6 Incompressibility Water is essentially incompressible under pressure

6. Incompressibility Water is essentially incompressible under pressure In actuality, it is slightly incompressible. If truly incompressible, oceans would be 30 meter higher 7. Capable of forming hydrostatic pressure

6. Incompressibility Water is essentially incompressible under pressure In actuality, it is slightly incompressible. If truly incompressible, oceans would be 30 meter higher 7. Capable of forming hydrostatic pressure First, some background

6. Incompressibility Water is essentially incompressible under pressure In actuality, it is slightly incompressible. If truly incompressible, oceans would be 30 meter higher 7. Capable of forming hydrostatic pressure First, some background What is a force?

6. Incompressibility Water is essentially incompressible under pressure In actuality, it is slightly incompressible. If truly incompressible, oceans would be 30 meter higher 7. Capable of forming hydrostatic pressure First, some background What is a force? F = ma

6. Incompressibility Water is essentially incompressible under pressure In actuality, it is slightly incompressible. If truly incompressible, oceans would be 30 meter higher 7. Capable of forming hydrostatic pressure First, some background What is a force? F = ma Units: m (kg), a (m/s 2) = kg m/s 2 = Newton (N)

6. Incompressibility Water is essentially incompressible under pressure In actuality, it is slightly incompressible. If truly incompressible, oceans would be 30 meter higher 7. Capable of forming hydrostatic pressure First, some background What is a force? F = ma Units: m (kg), a (m/s 2) = kg m/s 2 = Newton (N) What is stress (also called pressure)?

6. Incompressibility Water is essentially incompressible under pressure In actuality, it is slightly incompressible. If truly incompressible, oceans would be 30 meter higher 7. Capable of forming hydrostatic pressure First, some background What is a force? F = ma Units: m (kg), a (m/s 2) = kg m/s 2 = Newton (N) What is stress (also called pressure)? s = F/A

6. Incompressibility Water is essentially incompressible under pressure In actuality, it is slightly incompressible. If truly incompressible, oceans would be 30 meter higher 7. Capable of forming hydrostatic pressure First, some background What is a force? F = ma Units: m (kg), a (m/s 2) = kg m/s 2 = Newton (N) What is stress (also called pressure)? s = F/A Units: F = kg m/s 2 = Newton Area = m 2

6. Incompressibility Water is essentially incompressible under pressure In actuality, it is slightly incompressible. If truly incompressible, oceans would be 30 meter higher 7. Capable of forming hydrostatic pressure First, some background What is a force? F = ma Units: m (kg), a (m/s 2) = kg m/s 2 = Newton (N) What is stress (also called pressure)? s = F/A Units: F = kg m/s 2 = Newton Area = m 2 s = N/m 2 or kg/(ms 2) = Pascal (Pa)

What is strain or deformation (e)?

What is strain or deformation (e)? Strain = DL/L Units?

What is strain or deformation (e)? Strain = DL/L Units? Dimensionless – think of it as a fraction, or multiply by 100 to get %

What is strain or deformation (e)? Strain = DL/L Units? Dimensionless – think of it as a fraction, or multiply by 100 to get % Example: 1 meter long rod subjected to tension. 1. 1 m after.

What is strain or deformation (e)? Strain = DL/L Units? Dimensionless – think of it as a fraction, or multiply by 100 to get % Example: 1 meter long rod subjected to tension. 1. 1 m after.

What is strain or deformation (e)? Strain = DL/L Units? Dimensionless – think of it as a fraction, or multiply by 100 to get % Example: 1 meter long rod subjected to tension. 1. 1 m after. e = DL/L = 0. 1 m/1 m = 0. 1 (10%)

What is strain or deformation (e)? Strain = DL/L Units? Dimensionless – think of it as a fraction, or multiply by 100 to get % Example: 1 meter long rod subjected to tension. 1. 1 m after. e = DL/L = 0. 1 m/1 m = 0. 1 (10%). What is strain rate (e )?

What is strain or deformation (e)? Strain = DL/L Units? Dimensionless – think of it as a fraction, or multiply by 100 to get % Example: 1 meter long rod subjected to tension. 1. 1 m after. e = DL/L = 0. 1 m/1 m = 0. 1 (10%). What is strain rate (e )? . e = e/t = DL/L/t Units?

