60 th International Symposium on Molecular Spectroscopy Columbus
60 th International Symposium on Molecular Spectroscopy, Columbus (OH) How methyl tops talk with each other Melanie Schnell Fritz-Haber-Institut der Max-Planck-Gesellschaft Faradayweg 4 -6, D-14195 Berlin, Germany Jens-Uwe Grabow Gottfried-Wilhelm-Leibniz-Universität Institut für Physikalische Chemie & Elektrochemie Callinstrasse 3 A, D-30167 Hannover, Germany

60 th International Symposium on Molecular Spectroscopy, Columbus (OH) Motivation - learn more about the internal rotation behavior in C 3 v-symmetric molecules with three C 3 v-symmetric tops - studied so far: (CH 3)3 Si. Cl, (CH 3)3 Ge. Cl, (CH 3)3 Sn. Cl, (CH 3)3 Si. I, (CH 3)3 Ge. CCH … - large influence of the central atom on the chemical bond character and thus on the torsional behavior: top-top communication - enlarge the series to gain more information

60 th International Symposium on Molecular Spectroscopy, Columbus (OH) … unusual K=0 quadrupole patterns - observed for (CH 3)3 Sn. Cl, but not for (CH 3)3 Si. Cl and (CH 3)3 Ge. Cl. - hypothesis: low torsional barrier and thus large splitting strong rotorsional interaction K=± 1 mixing Idea ! -probe this effect using another molecule with different quadrupole coupling and barrier to internal rotation: (CH 3)3 Ge. Br - intermediate torsional coupling - large quadrupole coupling
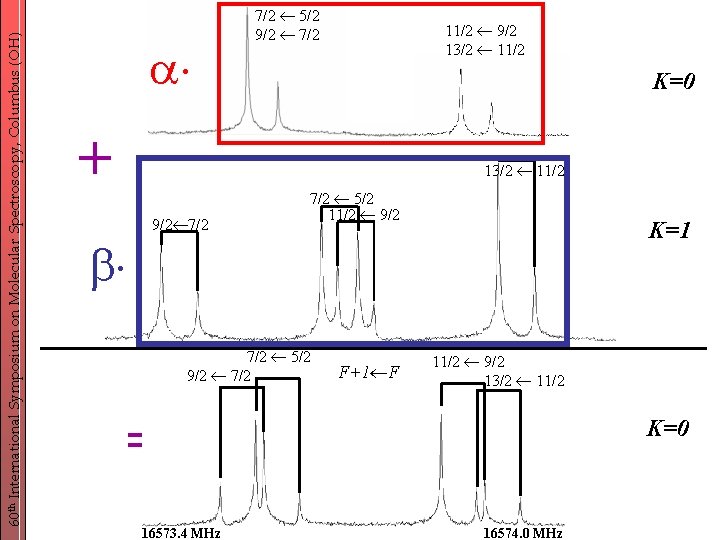
60 th International Symposium on Molecular Spectroscopy, Columbus (OH) + 7/2 5/2 9/2 7/2 11/2 9/2 13/2 11/2 K=0 13/2 11/2 9/2 7/2 5/2 11/2 9/2 7/2 5/2 9/2 7/2 F+1 F K=1 11/2 9/2 13/2 11/2 = 16573. 4 MHz K=0 16574. 0 MHz
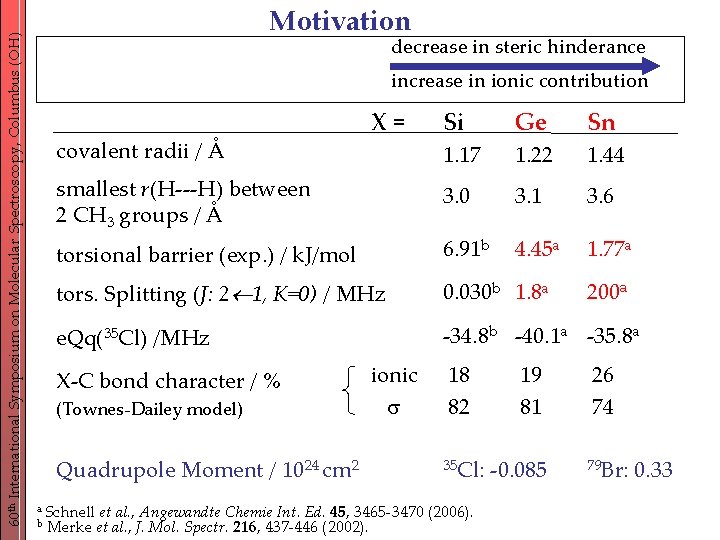
60 th International Symposium on Molecular Spectroscopy, Columbus (OH) Motivation decrease in steric hinderance A recent studya of the series (CH 3)3 XCl, X = Si, Ge, Sn revealed increase in ionic contribution the major influence of the central atom. Si Ge Sn 1. 17 1. 22 1. 44 smallest r(H---H) between 2 CH 3 groups / Å 3. 0 3. 1 3. 6 torsional barrier (exp. ) / k. J/mol 6. 91 b 4. 45 a 1. 77 a tors. Splitting (J: 2 1, K=0) / MHz 0. 