6 CHAPTER engel Boles Thermodynamics Entropy A Measure

6 CHAPTER Çengel Boles Thermodynamics Entropy: A Measure of Disorder Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998
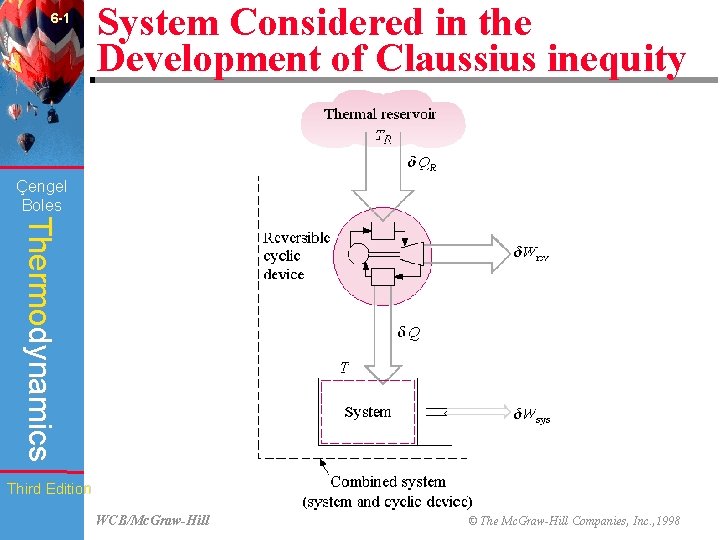
6 -1 System Considered in the Development of Claussius inequity Çengel Boles Thermodynamics Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998
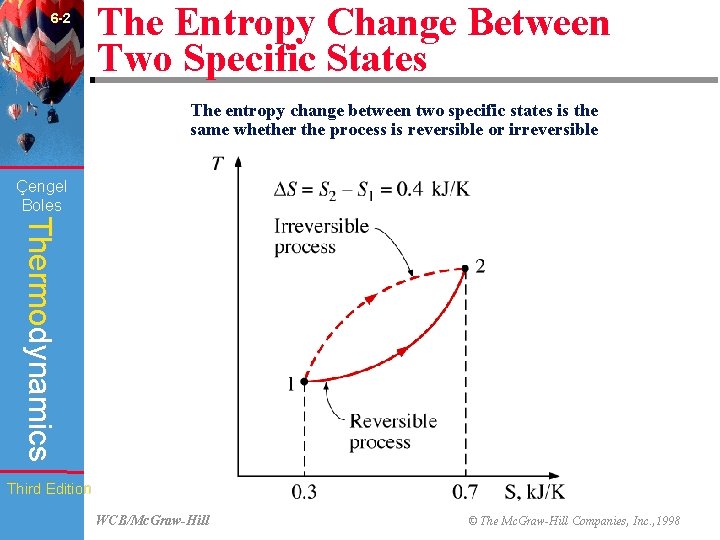
6 -2 The Entropy Change Between Two Specific States The entropy change between two specific states is the same whether the process is reversible or irreversible (Fig. 6 -3) Çengel Boles Thermodynamics Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998
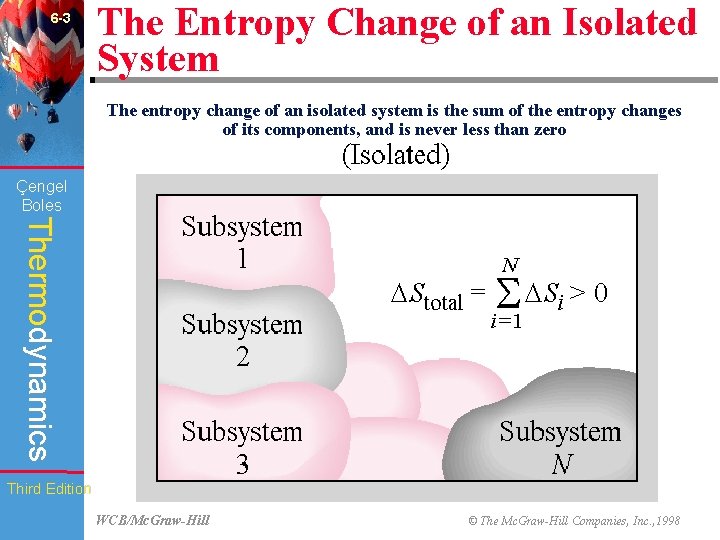
6 -3 The Entropy Change of an Isolated System The entropy change of an isolated system is the sum of the entropy changes of its components, and is never less than zero Çengel Boles Thermodynamics Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998
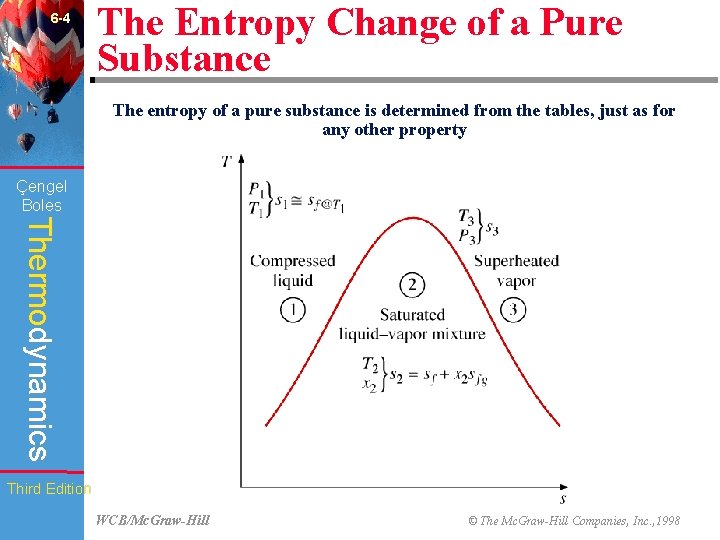
6 -4 The Entropy Change of a Pure Substance The entropy of a pure substance is determined from the tables, just as for any other property (Fig. 6 -10) Çengel Boles Thermodynamics Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998
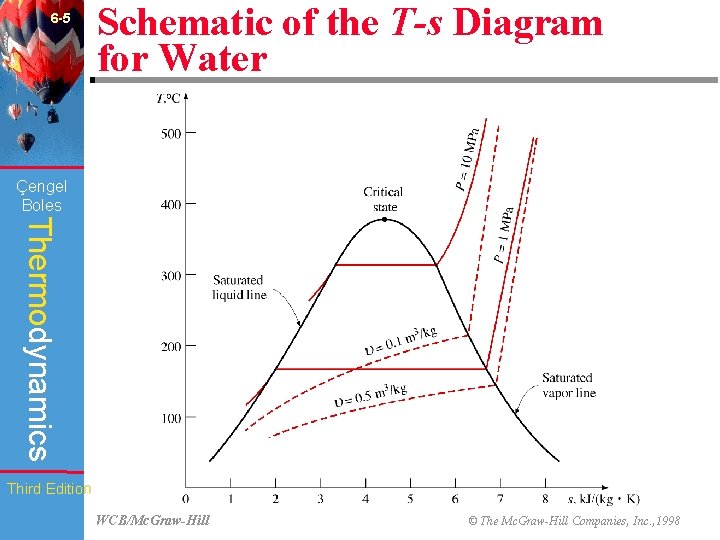
