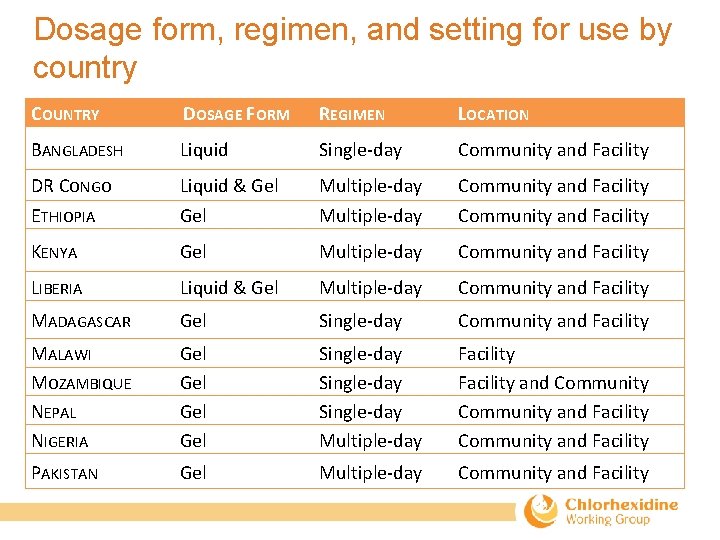
Dosage form, regimen, and setting for use by country COUNTRY DOSAGE FORM REGIMEN LOCATION BANGLADESH Liquid Single‐day Community and Facility DR CONGO ETHIOPIA Liquid & Gel Multiple‐day Community and Facility KENYA Gel Multiple‐day Community and Facility LIBERIA Liquid & Gel Multiple‐day Community and Facility MADAGASCAR Gel Single‐day Community and Facility MALAWI MOZAMBIQUE NEPAL NIGERIA Gel Gel Single‐day Multiple‐day Facility and Community and Facility PAKISTAN Gel Multiple‐day Community and Facility

Product availability and production strategy

Three-pronged approach to enable access to chlorhexidine products of high quality Facilitate product availability through inclusion of chlorhexidine in WHO Model List of Essential Medicines. Support sustainable supply of high‐ quality product through partnership with manufacturers in LMICs. Ensure approval and procurement of appropriate products through development and dissemination of standardized information.
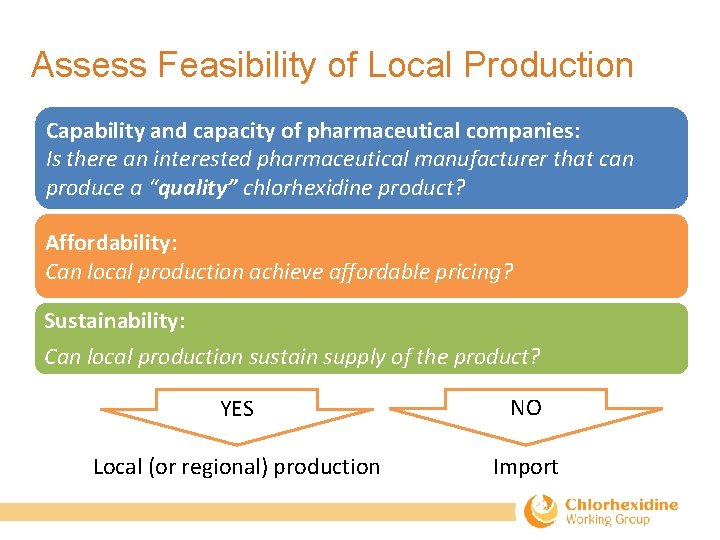
Assess Feasibility of Local Production Capability and capacity of pharmaceutical companies: Is there an interested pharmaceutical manufacturer that can produce a “quality” chlorhexidine product? Affordability: Can local production achieve affordable pricing? Sustainability: Can local production sustain supply of the product? YES NO Local (or regional) production Import
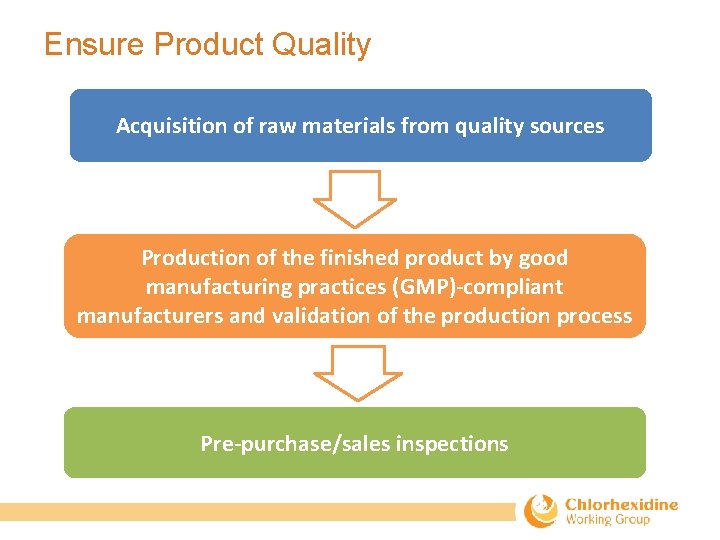
Ensure Product Quality Acquisition of raw materials from quality sources Production of the finished product by good manufacturing practices (GMP)-compliant manufacturers and validation of the production process Pre-purchase/sales inspections