What is strain or deformation (e)? Strain = DL/L Units? Dimensionless – think of it as a fraction, or multiply by 100 to get % Example: 1 meter long rod subjected to tension. 1. 1 m after. e = DL/L = 0. 1 m/1 m = 0. 1 (10%). What is strain rate (e )? . e = e/t = DL/L/t Units? s-1 (per second) Think of it as a fraction per second “It strained by 0. 1 per second”

Example: A rock stretched from 2 m to 2. 4 m in 20 seconds. What was the strain rate?
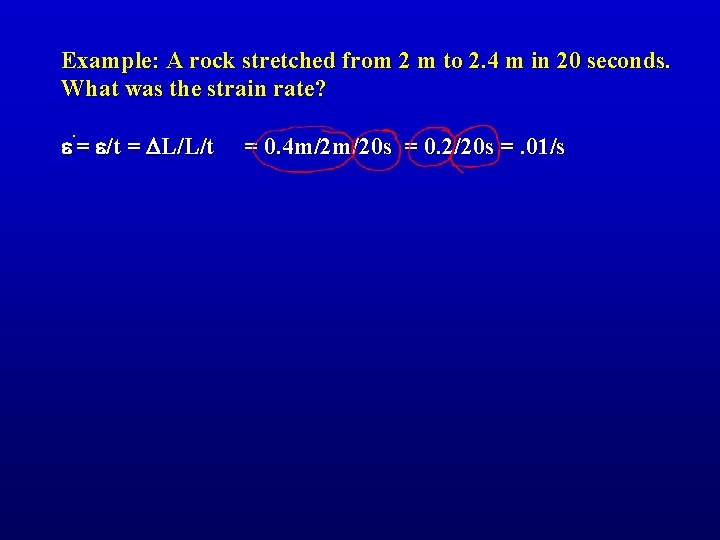
Example: A rock stretched from 2 m to 2. 4 m in 20 seconds. What was the strain rate? . e = e/t = DL/L/t = 0. 4 m/2 m/20 s = 0. 2/20 s =. 01/s

What is Hydrostatic Pressure?

What is Hydrostatic Pressure? Pressure from burial beneath a water column

What is Hydrostatic Pressure? Pressure from burial beneath a water column What is Lithostatic Pressure?

What is Hydrostatic Pressure? Pressure from burial beneath a water column What is Lithostatic Pressure? Pressure from burial beneath a rock column

What is Hydrostatic Pressure? Pressure from burial beneath a water column What is Lithostatic Pressure? Pressure from burial beneath a rock column What factors affect pressure of burial?

What is Hydrostatic Pressure? Pressure from burial beneath a water column What is Lithostatic Pressure? Pressure from burial beneath a rock column What factors affect pressure of burial? 1. Depth (m) 2. Density of overlying material (kg/m 3) 3. Gravity (m/s 2)

What is Hydrostatic Pressure? Pressure from burial beneath a water column What is Lithostatic Pressure? Pressure from burial beneath a rock column What factors affect pressure of burial? 1. Depth (m) 2. Density of overlying material (kg/m 3) 3. Gravity (m/s 2) If you multiply those three factors, what is the unit? kg/(m s 2) = Pascal = Unit of Pressure

What is Hydrostatic Pressure? Pressure from burial beneath a water column What is Lithostatic Pressure? Pressure from burial beneath a rock column What factors affect pressure of burial? 1. Depth (m) 2. Density of overlying material (kg/m 3) 3. Gravity (m/s 2) If you multiply those three factors, what is the unit? kg/(m s 2) = Pascal = Unit of Pressure sh = gdr gravity (9. 8 m/s 2), depth (m), and density (kg/m 3)

What is Hydrostatic Pressure? Pressure from burial beneath a water column What is Lithostatic Pressure? Pressure from burial beneath a rock column What factors affect pressure of burial? 1. Depth (m) 2. Density of overlying material (kg/m 3) 3. Gravity (m/s 2) If you multiply those three factors, what is the unit? kg/(m s 2) = Pascal = Unit of Pressure sh = gdr gravity (9. 8 m/s 2), depth (m), and density (kg/m 3) (1000 kg/m 3 or 1 g/cm 3 for water. 2 x for sed rock. 3 x for igneous)

Example: What is the hydrostatic pressure at the bottom of a 0. 3 km deep freshwater lake on Mars? sh = gdh g is 3. 3 m/s 2 on Mars

Example: What is the hydrostatic pressure at the bottom of a 0. 3 km deep freshwater lake on Mars? sh = gdh g is 3. 3 m/s 2 on Mars sh = 3. 3 m/s 2 x 300 m x 1000 kg/m 3 = 9. 9 x 105 Pa

