6 7 Electronegativity and Bond Polarity The electronegativity

6. 7 Electronegativity and Bond Polarity The electronegativity values of representative elements in Group 1 A (1) to Group 7 A (17). Learning Goal Use electronegativity to determine the polarity of a bond. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Electronegativity The electronegativity of an atom is its ability to attract the shared electrons in a bond. It • increases from left to right going across a period on the periodic table. • increases from the bottom to the top of the periodic table. • is high for the nonmetals, with fluorine as the highest. • is low for the metals. Core Chemistry Skill Using Electronegativity General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Electronegativity and the Periodic Table General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.
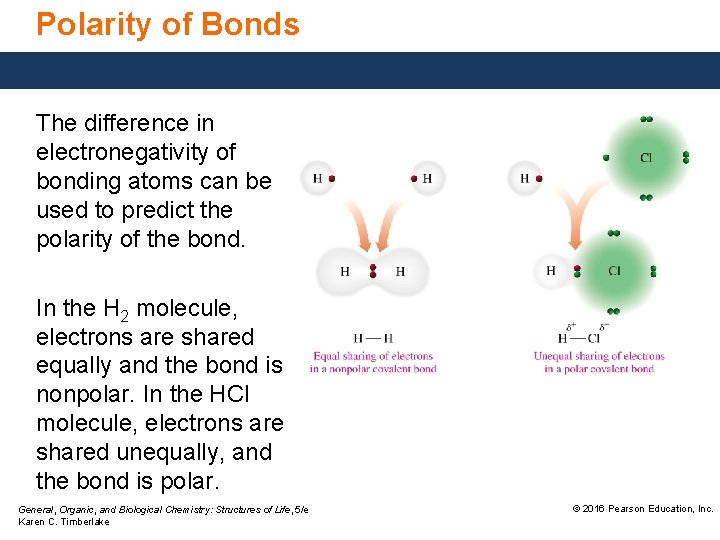
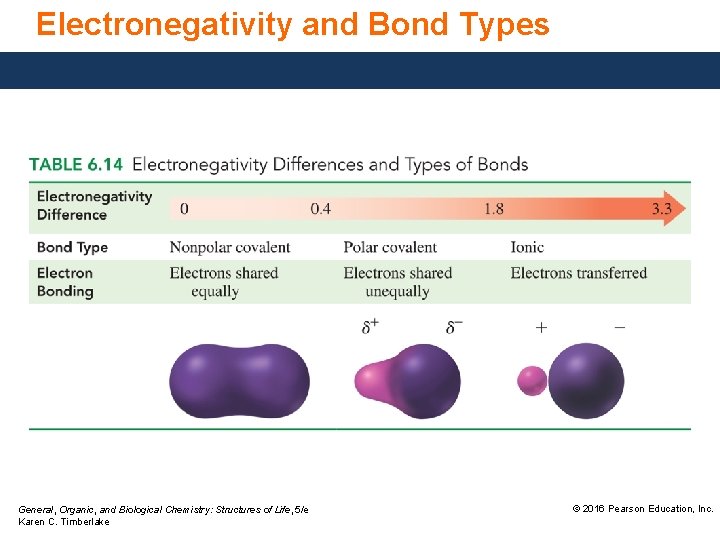
Polarity of Bonds The difference in electronegativity of bonding atoms can be used to predict the polarity of the bond. In the H 2 molecule, electrons are shared equally and the bond is nonpolar. In the HCl molecule, electrons are shared unequally, and the bond is polar. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.