Assuring product quality • Manufacturers should be certified with WHO c. GMP. Desirable to be audited by credible international agencies. • Manufacturers must register chlorhexidine for umbilical cord care in country of sales by conforming to national drug regulatory authority standards. • Make sure that the product are tested according to proper protocols before release. • Topical solution monograph included in USP‐NF. • Gel monograph to be included in the new global health section of USP‐NF. • Suppliers should be able to provide validation methods and a certificate of analysis (COA) together with the product. • Government tender documents should be clear about which dosage form they are requested (gel or liquid). • Make sure the product does not contain alcohol.
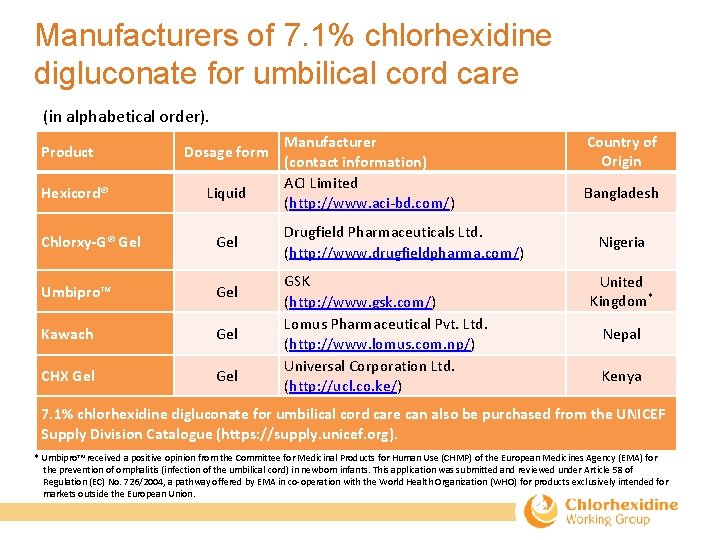
Manufacturers of 7. 1% chlorhexidine digluconate for umbilical cord care (in alphabetical order). Product Hexicord® Dosage form Liquid Chlorxy-G® Gel Umbipro™ Gel Kawach Gel CHX Gel Manufacturer (contact information) ACI Limited (http: //www. aci‐bd. com/) Drugfield Pharmaceuticals Ltd. (http: //www. drugfieldpharma. com/) GSK (http: //www. gsk. com/) Lomus Pharmaceutical Pvt. Ltd. (http: //www. lomus. com. np/) Universal Corporation Ltd. (http: //ucl. co. ke/) Country of Origin Bangladesh Nigeria United Kingdom* Nepal Kenya 7. 1% chlorhexidine digluconate for umbilical cord care can also be purchased from the UNICEF Supply Division Catalogue (https: //supply. unicef. org). * Umbipro™ received a positive opinion from the Committee for Medicinal Products for Human Use (CHMP) of the European Medicines Agency (EMA) for the prevention of omphalitis (infection of the umbilical cord) in newborn infants. This application was submitted and reviewed under Article 58 of Regulation (EC) No. 726/2004, a pathway offered by EMA in co‐operation with the World Health Organization (WHO) for products exclusively intended for markets outside the European Union.