6 -5 Schematic of the T-s Diagram for Water (Fig. 6 -11) Çengel Boles Thermodynamics Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998
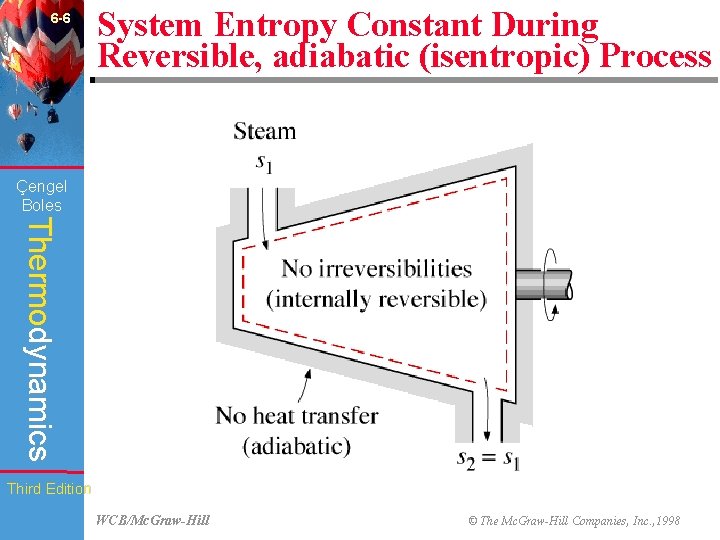
6 -6 System Entropy Constant During Reversible, adiabatic (isentropic) Process (Fig. 6 -14) Çengel Boles Thermodynamics Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998
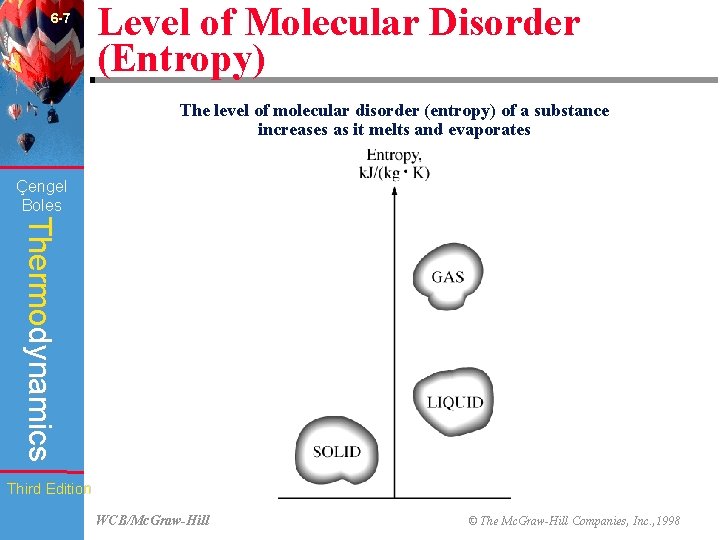
6 -7 Level of Molecular Disorder (Entropy) The level of molecular disorder (entropy) of a substance increases as it melts and evaporates (Fig. 6 -16) Çengel Boles Thermodynamics Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998
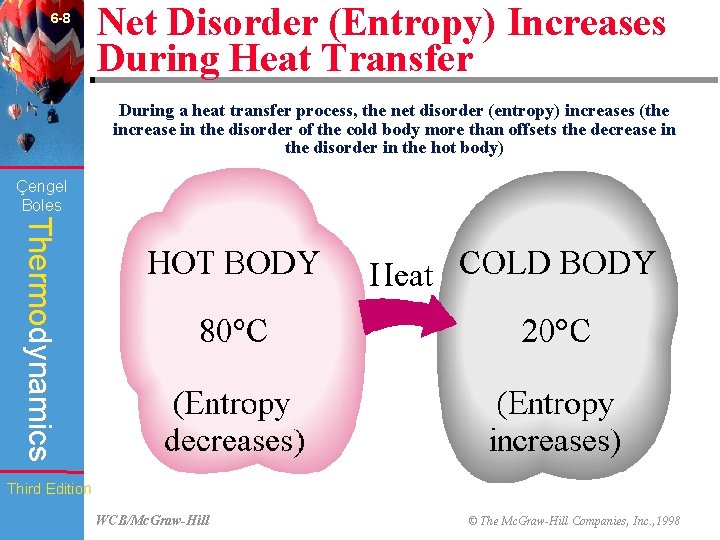
6 -8 Net Disorder (Entropy) Increases During Heat Transfer During a heat transfer process, the net disorder (entropy) increases (the increase in the disorder of the cold body more than offsets the decrease in the disorder in the hot body) Çengel Boles Thermodynamics Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998
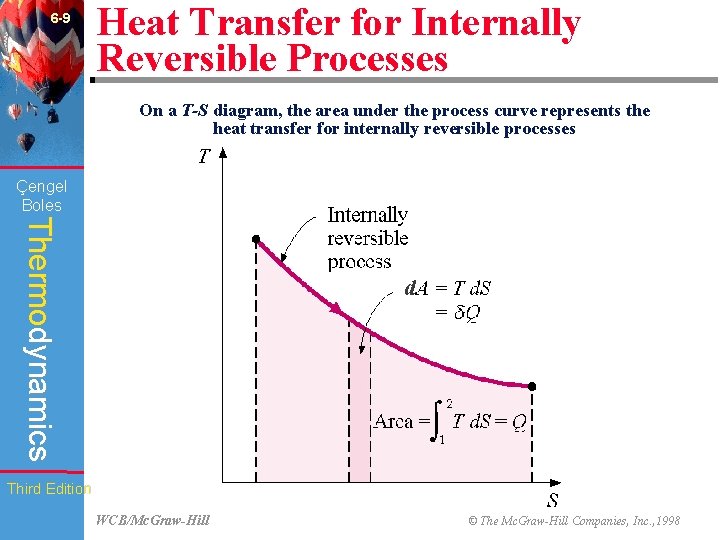
6 -9 Heat Transfer for Internally Reversible Processes On a T-S diagram, the area under the process curve represents the heat transfer for internally reversible processes (Fig. 6 -23) Çengel Boles Thermodynamics d Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998