Example: What is the hydrostatic pressure at the bottom of a 0. 3 km deep freshwater lake on Mars? sh = gdh g is 3. 3 m/s 2 on Mars sh = 3. 3 m/s 2 x 300 m x 1000 kg/m 3 = 9. 9 x 105 Pa How much lithostatic stress if you were buried under an equal depth of sedimentary rock on Mars (density is 2000 kg/m 3)

Example: What is the hydrostatic pressure at the bottom of a 0. 3 km deep freshwater lake on Mars? sh = gdh g is 3. 3 m/s 2 on Mars sh = 3. 3 m/s 2 x 300 m x 1000 kg/m 3 = 9. 9 x 105 Pa How much lithostatic stress if you were buried under an equal depth of sedimentary rock on Mars (density is 2000 kg/m 3) sh = 3. 3 m/s 2 x 300 m x 2000 kg/m 3 = 1. 8 x 106 Pa

8. Energy requirements to heat water and change phase A. Calorie – heat energy required to raise 1 g of H 2 O 1 degree C – specific heat of water 1 cal/g. C

8. Energy requirements to heat water and change phase A. Calorie – heat energy required to raise 1 g of H 2 O 1 degree C – specific heat of water 1 cal/g. C (1 “food” calorie is actually a kilocalorie, or 1000 calories)

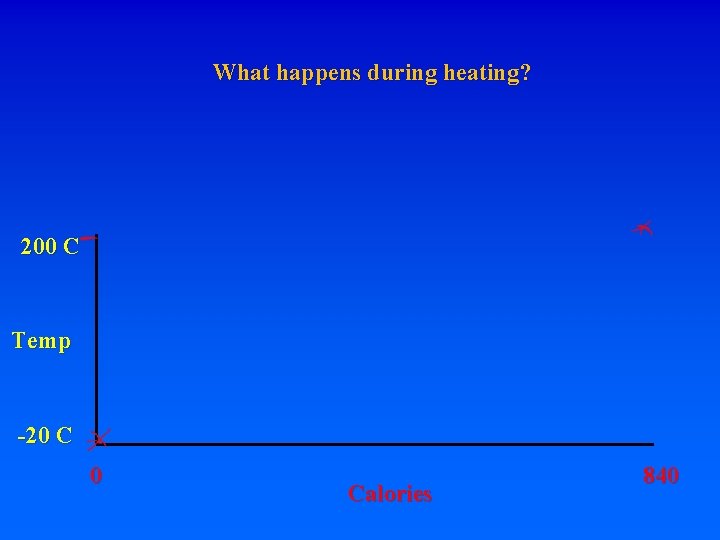
8. Energy requirements to heat water and change phase A. Calorie – heat energy required to raise 1 g of H 2 O 1 degree C – specific heat of water 1 cal/g. C (1 “food” calorie is actually a kilocalorie, or 1000 calories) Let’s start with 1 gram of ice at -20 C. How much energy is required to turn it into steam at 200 C?

8. Energy requirements to heat water and change phase A. Calorie – heat energy required to raise 1 g of H 2 O 1 degree C – specific heat of water 1 cal/g. C (1 “food” calorie is actually a kilocalorie, or 1000 calories) Let’s start with 1 gram of ice at -20 C. How much energy is required to turn it into steam at 200 C? Raise from -20 to 0 requires:

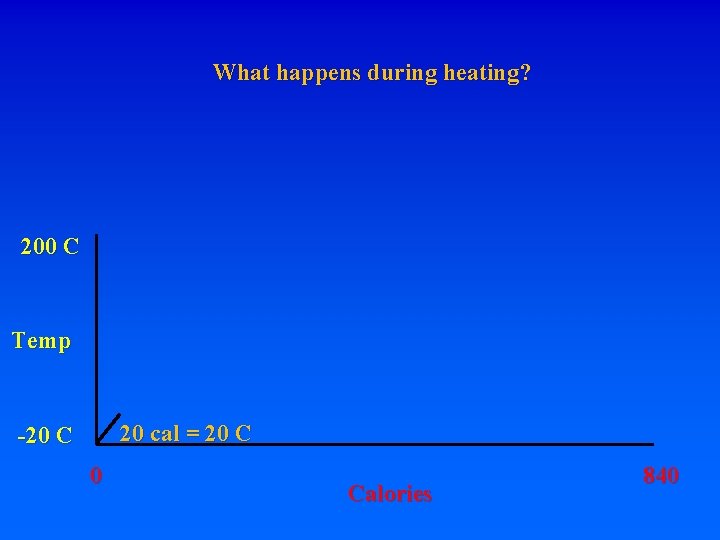
8. Energy requirements to heat water and change phase A. Calorie – heat energy required to raise 1 g of H 2 O 1 degree C – specific heat of water 1 cal/g. C (1 “food” calorie is actually a kilocalorie, or 1000 calories) Let’s start with 1 gram of ice at -20 C. How much energy is required to turn it into steam at 200 C? Raise from -20 to 0 requires: 20 calories. What do you have?

