6 Colour 11 Color has been described here






6





Colour 11 Color has been described here with more relevance to water. In water the colour may occur for any one or more reasons. It may be due to (i) The leaves and peat may add tannin, glucosides and their derivatives to water resulting in a yellow or brown hue. e. g. , :

12 (1) Yellow or brown hue in water may be due to tannin, glucosides and their derivatives (drawn) from coloured organic substances originating in the decay or aqueous extracts of natural vegetation. (ii) Extracts in soil and plants, e. g. , (2) Colour caused by iron, manganese and copper ions in the water. (iii) Coloured industrial discharges, e. g. , Textile wastes, pulp and paper etc. In water and wastewater colour can be true or apparent colour as distinguished here under

13 Apparent colour: Colour measured in water containing suspended matter is defined as apparent colour. True colour: Colour measured in water after filteration or centrifugation is defined as true colour. It is due to the dissolved organics, minerals or chemicals in water as discussed earlier. The term colour is used to mean true colour, that is, the colour of water from which suspended matter (turbidity) has been removed.

14 The term apparent colour includes colour due to dissolved and suspended matter. It is determined on the sample that has not been subjected to filteration or centrifugation. True colour of water sample is, therefore, substantially less than its apparent colour. In industrial wastewaters colour is mainly due to colloidal or suspended matter. In such cases measurement of true and apparent colour becomes necessary.

15 Colour is an indication of visible pollution. The appearance of colour in water is due to absorption of certain wavelengths of light by dissolved, colloidal and suspended matter. Obviously colour of industrial wastewater varies according to the type of industry, e. g. , colour of effluent from textile mill and slaughter house will be different. Knowledge about the cause and measurement of colour is important.

Measurement of Colour. 16 Primary requirement to measure colour is removal of turbidity either by filtration or by centrifugation. Two methods can be used to measure colour in water and wastewater: (a) Visual method: Platinum—Cobalt method (b) Instrumental method The visual i. e. , Platinum—Cobalt method is the simplest as it involves visual comparison of a water sample with a series of standard solutions in tubes or with coloured glass discs.

17 The standard solutions contain known amounts potassium chloroplatinate and cobaltous chloride. The amount of cobaltous chloride are varied to match the normal yellow or brown hue of the test water at a given water location. This method is called Platinum— Cobalt Standard method. This method is best suited to measure the colour of potable water and of water in which colour is due to naturally occurring material.

18 It is not applicable to most highly coloured industrial wastewaters or water polluted by them. This is so because this method was developed to analyse naturally coloured water. Other methods, such as spectrophotometry, are employed to measure colours originating from industrial wastewater or coloured minerals. The failure to compare colour of public water supply with standard colour solutions is, quite often, indicative of industrial pollution.

The Color unit: 19 Using visual method, colour is measured and expressed as True Color Units (TCU) or Platinum— Cobalt Unit which corresponds to color exhibited by a standard solution containing 1. 0 mg of Platinum/L in the form of the Chloroplatinate ion. To match the colour of natural waters, cobalt is added in the ratio of 1: 2 i. e. , 250 mg of Cobalt metal ion: 500 mg of platinum ion. However, this ratio may be varied to match the hue in special cases.

20 Thus, a water sample exhibiting a colour of 20 TCU compares more closely with standard solution containing 20 mg platinum per litre than with Standards containing 19 or 21 mg (platinum)/L. Most consumers can detect a colour of 15 TCU in a glass of water. However, in large volumes of water like bathtubs a colour of 5 TCU is also detectable.

p. H and Colour value: 21 The colour value of water is extremely p. H dependent and hence it must be recorded while measuring colour. The colour value invariably increases as the p. H of water is raised. The increase in the colour value with increasing p. H of the water sample is known as indicator effect. To allow for this effect the colour value and p. H must be recorded together.

22 For a given location and or an individual treatment plant all the colour measurements can be done at standard p. H of 8. 3. Significance of colour in water and wastewater’. Colour communicates differently for water and wastewater as under:

23 (a) Water: (i) Most consumers can detect a colour of 15 TCU in a glass of water. Colour is an aesthetic problem. (ii) A colour of 5 TCU is apparent in a bath tubs. (iii) Aesthetic objective for colour is set at <15 TCU. (iv) Red colour: Presence of Iron. (v) Black: Presence of manganese. (vi) The colour of surface water is predominantly due to organic substances of natural origin.