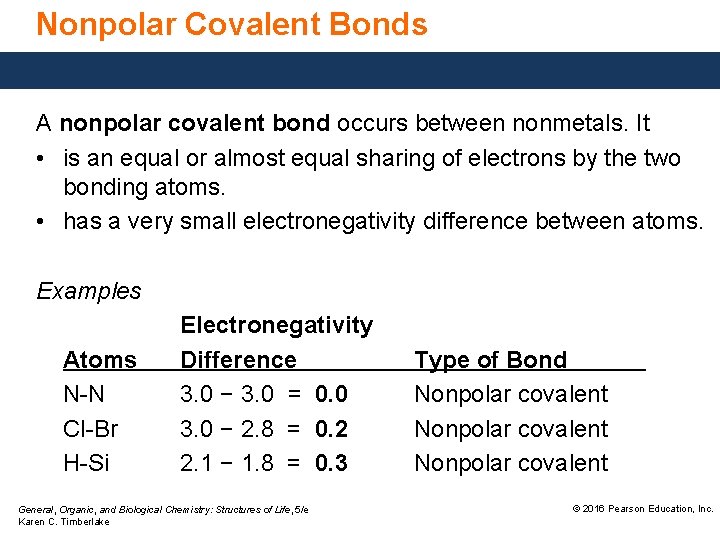
Nonpolar Covalent Bonds A nonpolar covalent bond occurs between nonmetals. It • is an equal or almost equal sharing of electrons by the two bonding atoms. • has a very small electronegativity difference between atoms. Examples Atoms N-N Cl-Br H-Si Electronegativity Difference 3. 0 − 3. 0 = 0. 0 3. 0 − 2. 8 = 0. 2 2. 1 − 1. 8 = 0. 3 General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake Type of Bond Nonpolar covalent © 2016 Pearson Education, Inc.
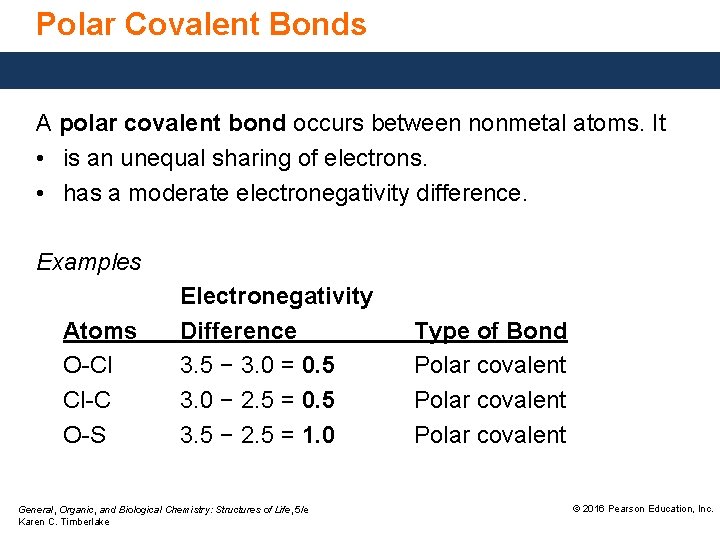
Polar Covalent Bonds A polar covalent bond occurs between nonmetal atoms. It • is an unequal sharing of electrons. • has a moderate electronegativity difference. Examples Atoms O-Cl Cl-C O-S Electronegativity Difference 3. 5 − 3. 0 = 0. 5 3. 0 − 2. 5 = 0. 5 3. 5 − 2. 5 = 1. 0 General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake Type of Bond Polar covalent © 2016 Pearson Education, Inc.

Dipoles and Bond Polarity A polar covalent bond becomes more polar as the difference in electronegativity increases. The separation of charges in a polar bond is called a dipole. The positive and negative ends of the dipole are located by using • the lowercase Greek letter delta with a positive or negative charge. • an arrow that points from the positive to the negative end of the dipole. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.