8. Energy requirements to heat water and change phase A. Calorie – heat energy required to raise 1 g of H 2 O 1 degree C – specific heat of water 1 cal/g. C (1 “food” calorie is actually a kilocalorie, or 1000 calories) Let’s start with 1 gram of ice at -20 C. How much energy is required to turn it into steam at 200 C? Raise from -20 to 0 requires: 20 calories. What do you have? How much energy to get liquid water at 0 C?

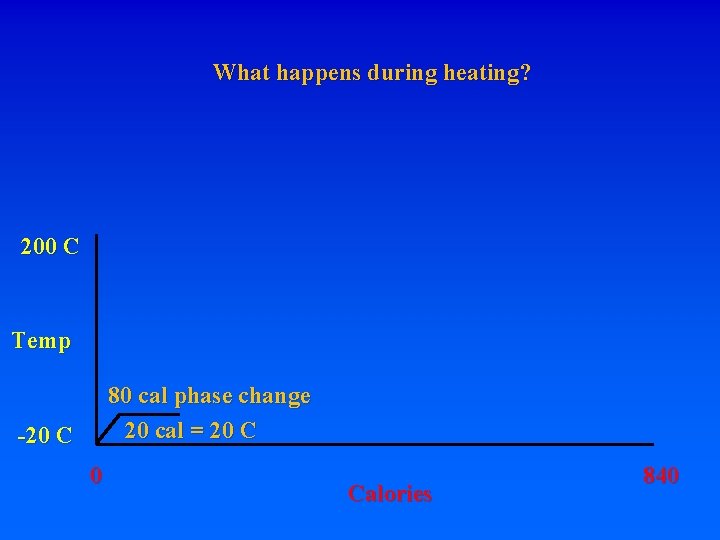
8. Energy requirements to heat water and change phase A. Calorie – heat energy required to raise 1 g of H 2 O 1 degree C – specific heat of water 1 cal/g. C (1 “food” calorie is actually a kilocalorie, or 1000 calories) Let’s start with 1 gram of ice at -20 C. How much energy is required to turn it into steam at 200 C? Raise from -20 to 0 requires: 20 calories. What do you have? How much energy to get liquid water at 0 C? MUST BREAK BONDS! Latent heat of fusion – 80 cal/gr for H 20

8. Energy requirements to heat water and change phase A. Calorie – heat energy required to raise 1 g of H 2 O 1 degree C – specific heat of water 1 cal/g. C (1 “food” calorie is actually a kilocalorie, or 1000 calories) Let’s start with 1 gram of ice at -20 C. How much energy is required to turn it into steam at 200 C? Raise from -20 to 0 requires: 20 calories. What do you have? How much energy to get liquid water at 0 C? MUST BREAK BONDS! Latent heat of fusion – 80 cal/gr for H 20 To go from ice at -20 to ice at 0 requires 20 cal

8. Energy requirements to heat water and change phase A. Calorie – heat energy required to raise 1 g of H 2 O 1 degree C – specific heat of water 1 cal/g. C (1 “food” calorie is actually a kilocalorie, or 1000 calories) Let’s start with 1 gram of ice at -20 C. How much energy is required to turn it into steam at 200 C? Raise from -20 to 0 requires: 20 calories. What do you have? How much energy to get liquid water at 0 C? MUST BREAK BONDS! Latent heat of fusion – 80 cal/gr for H 20 To go from ice at -20 to ice at 0 requires 20 cal To go from ice at 0 to liquid at 0 requires 80 cal!