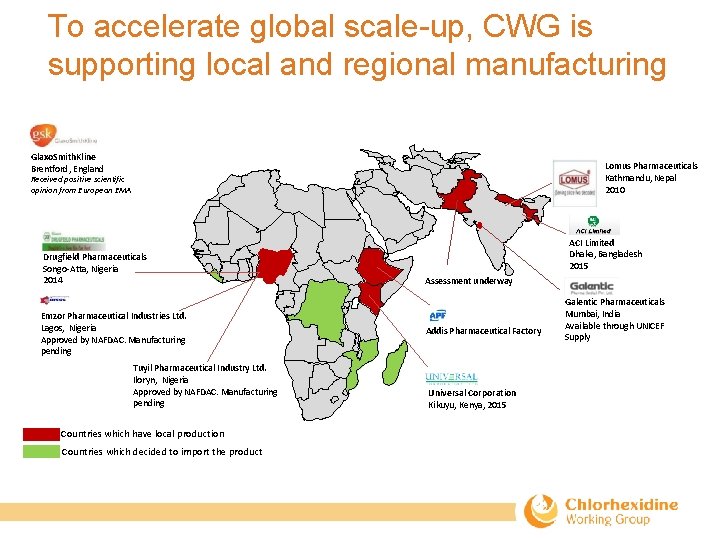
To accelerate global scale-up, CWG is supporting local and regional manufacturing Glaxo. Smith. Kline Brentford, England Lomus Pharmaceuticals Kathmandu, Nepal 2010 Received positive scientific opinion from European EMA Drugfield Pharmaceuticals Songo‐Atta, Nigeria 2014 Emzor Pharmaceutical Industries Ltd. Lagos, Nigeria Approved by NAFDAC. Manufacturing pending Tuyil Pharmaceutical Industry Ltd. Iloryn, Nigeria Approved by NAFDAC. Manufacturing pending Countries which have local production Countries which decided to import the product ACI Limited Dhaka, Bangladesh 2015 Assessment underway Addis Pharmaceutical Factory Universal Corporation Kikuyu, Kenya, 2015 Galentic Pharmaceuticals Mumbai, India Available through UNICEF Supply

Global collaboration for country introduction and scale

Chlorhexidine Working Group (CWG) An international collaboration of organizations dedicated to advancing the use of 7. 1% chlorhexidine digluconate (delivering 4% chlorhexidine) for umbilical cord care through advocacy and technical assistance. • • • • • PATH [CWG Secretariat] ayzh Bill & Melinda Gates Foundation Boston University Burnet Institute Centre for Infectious Disease Research in Zambia Clinton Health Access Initiative Drugfield Pharmaceuticals Ltd. (Nigeria) Duke University GSK (UK) Global Health Action Jhpiego John Snow, Inc. Johns Hopkins Bloomberg School of Public Health Johnson & Johnson (USA) Lomus Pharmaceuticals Pvt. Ltd. (Nepal) Maternal and Child Survival Program Ministry of Health, DRC (Reproductive Health) • • • • Ministry of Health, Ethiopia (Maternal & Child Health) Ministry of Health, Kenya (Child & Adolescent Health) Ministry of Health, Liberia (Family Health) Ministry of Health, Malawi (Reproductive Health) Ministry of Health, Mozambique (Child Health) PSI Promoting the Quality of Medicines/ United States Pharmacopeia Save the Children/Saving Newborn Lives SHOPS Plus/Abt Associates Systems for Improved Access to Pharmaceuticals and Services/Management Sciences for Health United Nations Children’s Fund United States Agency for International Development Universal Corporation Ltd. (Kenya) University of Illinois at Chicago School of Nursing University Research Co. , LLC | Center for Human Services World Health Organization

Coordinating global uptake The Chlorhexidine Working Group accelerates introduction and scale up of chlorhexidine for umbilical cord care by: Coordinating efforts for global policy development (e. g. , WHO, EML, and WHO cord care recommendation). Managing clinical, technical, and program knowledge. Ensuring rational decision making for resource allocation and priority setting. Identifying and troubleshooting issues that arise. Aligning demand with high-quality supply.