6 -11 h-s Diagram for Adiabatic Steady. Flow Devices For adiabatic steady-flow devices, the vertical distance ²h on an h-s diagram is a measure of work, and the horizontal distance ²s is a measure of irreversibilities Çengel Boles Thermodynamics Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998
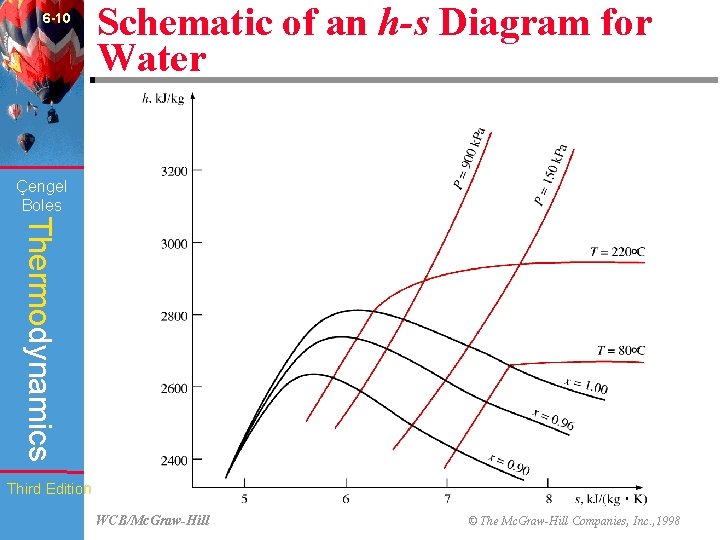
6 -10 Schematic of an h-s Diagram for Water (Fig. 6 -27) Çengel Boles Thermodynamics Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998