8. Energy requirements to heat water and change phase A. Calorie – heat energy required to raise 1 g of H 2 O 1 degree C – specific heat of water 1 cal/g. C (1 “food” calorie is actually a kilocalorie, or 1000 calories) Let’s start with 1 gram of ice at -20 C. How much energy is required to turn it into steam at 200 C? Raise from -20 to 0 requires: 20 calories. What do you have? How much energy to get liquid water at 0 C? MUST BREAK BONDS! Latent heat of fusion – 80 cal/gr for H 20 To go from ice at -20 to ice at 0 requires 20 cal To go from ice at 0 to liquid at 0 requires 80 cal! To go from liquid at 0 to liquid at 100 requires 100 cal

8. Energy requirements to heat water and change phase A. Calorie – heat energy required to raise 1 g of H 2 O 1 degree C – specific heat of water 1 cal/g. C (1 “food” calorie is actually a kilocalorie, or 1000 calories) Let’s start with 1 gram of ice at -20 C. How much energy is required to turn it into steam at 200 C? Raise from -20 to 0 requires: 20 calories. What do you have? How much energy to get liquid water at 0 C? MUST BREAK BONDS! Latent heat of fusion – 80 cal/gr for H 20 To go from ice at -20 to ice at 0 requires 20 cal To go from ice at 0 to liquid at 0 requires 80 cal! To go from liquid at 0 to liquid at 100 requires 100 cal To go from liquid at 100 to gas at 100 requires 540 cal!!! Latent heat of vaporization

8. Energy requirements to heat water and change phase A. Calorie – heat energy required to raise 1 g of H 2 O 1 degree C – specific heat of water 1 cal/g. C (1 “food” calorie is actually a kilocalorie, or 1000 calories) Let’s start with 1 gram of ice at -20 C. How much energy is required to turn it into steam at 200 C? Raise from -20 to 0 requires: 20 calories. What do you have? How much energy to get liquid water at 0 C? MUST BREAK BONDS! Latent heat of fusion – 80 cal/gr for H 20 To go from ice at -20 to ice at 0 requires 20 cal To go from ice at 0 to liquid at 0 requires 80 cal! To go from liquid at 0 to liquid at 100 requires 100 cal To go from liquid at 100 to gas at 100 requires 540 cal!!! Latent heat of vaporization Raise from -20 ice to 200 steam = 20+80+100+540+100=840 cal