030 b 1. 8 a e. Qq(35 Cl) /MHz -34. 8 b -40. 1 a -35. 8 a covalent radii / Å X-C bond character / % (Townes-Dailey model) X= ionic Quadrupole Moment / 1024 cm 2 a Schnell et al. , Angewandte Chemie Int. Ed. 45, 3465 -3470 b Merke et al. , J. Mol. Spectr. 216, 437 -446 (2002). 18 82 35 Cl: (2006). 19 81 -0. 085 200 a 26 74 79 Br: 0. 33
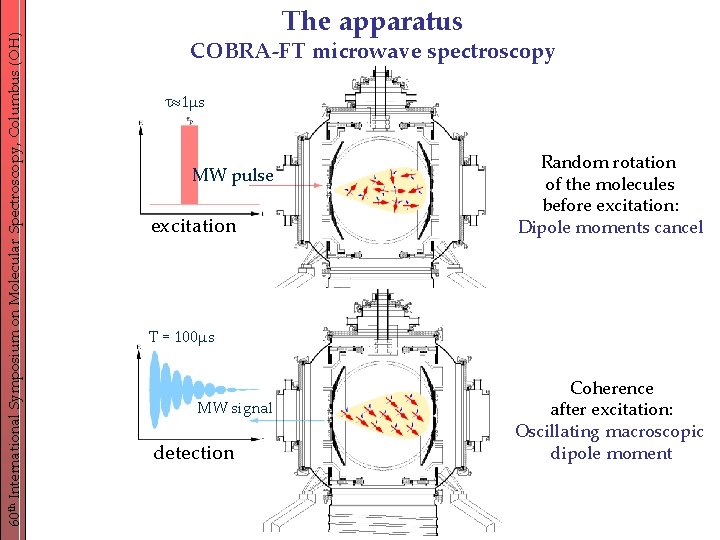
60 th International Symposium on Molecular Spectroscopy, Columbus (OH) The apparatus COBRA-FT microwave spectroscopy 1μs MW pulse excitation Random rotation of the molecules before excitation: Dipole moments cancel T = 100μs MW signal detection Coherence after excitation: Oscillating macroscopic dipole moment
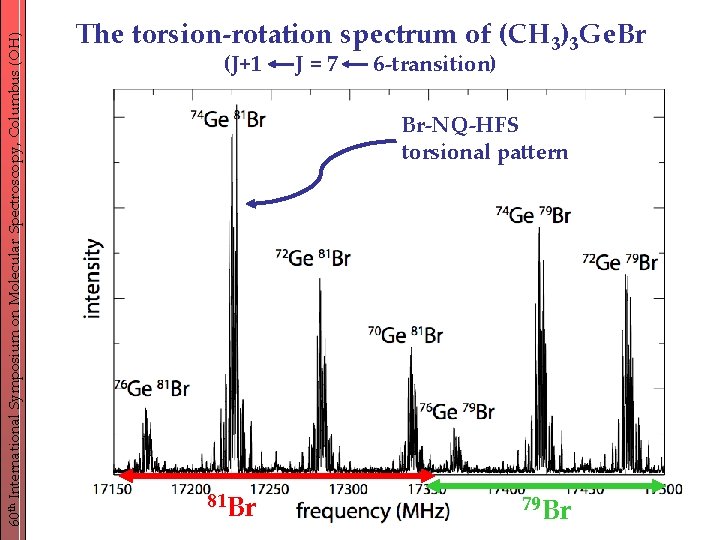
60 th International Symposium on Molecular Spectroscopy, Columbus (OH) The torsion-rotation spectrum of (CH 3)3 Ge. Br (J+1 J=7 6 -transition) Br-NQ-HFS torsional pattern 81 Br 79 Br
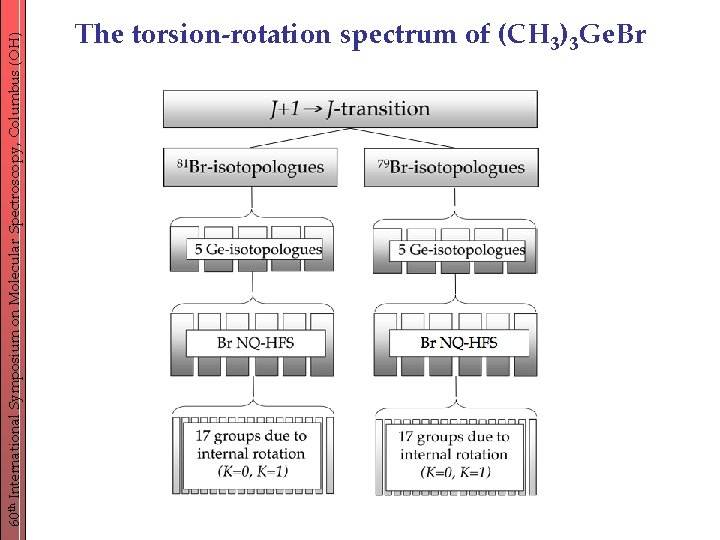
60 th International Symposium on Molecular Spectroscopy, Columbus (OH) The torsion-rotation spectrum of (CH 3)3 Ge. Br