6 -12 Entropy of an Ideal Gas The entropy of an ideal gas depends on both T and P. The function s° represents only the temperature-dependent part of entropy (Fig. 6 -33) Çengel Boles Thermodynamics Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998

6 -13 The Isentropic Relations of Ideal Gases The isentropic relations of ideal gases are valid for the isentropic processes of ideal gases only (Fig. 6 -36) Çengel Boles Thermodynamics Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998

6 -14 Using Pr data to Calculate Final Temperature During Isentropic Processes The T-ebow of an ordinary shower serves as the mixing chamber for hot- and cold-water streams. (Fig. 6 -37) Çengel Boles Thermodynamics Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998
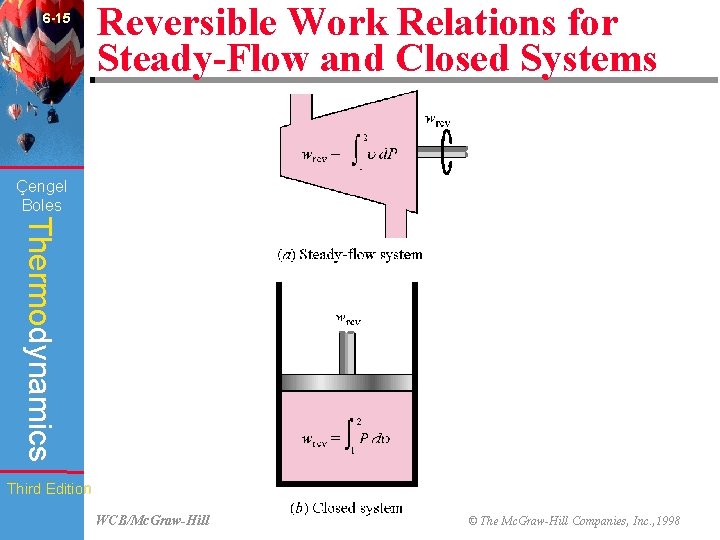
6 -15 Reversible Work Relations for Steady-Flow and Closed Systems (Fig. 6 -41) Çengel Boles Thermodynamics Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998