8. Energy requirements to heat water and change phase A. Calorie – heat energy required to raise 1 g of H 2 O 1 degree C – specific heat of water 1 cal/g. C (1 “food” calorie is actually a kilocalorie, or 1000 calories) Let’s start with 1 gram of ice at -20 C. How much energy is required to turn it into steam at 200 C? Raise from -20 to 0 requires: 20 calories. What do you have? How much energy to get liquid water at 0 C? MUST BREAK BONDS! Latent heat of fusion – 80 cal/gr for H 20 To go from ice at -20 to ice at 0 requires 20 cal To go from ice at 0 to liquid at 0 requires 80 cal! To go from liquid at 0 to liquid at 100 requires 100 cal To go from liquid at 100 to gas at 100 requires 540 cal!!! Latent heat of vaporization Raise from -20 ice to 200 steam = 20+80+100+540+100=840 cal 620 to change phases, 220 to raise temperature
What happens during heating? 200 C Temp -20 C 0 Calories 840
What happens during heating? 200 C Temp 20 cal = 20 C -20 C 0 Calories 840
What happens during heating? 200 C Temp 80 cal phase change 20 cal = 20 C -20 C 0 Calories 840
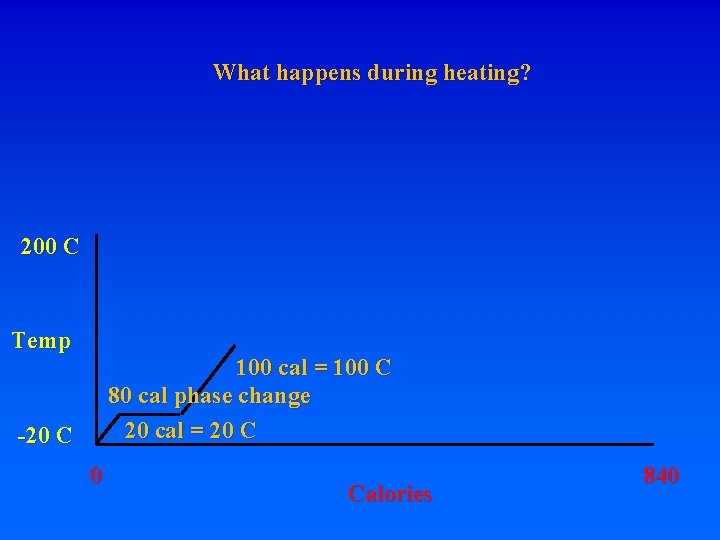
What happens during heating? 200 C Temp 100 cal = 100 C 80 cal phase change 20 cal = 20 C -20 C 0 Calories 840
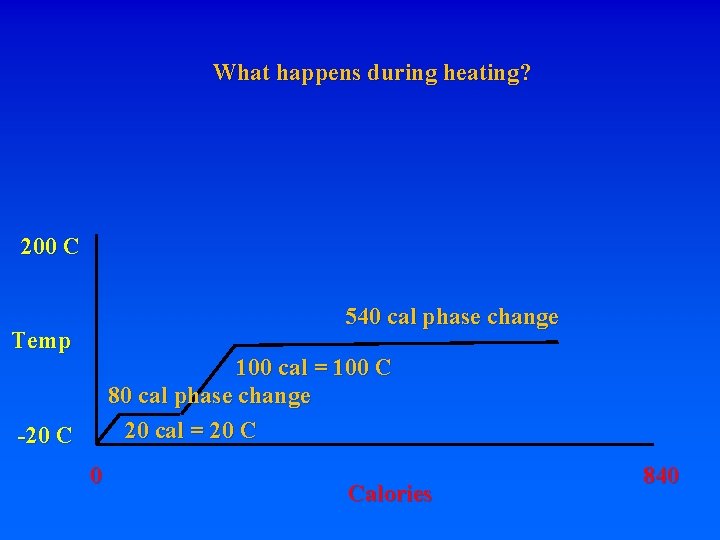
What happens during heating? 200 C 540 cal phase change Temp 100 cal = 100 C 80 cal phase change 20 cal = 20 C -20 C 0 Calories 840
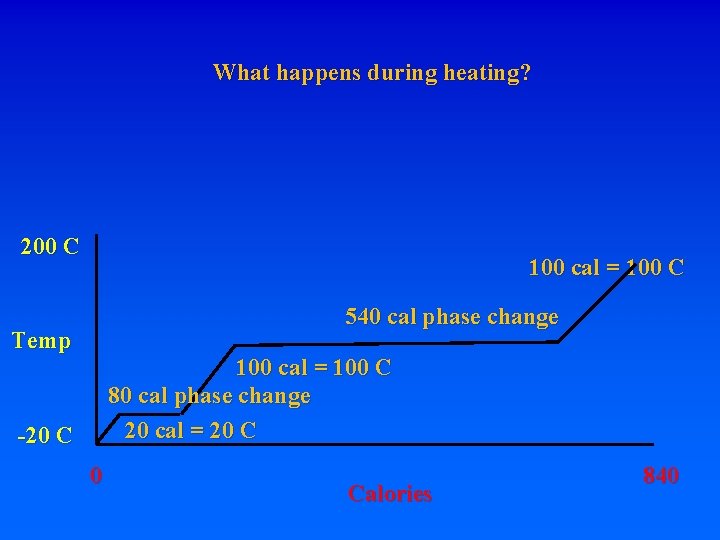
What happens during heating? 200 C 100 cal = 100 C 540 cal phase change Temp 100 cal = 100 C 80 cal phase change 20 cal = 20 C -20 C 0 Calories 840
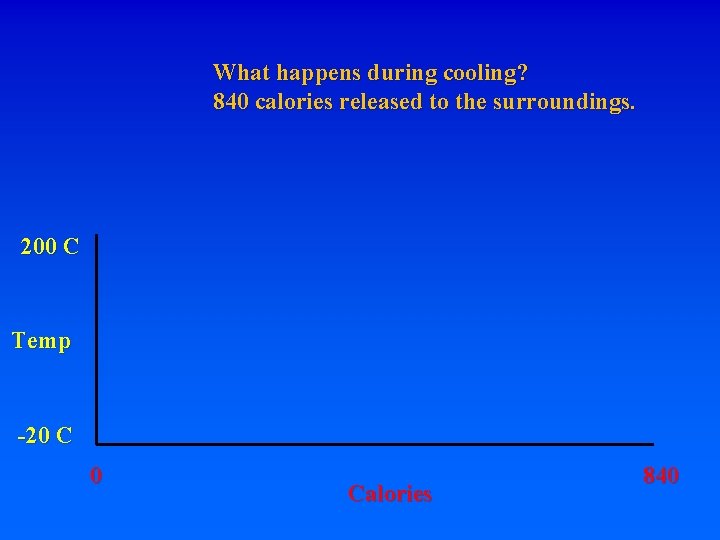
What happens during cooling? 840 calories released to the surroundings. 200 C Temp -20 C 0 Calories 840
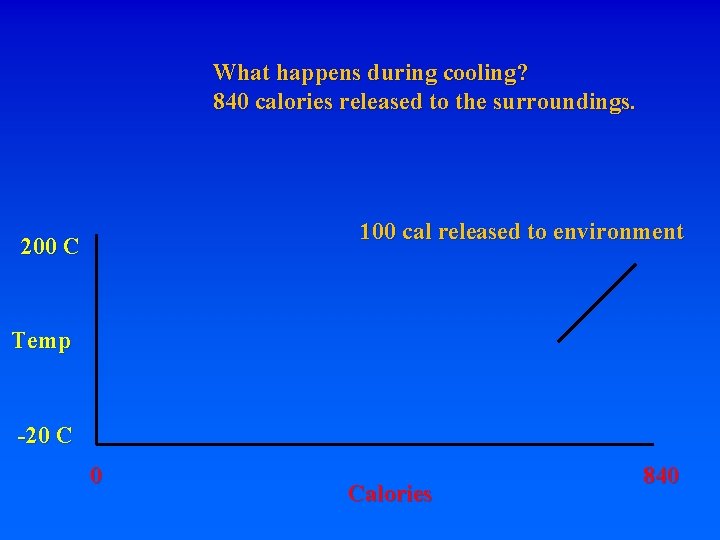
What happens during cooling? 840 calories released to the surroundings. 100 cal released to environment 200 C Temp -20 C 0 Calories 840
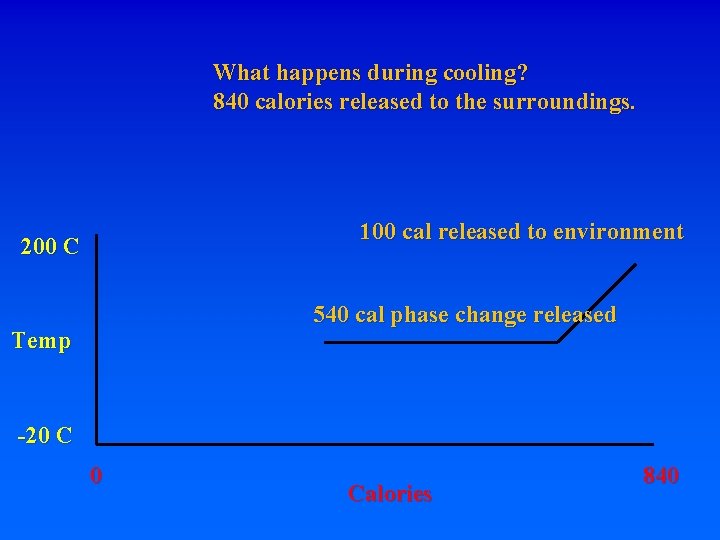