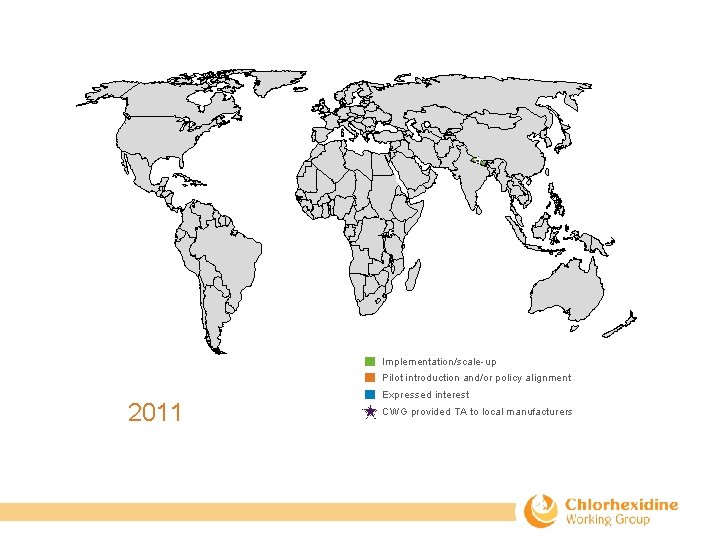
Implementation/scale-up Pilot introduction and/or policy alignment 2011 Expressed interest CWG provided TA to local manufacturers
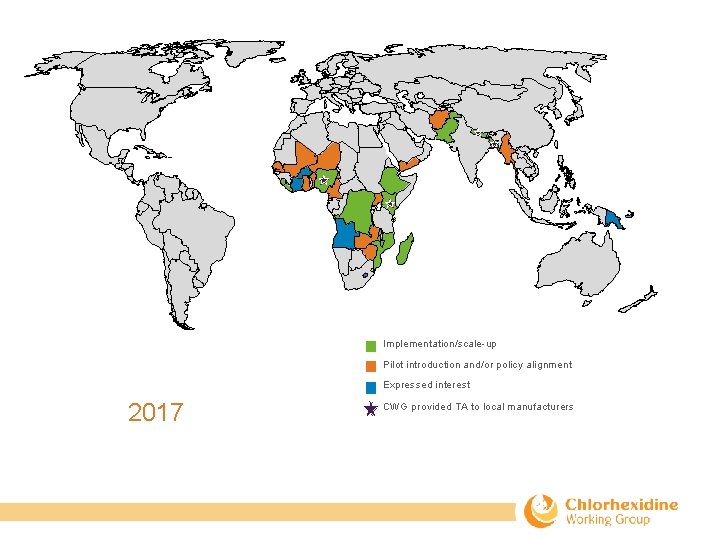
Implementation/scale-up Pilot introduction and/or policy alignment Expressed interest 2017 CWG provided TA to local manufacturers

Twelve countries have added 7. 1% CHX to national Essential Medicines List (n. EML) Country Year added Nepal 2011 DRC 2014 Ethiopia 2014 Mozambique 2014 Madagascar 2014 Myanmar 2014 Mali 2015 Kenya 2016 Nigeria 2016 Pakistan 2016 Afghanistan 2017 Ghana 2017
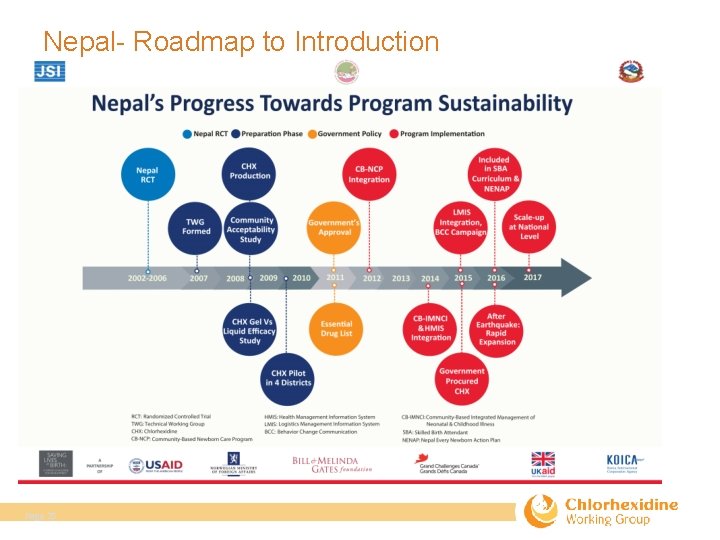
Nepal- Roadmap to Introduction Page 25
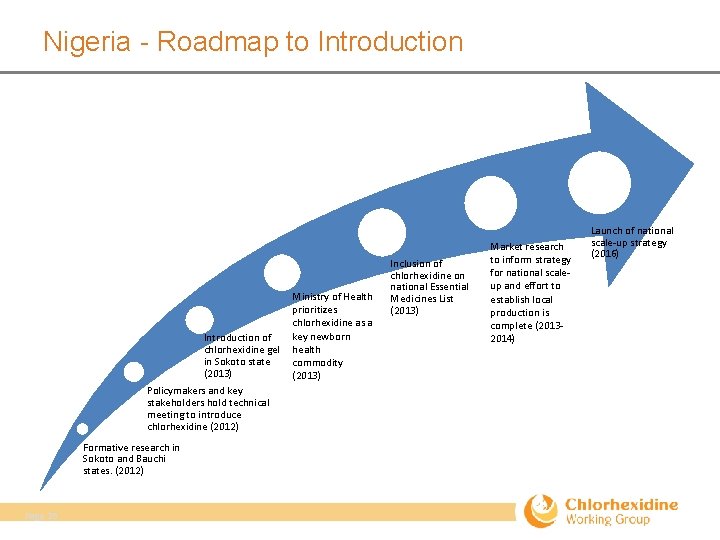
Nigeria - Roadmap to Introduction of chlorhexidine gel in Sokoto state (2013) Policymakers and key stakeholders hold technical meeting to introduce chlorhexidine (2012) Formative research in Sokoto and Bauchi states. (2012) Page 26 Ministry of Health prioritizes chlorhexidine as a key newborn health commodity (2013) Inclusion of chlorhexidine on national Essential Medicines List (2013) Market research to inform strategy for national scale‐ up and effort to establish local production is complete (2013‐ 2014) Launch of national scale‐up strategy (2016)
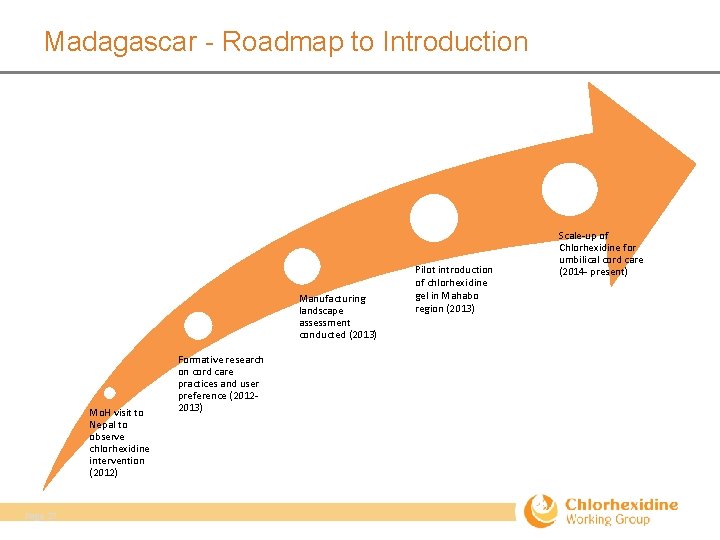