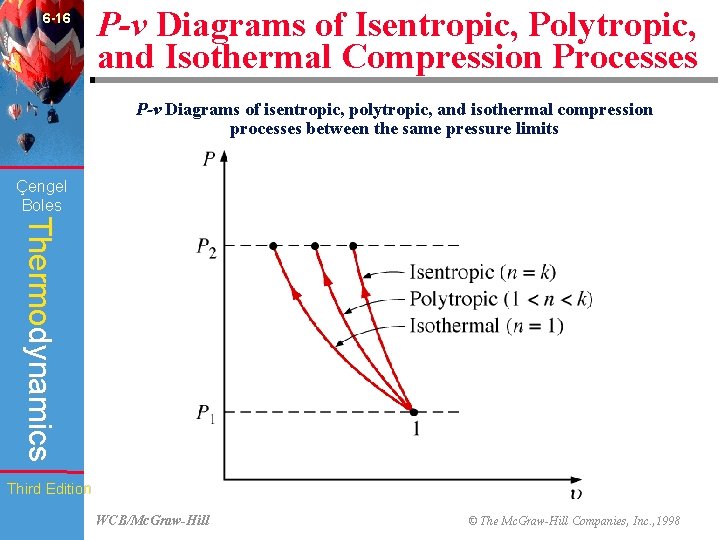
6 -16 P-v Diagrams of Isentropic, Polytropic, and Isothermal Compression Processes P-v Diagrams of isentropic, polytropic, and isothermal compression processes between the same pressure limits (Fig. 6 -45) Çengel Boles Thermodynamics Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998
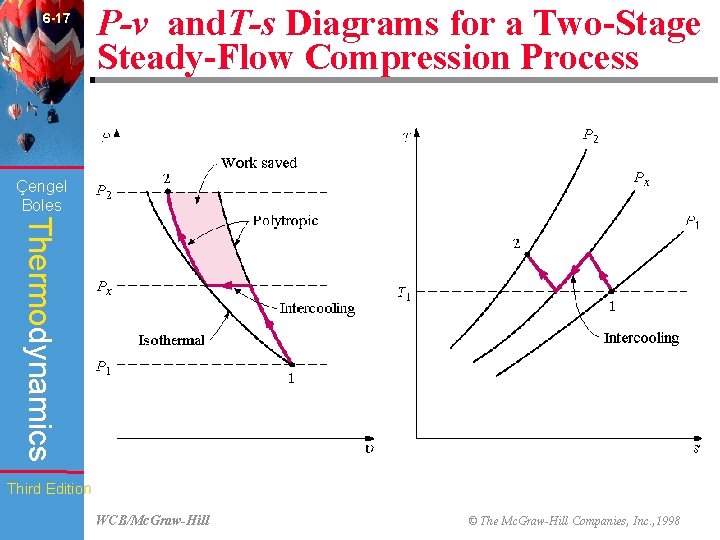
6 -17 P-v and. T-s Diagrams for a Two-Stage Steady-Flow Compression Process (Fig. 6 -46) Çengel Boles Thermodynamics Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998
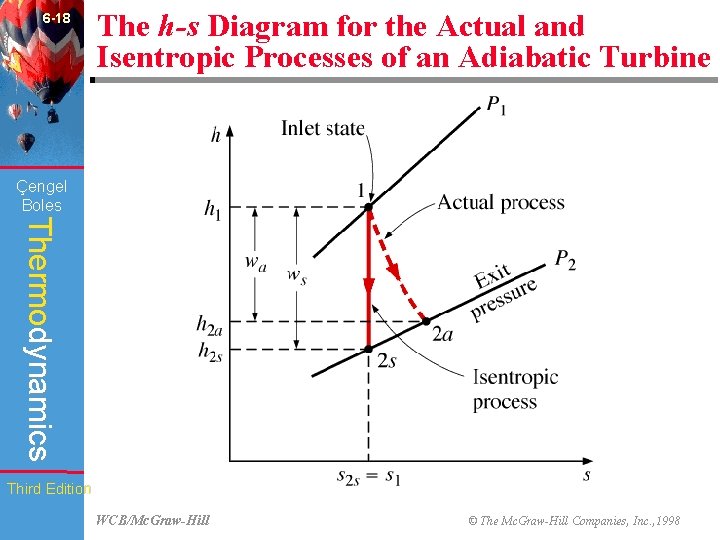
6 -18 The h-s Diagram for the Actual and Isentropic Processes of an Adiabatic Turbine (Fig. 6 -59) Çengel Boles Thermodynamics Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998
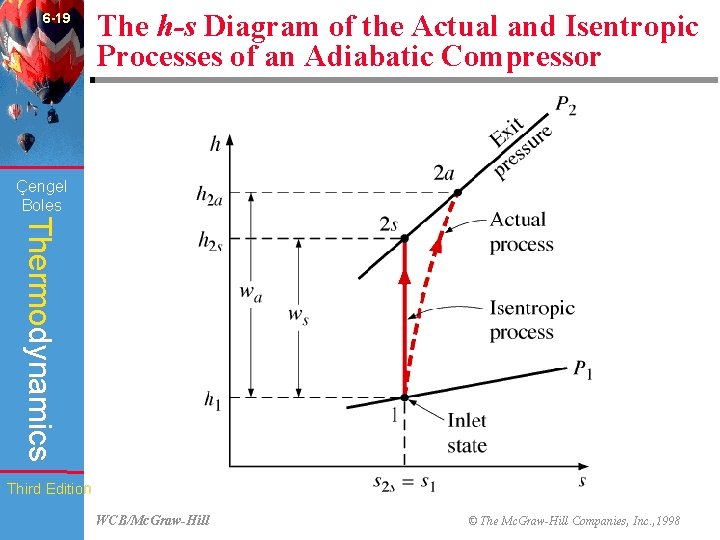
6 -19 The h-s Diagram of the Actual and Isentropic Processes of an Adiabatic Compressor (Fig. 6 -61) Çengel Boles Thermodynamics Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998