24 (vii) In general, hard surface waters are less highly coloured than soft water. (viii) The colour of ground water is due to its coloured mineral content.

25 (b) Wastewater: Domestic wastewater (i) Light brown: less than 6 hours old (ii) Light to medium grey: Some degree of decomposition in collection system (iii) Dark grey or Black: Septic—extensive bacterial decomposition under anaerobic conditions. Black colour is due to formation of various sulphides particularly ferrous sulphide.

26 (c) Industrial wastewater: Depends primarily on the type of industry, raw materials and the final product: Difficult to generalize but usually the coloured industrial wastewaters create curiosity about its pollution strength and toxicity.

3. 4. 1 Color 27 The true color of a sample of water or wastewater is primarily attributed to dissolved and colloidal substances. Apparent color is due to suspended substances that can be removed by settling and filtration. Color associated with dissolved and colloidal substances, however, can only be removed by adding a chemical that causes the substance to be precipitated or coagulated.

28 Powerful oxidizing agents, such as chlorine and ozone, can be used to oxidize color compounds into innocuous end-products. Most surface waters contain organic or vegetable extracts from the decomposition of leaves, pine needles, and lignin. In some cases, the water may appear dark brown even black, if the water originated from a swamp.

29 Not only is colored water aesthetically displeasing to consumers, but many organic compounds, such as humic and fulvic acids, will be converted to trihalomethanes (THMs) during chlorination of water. Trihalomethanes are suspected carcinogens, and the United States Environmental Protection Agency (EPA) has established a maximum contaminant level (MCL) of 0. 08 mg/L for total trihalomethanes (TTHMs).

30 When brought to the surface, groundwater that contains iron may exhibit a reddish-brown color due to oxidation of reduced iron (Fe 2+) to ferric iron (Fe 3+). Manganese in its reduced form (Mn 2+) will also be oxidized to Mn 3+ when groundwater is pumped from a well to the surface. Domestic wastewater generally appears to be dark brown to black. Many industrial wastewaters, such as textile, and pulp and paper mill wastes, will be highly colored.

31 Conventional wastewater treatment methods will not remove the contaminants causing color in these types of wastewater. Advanced treatment using activated carbon adsorption or advanced oxidation methods must be used. Typically, color is determined by visual comparison with a known standard solution of potassium chloroplatinate or calibrated color disks. The color produced by 1 mg/L of platinum (as K 2 Pt. Cl 6) serves as the standard unit of color.

32 A stock solution of potassium dichloroplatinate, containing 500 mg/L of platinum, is made. From this stock solution, color standards ranging from 5 -70 color units are prepared by diluting the stock with distilled water. The color value of water is p. H dependent, so the p. H should be measured and noted when reporting color.

33 An alternative method for measuring color of a filtered or centrifuged sample is to measure the transmittance at various wavelengths. The procedures for these and other methods for color determination are found in Standard Methods (1998).

(2) Colour l Color is the perception registered as radiation of various wavelengths strikes the retina of the eye. l This color may be objectionable not for health reasons but for aesthetics. l The presence of colour in water is not objectionable from health point of view, l but may spoil the colour of the clothes being washed in such waters, and is also objectionable from aesthetic and psycological point of view, l as people may not like to drink coloured waters. 34

l Stringent color limits are required for water use in many industries l beverage production, dairy and food processing, paper manufacturing, and textiles. l The colour in water can be easily detected by the naked eye. l Dissolved organic matter from decaying vegetation or some inorganic materials, such as coloured soils, etc. , may impart colour to the water. l The excessive growth of algae and aquatic microorganisms may also sometimes impart colour to the water. 35

l The color of a water is an indication of the organic content, including humic and fulvic acids, the presence of natural metallic ions such as iron and manganese, and turbidity. l Hues in water may result from l - natural minerals, such as iron and manganese, l - vegetable origins-humus material and tannins, l - or colored wastes discharged from a variety of industries including mining, refining, pulp and paper, chemicals, and food processing. 36