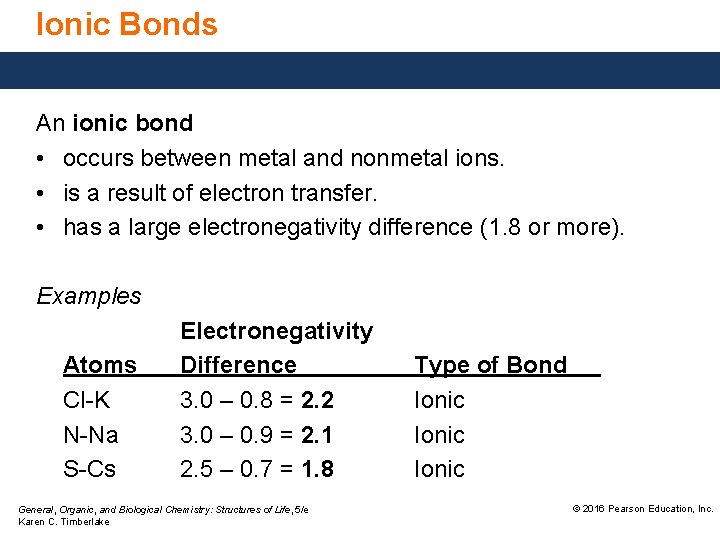
Ionic Bonds An ionic bond • occurs between metal and nonmetal ions. • is a result of electron transfer. • has a large electronegativity difference (1. 8 or more). Examples Atoms Cl-K N-Na S-Cs Electronegativity Difference 3. 0 – 0. 8 = 2. 2 3. 0 – 0. 9 = 2. 1 2. 5 – 0. 7 = 1. 8 General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake Type of Bond Ionic © 2016 Pearson Education, Inc.

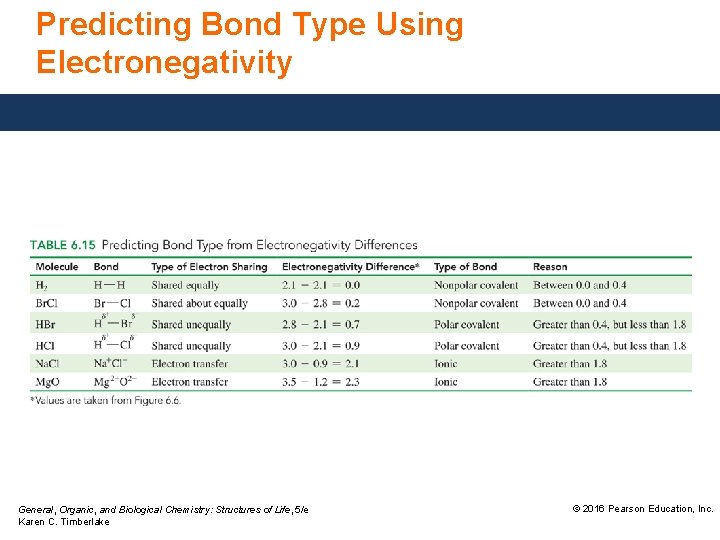
Variations in Bonding The difference in electronegativity values for two atoms can be used to predict the type of chemical bond. If the electronegativity difference is • between 0 and 0. 4, the bond is nonpolar covalent. • between 0. 5 and 1. 8, the bond is polar covalent. • greater than 1. 8, the bond is ionic and the electrons are considered transferred. General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.
Electronegativity and Bond Types General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.
Predicting Bond Type Using Electronegativity General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.

Study Check Use the electronegativity difference to identify the type of bond (nonpolar covalent [NP], polar covalent [P], or ionic [I]) between the following: A. K—N B. N—O C. Cl—Cl D. H—Cl General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.
![Solution Use the electronegativity difference to identify the type of bond (nonpolar covalent [NP], Solution Use the electronegativity difference to identify the type of bond (nonpolar covalent [NP],](http://slidetodoc.com/presentation_image/3855f24ef7c31a9861638133f6aeeab2/image-13.jpg)
Solution Use the electronegativity difference to identify the type of bond (nonpolar covalent [NP], polar covalent [P], or ionic [I]) between the following: Difference Type of bond A. K—N 2. 2 ionic (I) B. N—O 0. 5 polar covalent (P) C. Cl—Cl 0. 0 nonpolar covalent (NP) D. H—Cl 0. 9 polar covalent (P) General, Organic, and Biological Chemistry: Structures of Life, 5/e Karen C. Timberlake © 2016 Pearson Education, Inc.
- Slides: 13