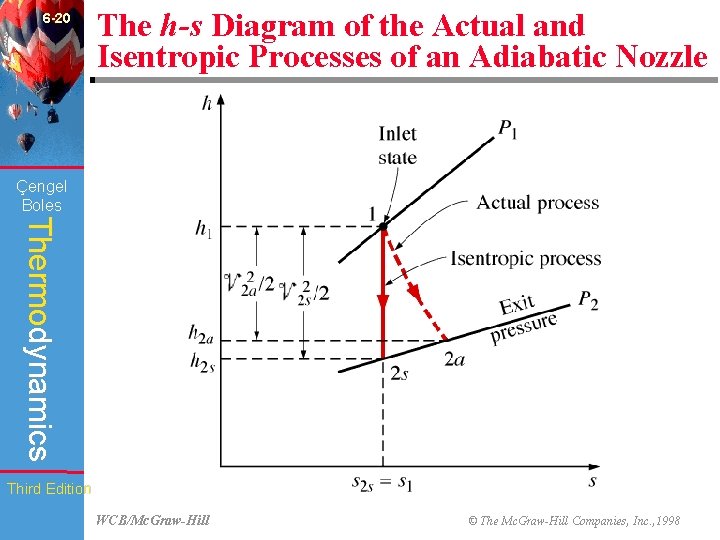
6 -20 The h-s Diagram of the Actual and Isentropic Processes of an Adiabatic Nozzle (Fig. 6 -64) Çengel Boles Thermodynamics Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998
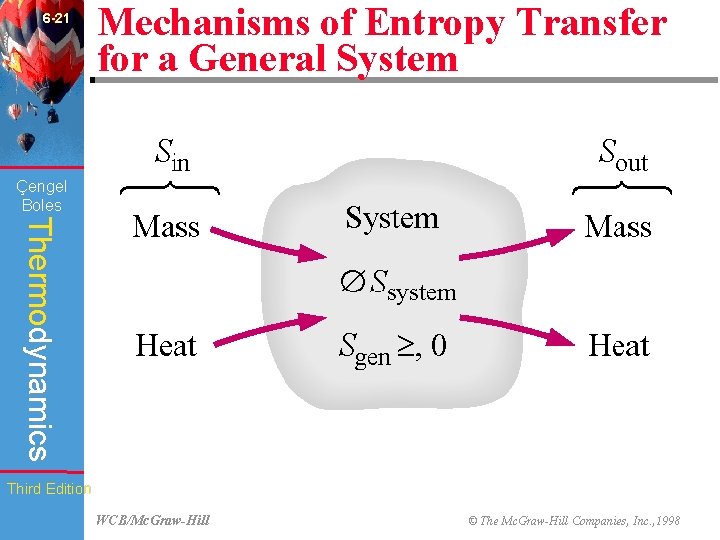
6 -21 Mechanisms of Entropy Transfer for a General System Çengel Boles Thermodynamics Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998
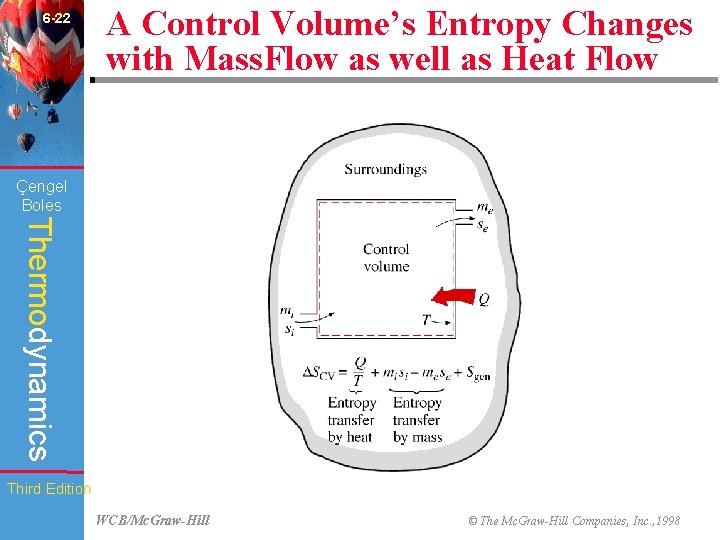
6 -22 A Control Volume’s Entropy Changes with Mass. Flow as well as Heat Flow (Fig. 6 -73) Çengel Boles Thermodynamics Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998
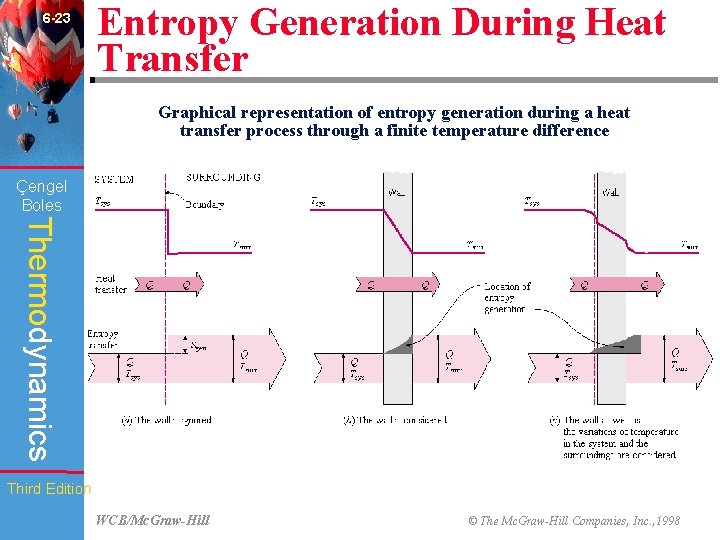
6 -23 Entropy Generation During Heat Transfer Graphical representation of entropy generation during a heat transfer process through a finite temperature difference Çengel Boles Thermodynamics Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998