Madagascar - Roadmap to Introduction Manufacturing landscape assessment conducted (2013) Mo. H visit to Nepal to observe chlorhexidine intervention (2012) Page 27 Formative research on cord care practices and user preference (2012‐ 2013) Pilot introduction of chlorhexidine gel in Mahabo region (2013) Scale‐up of Chlorhexidine for umbilical cord care (2014‐ present)
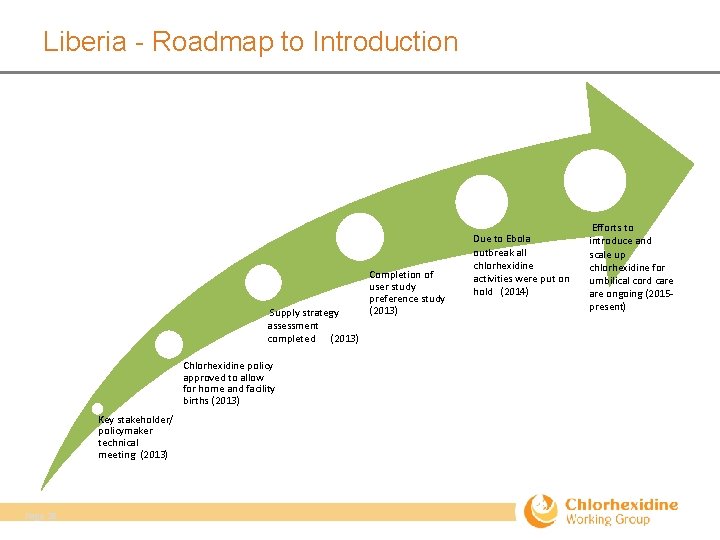
Liberia - Roadmap to Introduction Supply strategy assessment completed (2013) Chlorhexidine policy approved to allow for home and facility births (2013) Key stakeholder/ policymaker technical meeting (2013) Page 28 Completion of user study preference study (2013) Due to Ebola outbreak all chlorhexidine activities were put on hold (2014) Efforts to introduce and scale up chlorhexidine for umbilical cord care ongoing (2015‐ present)
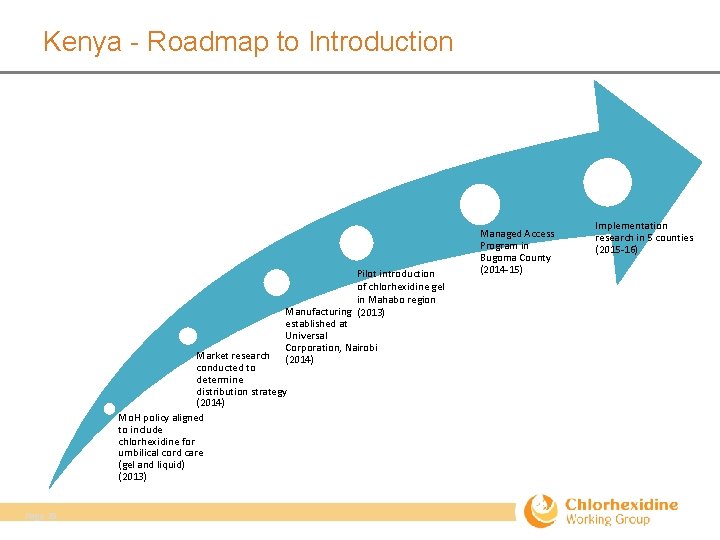
Kenya - Roadmap to Introduction Pilot introduction of chlorhexidine gel in Mahabo region Manufacturing (2013) established at Universal Corporation, Nairobi (2014) Market research conducted to determine distribution strategy (2014) Mo. H policy aligned to include chlorhexidine for umbilical cord care (gel and liquid) (2013) Page 29 Managed Access Program in Bugoma County (2014‐ 15) Implementation research in 5 counties (2015‐ 16)

Key Chlorhexidine Successes Inclusion of CHX in WHO EMLc, WHO postnatal care guidelines, Li. ST, MANDATE, and the DHS Newborn Module. CHX available from 1 global and 7 local manufacturers. 