What happens during cooling? 840 calories released to the surroundings. 100 cal released to environment 200 C 540 cal phase change released Temp -20 C 0 Calories 840
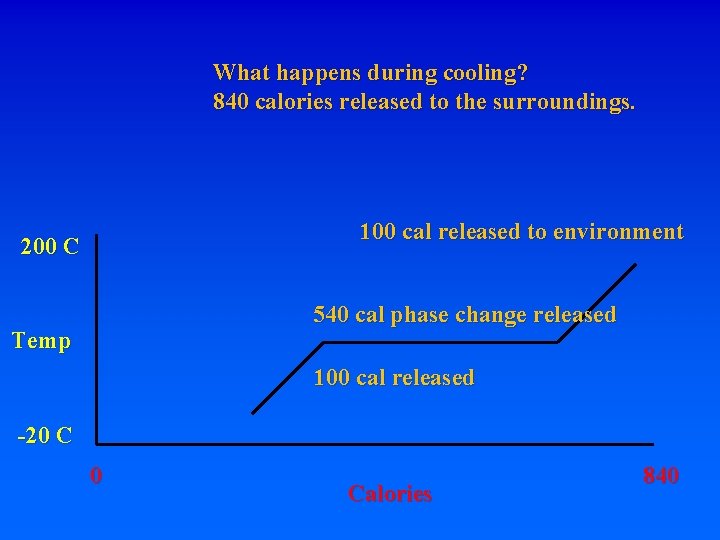
What happens during cooling? 840 calories released to the surroundings. 100 cal released to environment 200 C 540 cal phase change released Temp 100 cal released -20 C 0 Calories 840
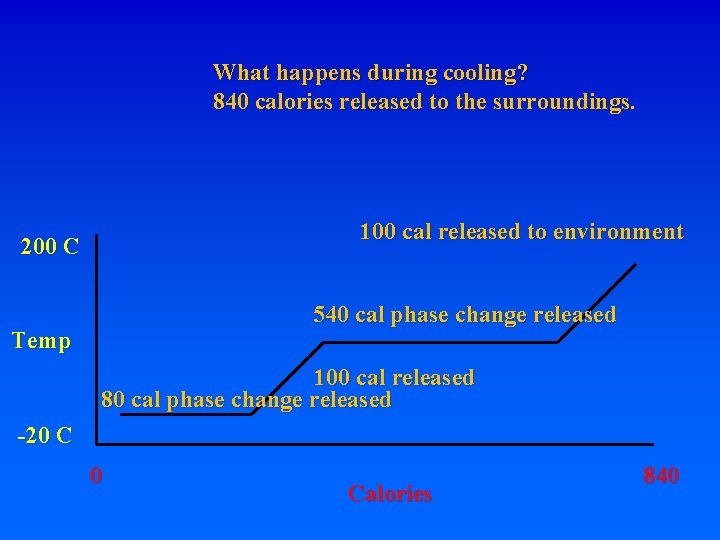
What happens during cooling? 840 calories released to the surroundings. 100 cal released to environment 200 C 540 cal phase change released Temp 100 cal released 80 cal phase change released -20 C 0 Calories 840
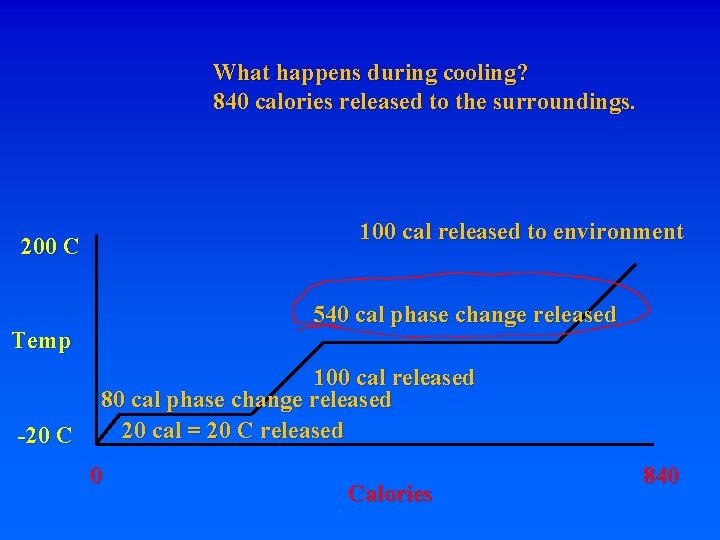
What happens during cooling? 840 calories released to the surroundings. 100 cal released to environment 200 C 540 cal phase change released Temp -20 C 100 cal released 80 cal phase change released 20 cal = 20 C released 0 Calories 840
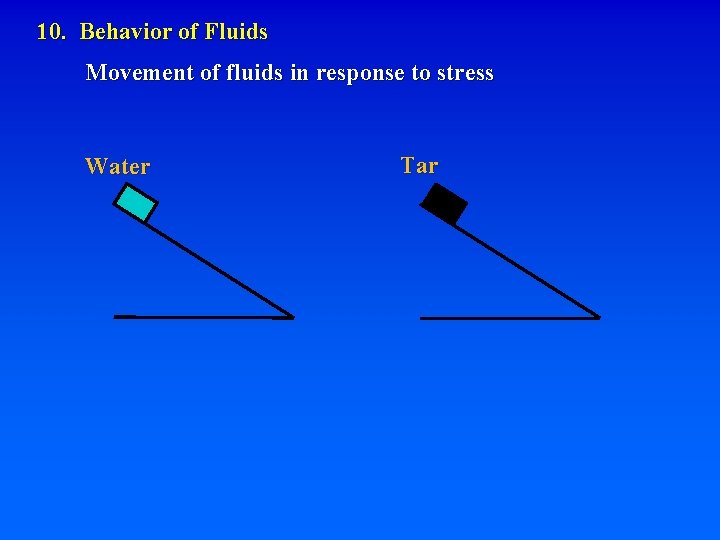
10. Behavior of Fluids Movement of fluids in response to stress Water Tar
- Slides: 59