6 -24 Chapter Summary Çengel Boles Thermodynamics • The second law of thermodynamics leads to the definition of a new property called entropy, which is a quantitative measure of microscopic disorder for a system. Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998

6 -25 Çengel Boles Chapter Summary • The definition of entropy is based on the Clausius inequality, given by Thermodynamics where the equality holds for internally or totally reversible processes and the inequality for irreversible processes. Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998

6 -26 Çengel Boles Chapter Summary Thermodynamics • Any quantity whose cyclic integral is zero is a property, and entropy is defined as Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998

6 -27 Çengel Boles Chapter Summary Thermodynamics • For the special case of an internally reversible, isothermal process, it gives Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998

6 -28 Çengel Boles Chapter Summary Thermodynamics • The inequality part of the Clausius inequality combined with the definition of entropy yields an inequality known as the increase of entropy principle, expressed as where Sgen is the entropy generated during the process. Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998

6 -29 Çengel Boles Chapter Summary Thermodynamics • Entropy change is caused by heat transfer, mass flow, and irreversibilities. Heat transfer to a system increases the entropy, and heat transfer from a system decreases it. The effect of irreversibilities is always to increase the entropy. Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998

6 -30 Çengel Boles Chapter Summary Thermodynamics • Entropy is a property, and it can be expressed in terms of more familiar properties through the Tds relations, expressed as Tds = du +Pdv and Tds = dh - vd. P Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998

6 -31 Çengel Boles Chapter Summary Thermodynamics • These two relations have many uses in thermodynamics and serve as the starting point in developing entropy-change relations for processes. The successful use of Tds relations depends on the availability of property relations. Such relations do not exist for a general pure substance but are available for incompressible substances (solids, liquids) and ideal gases. Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998

6 -32 Chapter Summary • The entropy-change and isentropic relations for a process can be summarized as follows: Çengel Boles Thermodynamics 1. Pure substances: Any process: s = s 2 - s 1 Isentropic process: [k. J/(kg-K)] s 2 = s 1 Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998

6 -33 Chapter Summary • The entropy-change and isentropic relations for a process can be summarized as follows: Çengel Boles Thermodynamics 2. Incompressible substances: Any process: s 2 - s 1 Isentropic process: T 2 = Cav 1 n T 1 [k. J/(kg-K)] T 2 = T 1 Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998

6 -34 Chapter Summary • The entropy-change and isentropic relations for a process can be summarized as follows: Çengel Boles Thermodynamics 3. Ideal gases: a. Constant specific heats (approximate treatment): Any process: T 2 v 2 s 2 - s 1 = Cv, av 1 n T + R 1 n v 1 1 [k. J/(kg-K)] and T 2 P 2 s 2 - s 1 = Cp, av 1 n T + R 1 n P 1 1 [k. J/(kg-K)] Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998

6 -35 Chapter Summary • The entropy-change and isentropic relations for a process can be summarized as follows: Çengel Boles Thermodynamics 3. Ideal gases: a. Constant specific heats (approximate treatment): On a unit-mole basis, T 2 v 2 s 2 - s 1 = Cv, av 1 n T + Ru 1 n v 1 1 [k. J/(kmol-K)] and T 2 P 2 s 2 - s 1 = Cp, av 1 n T + Ru 1 n P 1 1 [k. J/(kmol-K)] Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998

6 -36 Çengel Boles Chapter Summary 3. Ideal gases: a. Constant specific heats (approximate treatment): Isentropic process: Thermodynamics Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998