17 countries approved market registration of a CHX product (Bangladesh, Benin, Cameroon, Cote d’Ivoire, DRC, Ethiopia, Ghana, Kenya, Liberia, Madagascar, Mozambique, Nepal, Nigeria, Tanzania/Zanzibar, Uganda, Zambia) 12 country and regional stakeholder meetings (Afghanistan, DRC, Kenya, Liberia, Madagascar, Malawi, Mozambique, Nigeria, Pakistan, Uganda, and regional meeting of Burkina Faso, Côte d’Ivoire, Niger, Senegal). 12 countries added CHX to their national EML (Afghanistan, DRC, Ethiopia, Ghana, Kenya, Madagascar, Mali, Myanmar, Mozambique, Nepal, Nigeria, Pakistan) 12 countries implementing /scaling-up (Bangladesh, DRC, Ethiopia, Kenya, Liberia, Madagascar, Malawi, Mozambique, Nepal, Nigeria, Pakistan, Sierra Leone). 14 countries aligning policy/piloting the intervention (Afghanistan, Benin, Cameroon, Ghana, Haiti, Mali, Myanmar, Niger, Senegal, Timor-Leste, Uganda, Yemen, Zambia, Zimbabwe). 5 countries have expressed interest (Angola, Burkina Faso, Côte d’Ivoire, Lesotho, Papua New Guinea). 36 journal articles.

CWG resource page on Healthy Newborn Network (HNN) website http: //www. healthynewbornnetwork. org/issue/chlorhexidinefor-umbilical-cord-care/ Materials for: • Consensus building • Building evidence for implementation • Aligning policies and guidelines • Demand generation and training • Manufacturing and distribution • Monitoring and evaluation

For additional information, please visit the chlorhexidine resource page on the healthy newborn network site.
- Slides: 32