�Apparent color is measured on unfiltered samples and true color is measured in filtered samples (0. 45 -µm filter). �Turbidity increases the apparent color of water, while the true color is caused by dissolved species and is used to define the aesthetic quality of water. �The color of potable waters is typically assessed by visually comparing a water sample to known color solutions made from serial dilutions or concentrations of a standard platinum-cobalt solution. 37

l The unit of measurement of color is the platinum in potassium chloroplatinate (K 2 Pt. Cl 6). l One milligram per liter of Pt in K 2 Pt. Cl 6 is one unit of color. l The yellow-brownish hue produced by these metals in solution is similar to that found in natural waters. l The presence of color is reported in color units (c. u. ) at the p. H of the solution. 38

l Before testing the color of the water, first of all total suspended matter should be removed from the water by centrifugal force in a special apparatus. l After this the colour of the water is compared with standard colour solution or colour discs. l Standard color solutions are composed of potassium chloroplatinate (K 2 Pt. Cl 6) tinted with small amounts of cobalt chloride. 39

l The color produced by 1 mg/l of platinum in combination with 1/2 mg/l of metallic cobalt is taken as 1 standard color unit. l Comparison tubes containing standard platinum -cobalt solutions ranging from 0 to 70 color units are used for visual measurements; l however, laboratories often employ a colorimeter for readings. 40

l It can be measured by comparing the colour of water sample with other standard glass tubes (called Nessler tubes) containing solutions of different standard colour intensities. l The standard unit of colour is that which is produced by one milligram of platinum cobalt dissolved is one litre of distilled water. l Different standard colour intensities, representing as 1 ppm, 2 ppm, 3 ppm, . . . can thus be easily prepared by dissolving 1 mg, 2 mg, 3 mg. . . respectively of platinum cobalt in 1 litre of water. 41

�In water treatment, one of the difficulties with the comparison method is that at low levels of color it is difficult to differentiate between low values (e. g. , 2 versus 5 c. u. ). �If the water sample contains constituents (e. g. , industrial wastes) that produce unusual colors or hues that do not match the platinum-cobalt standards, then instrumental methods must be used. �Instrumental methods are used to determine (1) the hue (red, green, yellow, etc. ), (2) the luminance (brightness), and (3) the saturation (pale, deep, etc. ) of a solution. 42

l In turn, these three parameters can be related to the chromaticity. It should be noted that the results obtained with the two methods are not comparable. l For precise determination of small colour intensities, compact instrument properly lighted from inside, called a tintometer, is generally used. l The instrument contains an eye piece with two holes. l A slide of standard coloured water is seen through one hole, and the slide of water to be tested is seen from the other hole. 43

l The standard coloured slide is replaced by another, till a matching is obtained, as is done in a turbidimeter. l The standard colour intensity corresponding to this matching will represent the intensity of colour of the water sample. l For public supplies, the colour number on cobalt scale should not exceed 20, and should be preferably less than 10. 44

l A photoelectric colorimeter or spectrophotometer is used to measure the intensity of color developed in the treated sample, which can be related to the concentration of iron and manganese. l The portable colorimeter in Figure 2 -10 is shown with three sample cells and the optical system consisting of a tungsten-light source, an interchangeable filter module, and a silicon photo-diode detector. l The water sample is prepared by addition of reagents to develop a particular color with an intensity related to the concentration of the test substance. 45

46
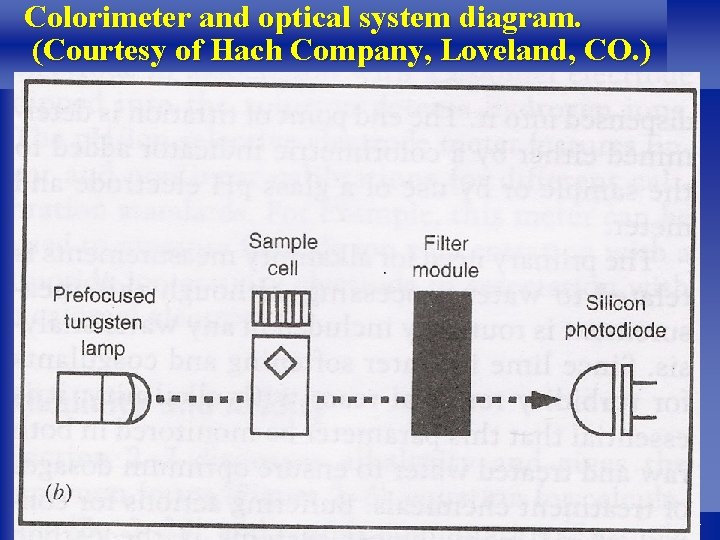
Colorimeter and optical system diagram. (Courtesy of Hach Company, Loveland, CO. ) 47