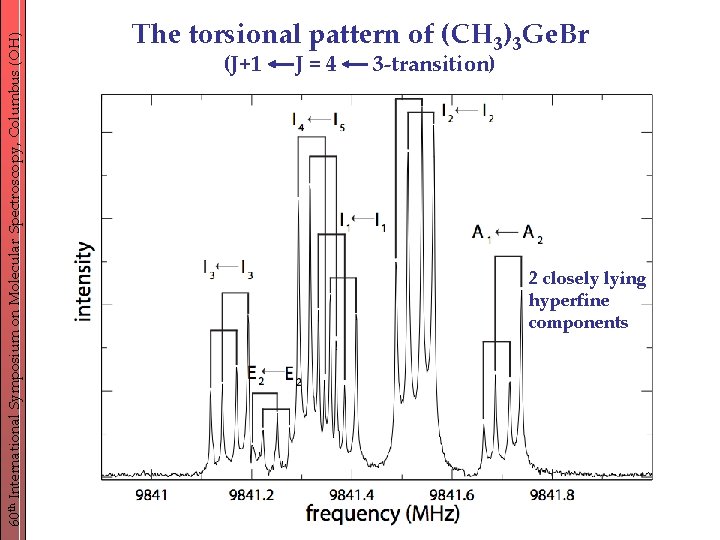
60 th International Symposium on Molecular Spectroscopy, Columbus (OH) The torsional pattern of (CH 3)3 Ge. Br (J+1 J=4 3 -transition) 2 closely lying hyperfine components

60 th International Symposium on Molecular Spectroscopy, Columbus (OH) Multidimensional high-barrier tunneling formalism (MS-group G 162) 27 different possibilities of orienting the CH 3 -groups with respect to each other (33) 27 frameworks 5 topologically inequivalent tunneling pathways: HR: rotation of 1 single CH 3 -group HA: anti-geared rotation of 2 CH 3 -groups HG: geared rotation of 2 CH 3 -groups HL: rotation of 3 CH 3 -groups with the same sense HE: rotation of 3 CH 3 -groups with different sense

60 th International Symposium on Molecular Spectroscopy, Columbus (OH) MS-group G 162 = (1, 2, 3), = (4, 5, 6), = (7, 8, 9); D = (a, b, c)(1, 4, 7)(2, 5, 8)(3, 6, 9); R = (b, c)(2, 3)(4, 7)(5, 9)(6, 8)* torsions C 3 -overall molecular rotation K. D. Möller, H. G. Andresen, J. Chem. Phys. 39, 17 (1963). Permution-Inversion reflection
