6 6 Single and Double Displacement Synthesis Decomposition
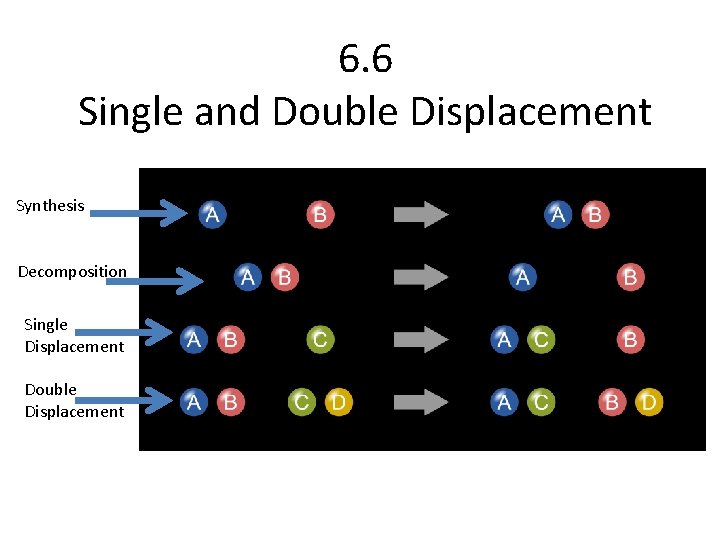
6. 6 Single and Double Displacement Synthesis Decomposition Single Displacement Double Displacement

Single Displacement • Single Displacement: – One element replaces another in a compound – Non-metals replace non-metals – Metals replace metals – Two people are dancing, and someone cuts in…

Single Displacement • Single Displacement: – Be careful! Follow rules of metal replaces metal, and non-metal replaces non-metal – If A is a metal, B is a non-metal……what is C?

Single Displacement • Example 1: Zinc reacts with copper (II) sulphate to make ____ and _____. Word equation: zinc + copper (II) sulphate ? + ? Formula Zn + Cu. SO 4 ___? ____ + __? ___

Single Displacement • Zn + Cu. SO 4 ___? ____ + __? ___ – Zinc will form Zn 2+ – Copper will form Cu 2+ – Sulphate is the complex ion SO 42 - – Can copper and zinc form a compound? ____ – Can sulphate and zinc form a compound? ____ – Why? __________________

Single Displacement Zinc + copper(II) sulphate copper + zinc sulphate Zn + Cu. SO 4 Cu + Zn. SO 4 More examples in page 240 in your textbook

Single Displacement Magnesium reacts with carbon dioxide to form magnesium oxide and carbon • Word equation ____ + _____ • Chem equation ____ + _____ • Balanced equation ____ + _____

Double Displacement • One element in a compound displaces or replaces an element in a compound – Many happen between ionic compounds • Non-metal switches with non-metal

Double Displacement • Ag. NO 3 + Na. Cl
- Slides: 9