6 -37 Chapter Summary • The entropy-change and isentropic relations for a process can be summarized as follows: Çengel Boles Thermodynamics 3. Ideal gases: b. Variable specific heats (exact treatment): Any process, s 2 - s 1 = s 2 o- s 1 o- R 1 n P 2 P 1 [k. J/(kg-K)] or P 2 s 2 - s 1 = s 2 - s 1 - Ru 1 n P 1 o o [k. J/(kmol-K)] Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998

6 -38 Çengel Boles Thermodynamics Third Edition Chapter Summary 3. Ideal gases: b. Variable specific heats (exact treatment): Isentropic process, P s 2 o= s 1 o - R 1 n 2 [k. J/(kg-K)] P 1 where Pr is the relative pressure and vr is the relative specific volume. The function so depends on temperature only. WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998

6 -39 Çengel Boles Chapter Summary • The steady-flow work for a reversible process can be expressed in terms of the fluid properties as Thermodynamics Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998

6 -40 Çengel Boles Chapter Summary Thermodynamics • For incompressible substances (v = constant) steady-flow work for a reversible process simplifies to Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998

6 -41 Çengel Boles Chapter Summary Thermodynamics • The work done during a steady-flow process is proportional to the specific volume. Therefore, v should be kept as small as possible during a compression process to minimize the work input and as large as possible during an expansion process to maximize the work output. Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998

6 -42 Çengel Boles Chapter Summary Thermodynamics • The reversible work inputs to a compressor compressing an ideal gas from T 1, P 1, to P 2 in an isentropic (Pvk = constant), polytropic (Pvn = constant), or isothermal (Pv = constant) manner, are determined by integration for each case with the following results: Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998

6 -43 Chapter Summary • Isentropic: Çengel Boles Thermodynamics (k. J/kg) Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998

6 -44 Chapter Summary • Polytropic: Çengel Boles Thermodynamics (k. J/kg) Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998

6 -45 Chapter Summary • Isothermal: Çengel Boles Thermodynamics (k. J/kg) Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998

6 -46 Chapter Summary Çengel Boles Thermodynamics • The work input to a compressor can be reduced by using multistage compression with intercooling. For maximum savings from the work input, the pressure ratio across each stage of the compressor must be the same. Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998

6 -47 Chapter Summary Çengel Boles Thermodynamics • Most steady-flow devices operate under adiabatic conditions, and the ideal process for these devices is the isentropic process. Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998

6 -48 Chapter Summary • The parameter that describes how efficiently a device approximates a corresponding isentropic device is called isentropic or adiabatic efficiency. It is expressed for turbines, compressors, and nozzles as follows: Çengel Boles Thermodynamics Actual turbine work wa ~ h 1 - h 2 a = = = Isentropic turbine work ws h 1 - h 2 s Isentropic compressor work ws ~ h 2 s - h 1 = = = Actual compressor work wa h 2 a - h 1 2 Actual KE at nozzle exit V 2 a ~ h 1 - h 2 a = = 2 = Isentropic KE at nozzle exit h 1 - h 2 s V 2 s In the relations above, h 2 a and h 2 s are the enthalpy values at the exit state for actual and isentropic processes, respectively. Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998

6 -49 Çengel Boles Chapter Summary Thermodynamics • The entropy balance for any system undergoing any process can be expressed in the general form as Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998
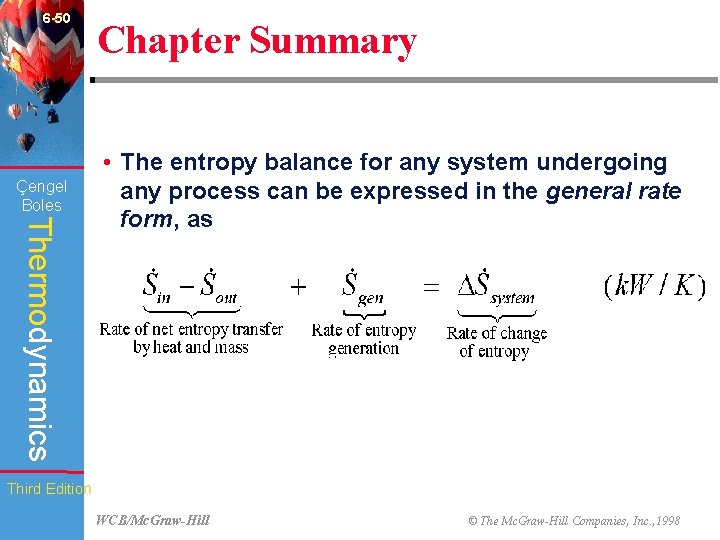
6 -50 Çengel Boles Chapter Summary Thermodynamics • The entropy balance for any system undergoing any process can be expressed in the general rate form, as Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998

6 -51 Çengel Boles Chapter Summary • For a general steady-flow process the entropy balance simplifies to Thermodynamics Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998
- Slides: 52