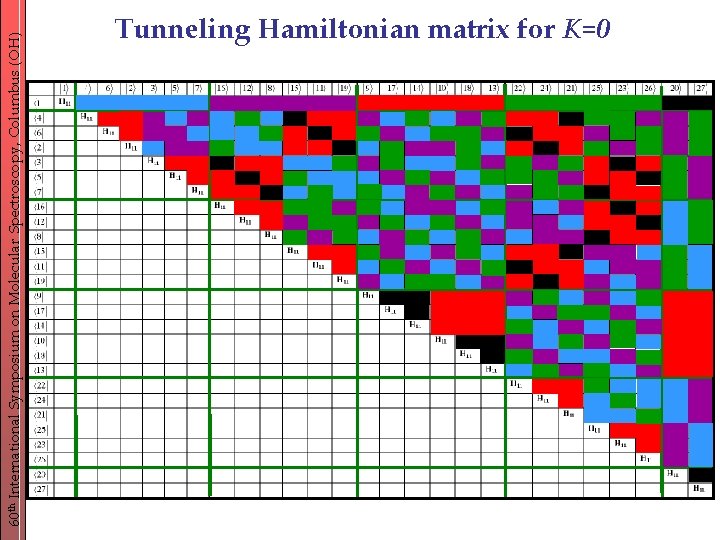
60 th International Symposium on Molecular Spectroscopy, Columbus (OH) Tunneling Hamiltonian matrix for K=0
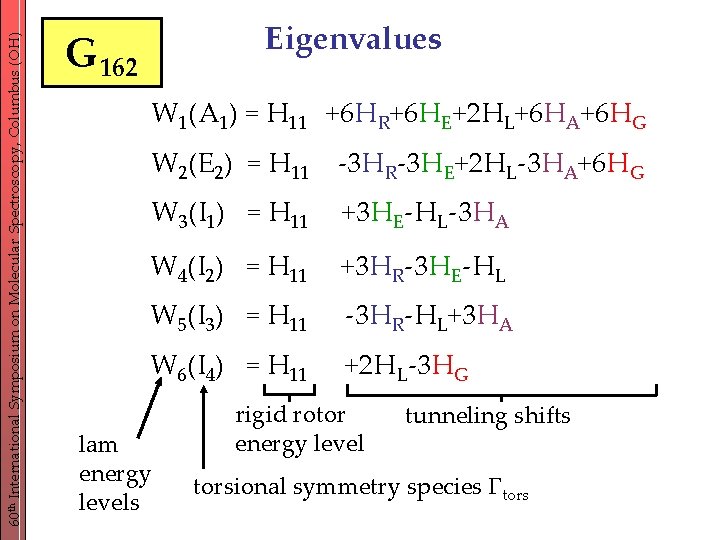
60 th International Symposium on Molecular Spectroscopy, Columbus (OH) Eigenvalues G 162 W 1(A 1) = H 11 +6 HR+6 HE+2 HL+6 HA+6 HG W 2(E 2) = H 11 -3 HR-3 HE+2 HL-3 HA+6 HG W 3(I 1) = H 11 +3 HE-HL-3 HA W 4(I 2) = H 11 +3 HR-3 HE-HL W 5(I 3) = H 11 -3 HR-HL+3 HA W 6(I 4) = H 11 +2 HL-3 HG lam energy levels rigid rotor energy level tunneling shifts torsional symmetry species tors
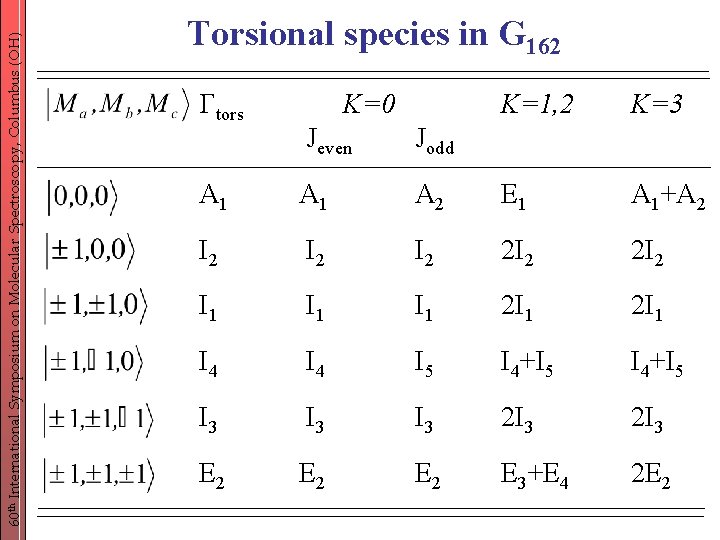
60 th International Symposium on Molecular Spectroscopy, Columbus (OH) Torsional species in G 162 tors K=0 K=1, 2 K=3 Jeven Jodd A 1 A 2 E 1 A 1+A 2 I 2 I 2 2 I 2 I 1 I 1 2 I 1 I 4 I 5 I 4+I 5 I 3 I 3 2 I 3 E 2 E 2 E 3+E 4 2 E 2
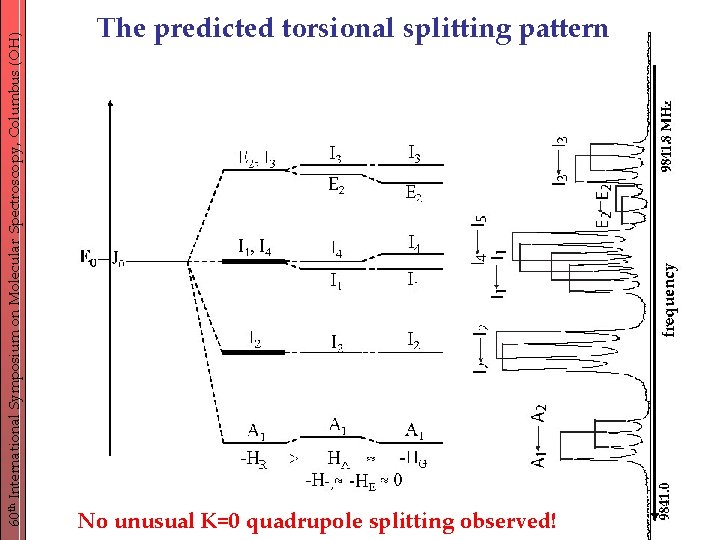
60 th International Symposium on Molecular Spectroscopy, Columbus (OH) The predicted torsional splitting pattern No unusual K=0 quadrupole splitting observed!