l The sample cell is placed in the light path of the colorimeter along with a filter module. l The appropriate optical filter provides a monochromatic light beam of the desired wavelength (420 to 810 nm) for the test substance. l The module contains stored calibrations and the electronics so that direct readout of results are displayed on the colorimeter screen. l The most accurate laboratory apparatus used in colorimetric measurements is the spectrophotometer. 48

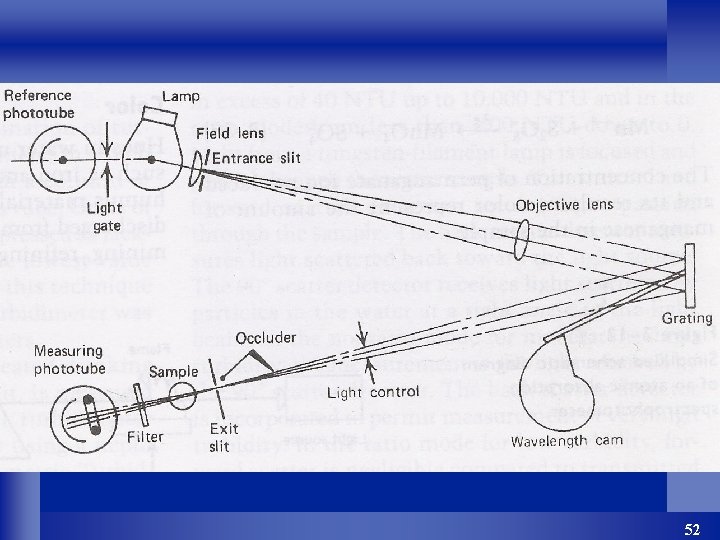
l It operates in a fashion similar to the photoelectric colorimeter, but the monochromatic light is developed by a precise prism or grating system that allows the selection of any wavelength in the visible spectrum. l Figure 2 -11 shows a picture and schematic diagram of a spectrophotometer. 49

�Iron in water contributes to color and varies with p. H and organic particle size. �Color quantification is useful as an aesthetic check on finished drinking waters, and color is regulated under the National Secondary Drinking Water Regulations (secondary standards) �To fulfill these requirements, the U. S. Environmental Protection Agency (USEPA) establishes maximum contaminant levels (MCL). �The secondary MCL for color is 15 color units. 50
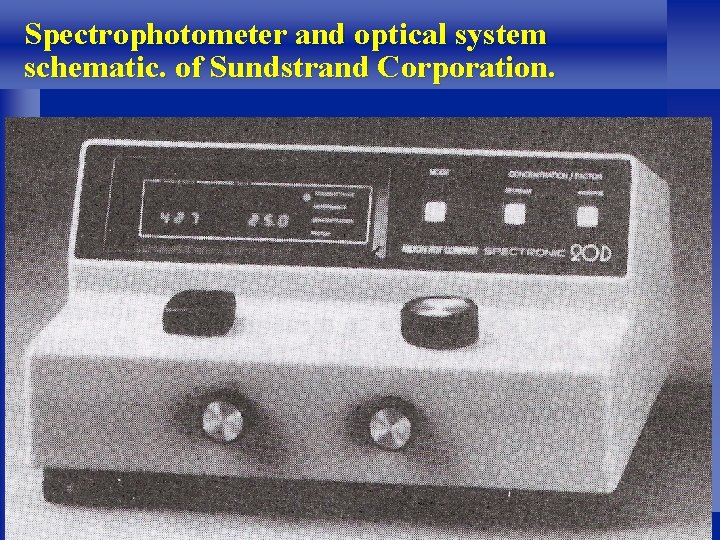
Spectrophotometer and optical system schematic. of Sundstrand Corporation. 51
52
- Slides: 52