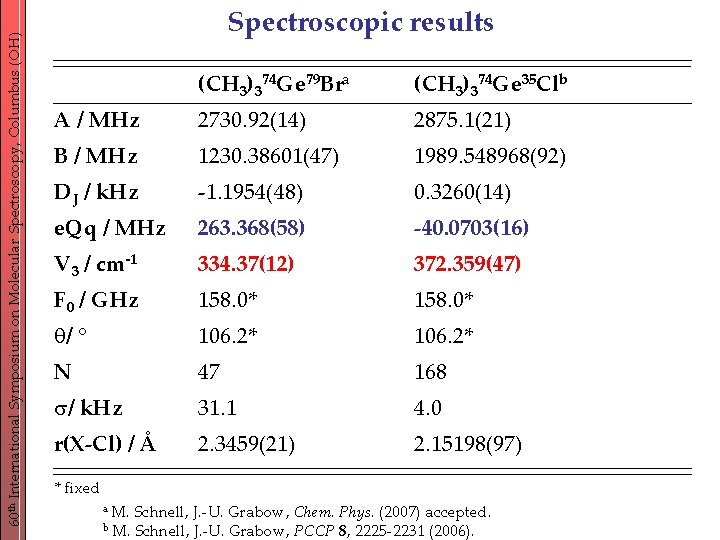
60 th International Symposium on Molecular Spectroscopy, Columbus (OH) Spectroscopic results (CH 3)374 Ge 79 Bra (CH 3)374 Ge 35 Clb A / MHz 2730. 92(14) 2875. 1(21) B / MHz 1230. 38601(47) 1989. 548968(92) DJ / k. Hz -1. 1954(48) 0. 3260(14) e. Qq / MHz 263. 368(58) -40. 0703(16) V 3 / cm-1 334. 37(12) 372. 359(47) F 0 / GHz 158. 0* q/ ° 106. 2* N 47 168 / k. Hz 31. 1 4. 0 r(X-Cl) / Å 2. 3459(21) 2. 15198(97) * fixed a M. b M. Schnell, J. -U. Grabow, Chem. Phys. (2007) accepted. Schnell, J. -U. Grabow, PCCP 8, 2225 -2231 (2006).

60 th International Symposium on Molecular Spectroscopy, Columbus (OH) Character of the chemical bonds (CH 3)374 Ge 79 Bra (CH 3)374 Ge 35 Clb covalent 62. 5 % 46. 5 % double 43. 9 % ionic -6. 4 % 15. 9 % Br towards the Ge 37. 6 % exceeds polarization covalent 76. 6 % 81 % ionic 23. 4 % 19 % Ge-X of the -bond: positive charge results on the coupling halogen Ge-C -backbonding is not possible a M. b M. backbonding of the Schnell, J. -U. Grabow, Chem. Phys. (2007) accepted. Schnell, J. -U. Grabow, PCCP 8, 2225 -2231 (2006).

60 th International Symposium on Molecular Spectroscopy, Columbus (OH) Conclusions - torsion-rotation spectrum recorded from 2. 4 -20 GHz - similar torsional splitting pattern as (CH 3)3 Ge. Cl barrier heights: 334 cm-1 (Br) vs. 372 cm-1 (Cl) Bromine compound: increased ionic character lower barrier - r(Ge-Br) = 2. 3459 Å compared to r(Ge-Cl) = 2. 15198 Å - Br NQ-hyperfine structure dominates the spectrum e. Qq: 263. 368 MHz(Br) vs. -40. 0703 MHz(Cl) - no unusual additional quadrupole splitting observed like (CH 3)3 Ge. Cl but unlike (CH 3)3 Sn. Cl

60 th International Symposium on Molecular Spectroscopy, Columbus (OH) Thanks! Wolfgang Rogge, electronic shop PCI, GWLU Hannover Mechanical shop PCI, GWLU Hannover Fonds der Chemischen Industrie Deutsche Forschungsgemeinschaft Land Niedersachsen
- Slides: 19